1.6: All About the Elements
- Page ID
- 204159
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Basic Arrangement of Periodic Table
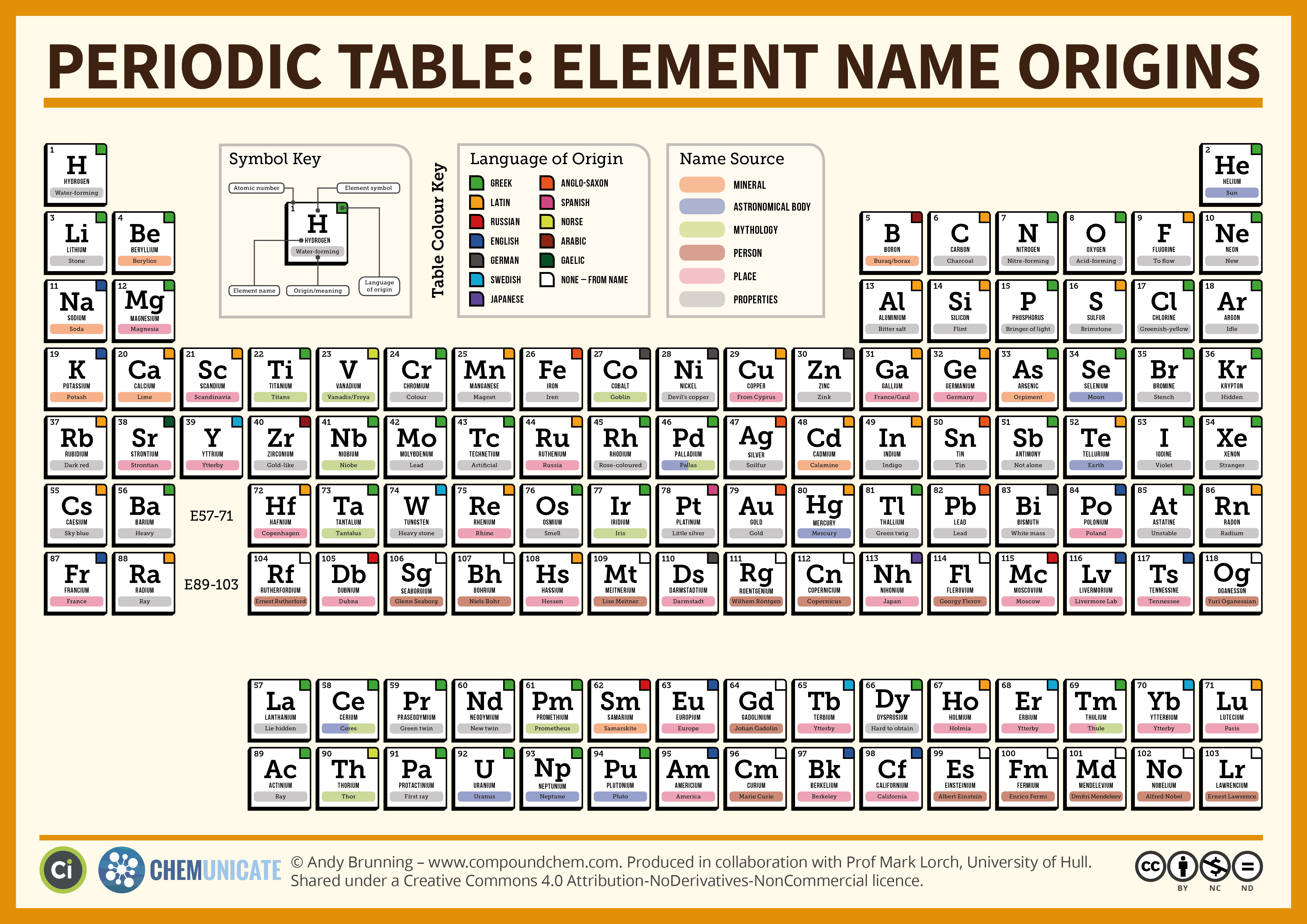
In Chapter 3, we will delve more into the initial construction and modifications of today's periodic table. For now, you need to be aware that the periodic table has columns (known as families/groups). Horizontal rows are called periods. On the table below, the element symbols are either one of two letters. Previously, some of the newly discovered elements (see period seven on other tables) used three letter symbols. These three letter symbols corresponded to a Latin numbering system until the elements were given official names.
Languages of Element Names
Interestingly, different languages are used on the periodic table. Depending on your background, you might be more familiar with some elemental symbols than others. For example, Hydrogen is represented by the symbol H. This element's name is derived from the Greek hydro, and genes which means to generate water. Other elemental symbols like copper (which has a symbol of Cu) are not so predictable. This particular element's name originated from the Latin cuprum (from the island of Cyprus). A variety of languages are displayed on the periodic table. For example, German roots are present in the element Tungsten. The symbol for this element is W which corresponds to the German word, wofram. . For this particular class, you will find a list of required elements/symbols that need to be memorized. Please locate this document on the moodle page for this course.
Meanings of Element Names
Many elements are named in honor of people. Some familiar names you might see are curium (Marie and Pierre Curie), einstenium (Albert Einstein), and medelelvium (Dimitri Mendeleev). IUPAC (International Union of Pure and Applied Chemistry) requires these types of names to be done posthumously. In other words, the scientist who discovered the element could not name it after her/himself or anyone who is still currently living. Over the years, there have two exceptions. The elements of seaborgium (Sg) and oganesson (Og) were named while Glen Seaborg and Yuri Oganessian were still living. Seaborgium was named in honor of an American nuclear chemist who synthesized the element plutonium. He is also credited with assisting the discovery of other elements like c, americium (element 95), curium (element 96), and fermium (element 100), . His accomplishments encouraged his fellow researcher, Albert Ghiorso, to name Seaborgium in honor of him. As for element 118 (oganessian), a joint research commission of Russian and American scientists named this element for Russian physcist, Yuri Oganessian. As of 2017, Oganessian is still living and is responsible for the discovery of super heavy elements. For test 1, please research an element that has not been mentioned on this page or on your list to be memorized. By accessing this link, information about a selected an element can be obtained. You will need provide the symbol and interesting factoid for five point question.
It is acceptable to name an element in honor of a geographical location. Some notable Earth locations would include: Am (americium, American continent), Ge (germanium, Germany), Fr (francium, France), and Eu (europium, European continent). Some scientists have ventured past Earth to name their elements in honor of other places. Examples of these would include Pu (plutonium, Pluto), U (uranium, Uranus), and Np (neptunium, Neptune).
Other options for naming elements are shown in the table below. Recently, elements 113-118 received their official names. Watch this ACS video to get some background regarding these new members of the table.
Example \(\PageIndex{1}\)
List the element names contained in the compounds below. Classify each compound as being organic or inorganic.
- Na2SO4
- HCl
- Cu(NO3)2 · H2O
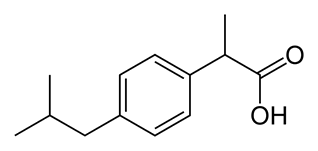
- CO2
SOLUTION
- sodium, sulfur, and oxygen. This compound is inorganic.
- hydrogen and chlorine. This compound is inorganic
- copper, nitrogen, oxygen, and hydrogen. This compound is inorganic.
- The corners of each line drawing represent carbons. You can assume for the single lines, the carbons have hydrogens attached (organic chemists tend not to draw all atoms). This compound is organic and contains carbon, hydrogen and oxygen.
- carbon and oxygen. This compound is inorganic.
Example \(\PageIndex{2}\)
Locate and name all the elements you see below in the compounds that are given below. Circle the organic compounds.
\[ \begin{align*} &\ce{LiOCN} \quad &\ce{MgSO4} \quad &\ce{Ca(OH)2} \quad &\ce{H2Cr2O7} \\ &\ce{C6H12O6}\quad &\ce{Pb3N4}\quad &\ce{CO2} \quad&\ce{UF6} \\ &\ce{Ch2CH2OH} \quad&\ce{Cl2} \quad&\ce{AuBr2}\quad &\ce{MnCO3} \end{align*}\]
Watch the video below and answer questions: https://www.youtube.com/watch?v=ItrYpHsymfI
- How were the last four elements made?
- How many total elements do we have now?
- How many elements are named in honor of female scientists?
- What would you name your new element and what would its symbol might be?
- Answer
-
(elements only)

Contributors
- Hayden Cox (Furman University)


