7.1: Carbohydrates
- Page ID
- 288509
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- To identify a chemical structure as a carbohydrate
- To classify carbohydrates as monosaccharides, disaccharides, or polysaccharides
Recognizing & Classifying Carbohydrates
Carbohydrates contain only three elements: carbon, hydrogen, and oxygen. They also contain a limited number of functional groups. Each carbohydrate contains several alcohol groups and at least one aldehyde or ketone group. Despite the similarities between carbohydrate molecules, there are still many different carbohydrates due to the wide variety of sizes among carbohydrates. Because some carbohydrates are polymers these biomolecules can be classified as monosaccharides, disaccharides, and polysaccharides.
In addition to being the building blocks of larger carbohydrates, monosaccharides are often found in fruits, cane sugar, and honey. Examples of these molecules, also called simple sugars, include fructose and glucose. Monosaccharides can be classified by the number of carbon atoms (e.g. 3 = tri, 4 = tetra, etc.) and whether they contain an aldehyde or ketone functional group (an aldo- or keto- prefix is used). As you will see in the table below and throughout the carbohydrate section, names of these molecules often end in -ose.
|
Number of Carbon Atoms |
Aldose |
Ketose |
|---|---|---|
|
3 |
aldotriose |
ketotriose |
|
4 |
aldotetrose |
ketotetrose |
|
5 |
aldopentose |
ketopentose |
|
6 |
aldohexose |
ketohexose |
|
7 |
aldoheptose |
ketoheptose |
The images shown are called Fischer projections. They are similar to the skeletal structures introduced in the Organic Chemistry chapter, but also provide information about chirality. At each intersection of the lines there is a carbon atom; all other atoms are shown explicitly. The L- and D- forms of glucose are two different isomers (enantiomers).
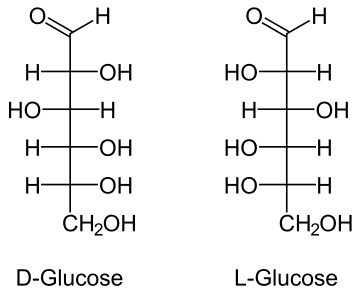
Each monosaccharide has several chiral carbon atoms. (Can you identify them in the examples above? Remember that a chiral center is a carbon atom with four different groups attached.) To distinguish between different forms prefixes L- and D- are used, for example L-glucose and D-glucose. Interestingly, most monosaccharides found in nature are the D- form. Monosaccharides often exist in a cyclic form rather than the straight chain forms. The cyclic form of D-glucose is shown below.

The ring is formed by a reaction that forms a single bond between the oxygen on carbon 5 and carbon 1 (where the aldehyde group was). All of the same atoms are present, but the hydrogen atom that had been part of the hydroxyl (-OH) group on carbon 5 is now bonded to the oxygen atom on carbon 1 (formally the carbonyl oxygen).
Which of the following are monosaccharides? Choose all that apply.




- Answer
-
Structures 2, 3, and 4 are monosaccharides. Carbohydrates have the elements C, H, and O with approximately equal numbers of C and O. Almost every carbon atom has an OH group. Remember that the carboxylic acid group in Structure 1 is not the same as the alcohol functional group. Monosaccharides may be either cyclic or acyclic.
Disaccharides
As the name implies, disaccharides are carbohydrates made from two monosaccharides. A common example is lactose which contains the monomers glucose and galactose.

Oligosaccharides
Oligosaccharides are moderately large carbohydrates consisting of 3 - 10 monosaccharides bonded together. Each monosaccharide is in its cyclic form and the rings are bonded together by an oxygen atom with two single bonds: -O-. Examples of oligosaccharides include the A and B antigens on red blood cells that determine blood type.

Figure \(\PageIndex{1}\): Oligosaccharides that form antigens and result in type A, B, AB, and O blood.
Polysaccharides
Polysaccharides are natural polymers consisting of many monosaccharide units. Examples include starch, cellulose, and glycogen. Starch is a complex carbohydrate found in potatoes and other foods. Cellulose gives structure to cell walls in plants and cannot be digested by humans because we cannot break the bond between the monosaccharide units. Glycogen is a polysaccharide produced in the body to store excess glucose for later use.
Amylose, a starch, is a polymer made from glucose monomers. Notice that the figure below has a glucose monomer in brackets with a subscript of 300-600 indicating that there are hundreds of glucose monomers in a chain of amylose.
![Amylose, a polysaccharide consisting of hundreds of glucose units (designated by [glucose]_300-600)](https://chem.libretexts.org/@api/deki/files/408609/Screen_Shot_2022-06-06_at_11.58.07_AM.png?revision=1&size=bestfit&width=583&height=230)
Classify the following carbohydrate as a monosaccharide, disaccharide, or oligosaccharide.



- Answer
-
- Structure 1 is a disaccharide because it has two monosaccharides bonded together (each of the rings is a monosaccharide).
- Structure 2 is a monosaccharide it is a carbohydrate consisting of only one ring.
- Structure 3 is an oligosaccharide because it has more than two monosaccharides bonded together.
Summary
- Carbohydrates generally contain carbon, hydrogen, and oxygen with an approximately equal number of carbon and oxygen atoms. They have many hydroxy groups (OH).
- Monosaccharides may be cyclic or acyclic. If cyclic, they contain only one ring. Monosaccharides have either an aldehyde or a ketone functional group in addition to many hydroxy groups.
- Disaccharides consist of two monosaccharides bonded together. They have two rings connected by an oxygen atom with two single bonds.
- Oligosaccharides and polysaccharides contain many monosaccharides bonded together. Each monosaccharide is a ring, typically of 5 - 6 atoms, and the rings are bonded together by the same -O- pattern found in disaccharides.

