3.4: Determining Empirical and Molecular Formulas
- Page ID
- 232996
Learning Objectives
By the end of this section, you will be able to:
- Compute the percent composition of a compound
- Determine the empirical formula of a compound
- Determine the molecular formula of a compound
In the previous section, we discussed the relationship between the bulk mass of a substance and the number of atoms or molecules it contains (moles). Given the chemical formula of the substance, we were able to determine the amount of the substance (moles) from its mass, and vice versa. But what if the chemical formula of a substance is unknown? In this section, we will explore how to apply these very same principles in order to derive the chemical formulas of unknown substances from experimental mass measurements.
Percent Composition
The elemental makeup of a compound defines its chemical identity, and chemical formulas are the most succinct way of representing this elemental makeup. When a compound’s formula is unknown, measuring the mass of each of its constituent elements is often the first step in the process of determining the formula experimentally. The results of these measurements permit the calculation of the compound’s percent composition, defined as the percentage by mass of each element in the compound. For example, consider a gaseous compound composed solely of carbon and hydrogen. The percent composition of this compound could be represented as follows:
If analysis of a 10.0-g sample of this gas showed it to contain 2.5 g H and 7.5 g C, the percent composition would be calculated to be 25% H and 75% C:
Example 1: Calculation of Percent Composition
Analysis of a 12.04-g sample of a liquid compound composed of carbon, hydrogen, and nitrogen showed it to contain 7.34 g C, 1.85 g H, and 2.85 g N. What is the percent composition of this compound?
[reveal-answer q=”82650″]Show Answer[/reveal-answer]
[hidden-answer a=”82650″]
To calculate percent composition, we divide the experimentally derived mass of each element by the overall mass of the compound, and then convert to a percentage:
The analysis results indicate that the compound is 61.0% C, 15.4% H, and 23.7% N by mass.
[/hidden-answer]
Check Your Learning
A 24.81-g sample of a gaseous compound containing only carbon, oxygen, and chlorine is determined to contain 3.01 g C, 4.00 g O, and 17.81 g Cl. What is this compound’s percent composition?
[reveal-answer q=”39718″]Show Answer[/reveal-answer]
[hidden-answer a=”39718″]12.1% C, 16.1% O, 71.8% Cl[/hidden-answer]
Determining Percent Composition from Formula Mass
Percent composition is also useful for evaluating the relative abundance of a given element in different compounds of known formulas. As one example, consider the common nitrogen-containing fertilizers ammonia (NH3), ammonium nitrate (NH4NO3), and urea (CH4N2O). The element nitrogen is the active ingredient for agricultural purposes, so the mass percentage of nitrogen in the compound is a practical and economic concern for consumers choosing among these fertilizers. For these sorts of applications, the percent composition of a compound is easily derived from its formula mass and the atomic masses of its constituent elements. A molecule of NH3 contains one N atom weighing 14.01 amu and three H atoms weighing a total of (3 × 1.008 amu) = 3.024 amu The formula mass of ammonia is therefore (14.01 amu + 3.024 amu) = 17.03 amu, and its percent composition is:
This same approach may be taken considering a pair of molecules, a dozen molecules, or a mole of molecules, etc. The latter amount is most convenient and would simply involve the use of molar masses instead of atomic and formula masses, as demonstrated in the example problem below. As long as we know the chemical formula of the substance in question, we can easily derive percent composition from the formula mass or molar mass.
Example 2: Determining Percent Composition from a Molecular Formula
Aspirin is a compound with the molecular formula C9H8O4. What is its percent composition?
[reveal-answer q=”97551″]Show Answer[/reveal-answer]
[hidden-answer a=”97551″]
To calculate the percent composition, we need to know the masses of C, H, and O in a known mass of C9H8O4. It is convenient to consider 1 mol of C9H8O4 and use its molar mass (180.159 g/mole, determined from the chemical formula) to calculate the percentages of each of its elements:
%C =60.00% C
%H = 4.476% H
%O = 35.52% O
Note that these percentages sum to equal 100.00% when appropriately rounded.
[/hidden-answer]
Check Your Learning
To three significant digits, what is the mass percentage of iron in the compound Fe2O3?
[reveal-answer q=”992530″]Show Answer[/reveal-answer]
[hidden-answer a=”992530″]69.9% Fe[/hidden-answer]
Exercises
- Calculate the following to four significant figures:
- the percent composition of ammonia, NH3
- the percent composition of photographic “hypo,” Na2S2O3
- the percent of calcium ion in Ca3(PO4)2
- Determine the following to four significant figures:
- the percent composition of hydrazoic acid, HN3
- the percent composition of TNT, C6H2(CH3)(NO2)3
- the percent of SO42– in Al2(SO4)3
- Determine the percent ammonia, NH3, in Co(NH3)6Cl3, to three significant figures.
- Determine the percent water in CuSO4∙5H2O to three significant figures.
[reveal-answer q=”954320″]Show Selected Answers[/reveal-answer]
[hidden-answer a=”954320″]
1. In each of these exercises asking for the percent composition, divide the molecular weight of the desired element or group of elements (the number of times it/they occur in the formula times the molecular weight of the desired element or elements) by the molecular weight of the compound.
3.
[/hidden-answer]
Determination of Empirical Formulas
As previously mentioned, the most common approach to determining a compound’s chemical formula is to first measure the masses of its constituent elements. However, we must keep in mind that chemical formulas represent the relative numbers, not masses, of atoms in the substance. Therefore, any experimentally derived data involving mass must be used to derive the corresponding numbers of atoms in the compound. To accomplish this, we can use molar masses to convert the mass of each element to a number of moles. We then consider the moles of each element relative to each other, converting these numbers into a whole-number ratio that can be used to derive the empirical formula of the substance. Consider a sample of compound determined to contain 1.71 g C and 0.287 g H. The corresponding numbers of atoms (in moles) are:
Thus, we can accurately represent this compound with the formula C0.142H0.248. Of course, per accepted convention, formulas contain whole-number subscripts, which can be achieved by dividing each subscript by the smaller subscript:
(Recall that subscripts of “1” are not written but rather assumed if no other number is present.)
The empirical formula for this compound is thus CH2. This may or not be the compound’s molecular formula as well; however, we would need additional information to make that determination (as discussed later in this section).
Consider as another example a sample of compound determined to contain 5.31 g Cl and 8.40 g O. Following the same approach yields a tentative empirical formula of:
In this case, dividing by the smallest subscript still leaves us with a decimal subscript in the empirical formula. To convert this into a whole number, we must multiply each of the subscripts by two, retaining the same atom ratio and yielding Cl2O7 as the final empirical formula.
In summary, empirical formulas are derived from experimentally measured element masses by:
- Deriving the number of moles of each element from its mass
- Dividing each element’s molar amount by the smallest molar amount to yield subscripts for a tentative empirical formula
- Multiplying all coefficients by an integer, if necessary, to ensure that the smallest whole-number ratio of subscripts is obtained
Figure 1 outlines this procedure in flow chart fashion for a substance containing elements A and X.
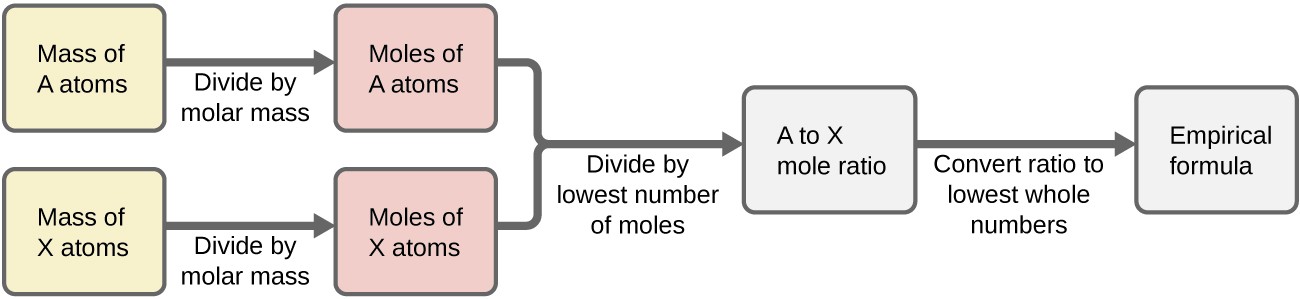
Example 3: Determining a Compound’s Empirical Formula from the Masses of Its Elements
A sample of the black mineral hematite (Figure 2), an oxide of iron found in many iron ores, contains 34.97 g of iron and 15.03 g of oxygen. What is the empirical formula of hematite?

[reveal-answer q=”235849″]Show Answer[/reveal-answer]
[hidden-answer a=”235849″]
For this problem, we are given the mass in grams of each element. Begin by finding the moles of each:
Next, derive the iron-to-oxygen molar ratio by dividing by the lesser number of moles:
The ratio is 1.000 mol of iron to 1.500 mol of oxygen (Fe1O1.5). Finally, multiply the ratio by two to get the smallest possible whole number subscripts while still maintaining the correct iron-to-oxygen ratio:
2(Fe1O1.5) = Fe2O3
The empirical formula is Fe2O3.
[/hidden-answer]
Check Your Learning
What is the empirical formula of a compound if a sample contains 0.130 g of nitrogen and 0.370 g of oxygen?
[reveal-answer q=”961821″]Show Answer[/reveal-answer]
[hidden-answer a=”961821″]N2O5[/hidden-answer]
For additional worked examples illustrating the derivation of empirical formulas, watch the brief video clip below.
Deriving Empirical Formulas from Percent Composition
Finally, with regard to deriving empirical formulas, consider instances in which a compound’s percent composition is available rather than the absolute masses of the compound’s constituent elements. In such cases, the percent composition can be used to calculate the masses of elements present in any convenient mass of compound; these masses can then be used to derive the empirical formula in the usual fashion.
Example 4: Determining an Empirical Formula from Percent Composition
The bacterial fermentation of grain to produce ethanol forms a gas with a percent composition of 27.29% C and 72.71% O (Figure 3). What is the empirical formula for this gas?

[reveal-answer q=”93530″]Show Answer[/reveal-answer]
[hidden-answer a=”93530″]
Since the scale for percentages is 100, it is most convenient to calculate the mass of elements present in a sample weighing 100 g. The calculation is “most convenient” because, per the definition for percent composition, the mass of a given element in grams is numerically equivalent to the element’s mass percentage. This numerical equivalence results from the definition of the “percentage” unit, whose name is derived from the Latin phrase per centum meaning “by the hundred.” Considering this definition, the mass percentages provided may be more conveniently expressed as fractions:
The molar amounts of carbon and hydrogen in a 100-g sample are calculated by dividing each element’s mass by its molar mass:
Coefficients for the tentative empirical formula are derived by dividing each molar amount by the lesser of the two:
Since the resulting ratio is one carbon to two oxygen atoms, the empirical formula is CO2.
[/hidden-answer]
Check Your Learning
What is the empirical formula of a compound containing 40.0% C, 6.71% H, and 53.28% O?
[reveal-answer q=”381880″]Show Answer[/reveal-answer]
[hidden-answer a=”381880″]CH2O[/hidden-answer]
Derivation of Molecular Formulas
Recall that empirical formulas are symbols representing the relative numbers of a compound’s elements. Determining the absolute numbers of atoms that compose a single molecule of a covalent compound requires knowledge of both its empirical formula and its molecular mass or molar mass. These quantities may be determined experimentally by various measurement techniques. Molecular mass, for example, is often derived from the mass spectrum of the compound (see discussion of this technique in the previous chapter on atoms and molecules). Molar mass can be measured by a number of experimental methods, many of which will be introduced in later chapters of this text.
Molecular formulas are derived by comparing the compound’s molecular or molar mass to its empirical formula mass. As the name suggests, an empirical formula mass is the sum of the average atomic masses of all the atoms represented in an empirical formula. If we know the molecular (or molar) mass of the substance, we can divide this by the empirical formula mass in order to identify the number of empirical formula units per molecule, which we designate as n:
The molecular formula is then obtained by multiplying each subscript in the empirical formula by n, as shown below for the generic empirical formula AxBy:
Callstack:
at (Courses/Lumen_Learning/Book:_General_Chemistry__Lecture_and_Lab_(Lumen)/03:_Composition_of_Substances_and_Solutions/3.04:_Determining_Empirical_and_Molecular_Formulas), /content/body/div[1]/div[4]/p[5]/img/@alt, line 1, column 3
For example, consider a covalent compound whose empirical formula is determined to be CH2O. The empirical formula mass for this compound is approximately 30 amu (the sum of 12 amu for one C atom, 2 amu for two H atoms, and 16 amu for one O atom). If the compound’s molecular mass is determined to be 180 amu, this indicates that molecules of this compound contain six times the number of atoms represented in the empirical formula:
Molecules of this compound are then represented by molecular formulas whose subscripts are six times greater than those in the empirical formula:
Note that this same approach may be used when the molar mass (g/mol) instead of the molecular mass (amu) is used. In this case, we are merely considering one mole of empirical formula units and molecules, as opposed to single units and molecules.
Example 5: Determination of the Molecular Formula for Nicotine
Nicotine, an alkaloid in the nightshade family of plants that is mainly responsible for the addictive nature of cigarettes, contains 74.02% C, 8.710% H, and 17.27% N. If 40.57 g of nicotine contains 0.2500 mol nicotine, what is the molecular formula?
[reveal-answer q=”658532″]Show Answer[/reveal-answer]
[hidden-answer a=”658532″]
Determining the molecular formula from the provided data will require comparison of the compound’s empirical formula mass to its molar mass. As the first step, use the percent composition to derive the compound’s empirical formula. Assuming a convenient, a 100-g sample of nicotine yields the following molar amounts of its elements:
Next, we calculate the molar ratios of these elements.
The C-to-N and H-to-N molar ratios are adequately close to whole numbers, and so the empirical formula is C5H7N. The empirical formula mass for this compound is therefore 81.13 amu/formula unit, or 81.13 g/mol formula unit.
We calculate the molar mass for nicotine from the given mass and molar amount of compound:
Comparing the molar mass and empirical formula mass indicates that each nicotine molecule contains two formula units:
Thus, we can derive the molecular formula for nicotine from the empirical formula by multiplying each subscript by two:
Callstack:
at (Courses/Lumen_Learning/Book:_General_Chemistry__Lecture_and_Lab_(Lumen)/03:_Composition_of_Substances_and_Solutions/3.04:_Determining_Empirical_and_Molecular_Formulas), /content/body/div[1]/div[4]/div[1]/div/p[11]/img/@alt, line 1, column 3
[/hidden-answer]
Check Your Learning
What is the molecular formula of a compound with a percent composition of 49.47% C, 5.201% H, 28.84% N, and 16.48% O, and a molecular mass of 194.2 amu?
[reveal-answer q=”509990″]Show Answer[/reveal-answer]
[hidden-answer a=”509990″]C8H10N4O2[/hidden-answer]
Key Concepts and Summary
The chemical identity of a substance is defined by the types and relative numbers of atoms composing its fundamental entities (molecules in the case of covalent compounds, ions in the case of ionic compounds). A compound’s percent composition provides the mass percentage of each element in the compound, and it is often experimentally determined and used to derive the compound’s empirical formula. The empirical formula mass of a covalent compound may be compared to the compound’s molecular or molar mass to derive a molecular formula.
Key Equations
- (AxBy)n = AnxBny
Exercises
- What information do we need to determine the molecular formula of a compound from the empirical formula?
- Determine the empirical formulas for compounds with the following percent compositions:
- 15.8% carbon and 84.2% sulfur
- 40.0% carbon, 6.7% hydrogen, and 53.3% oxygen
- Determine the empirical formulas for compounds with the following percent compositions:
- 43.6% phosphorus and 56.4% oxygen
- 28.7% K, 1.5% H, 22.8% P, and 47.0% O
- Polymers are large molecules composed of simple units repeated many times. Thus, they often have relatively simple empirical formulas. Calculate the empirical formulas of the following polymers:
- Lucite (Plexiglas); 59.9% C, 8.06% H, 32.0% O
- Saran; 24.8% C, 2.0% H, 73.1% Cl
- polyethylene; 86% C, 14% H
- polystyrene; 92.3% C, 7.7% H
- Orlon; 67.9% C, 5.70% H, 26.4% N
- A compound of carbon and hydrogen contains 92.3% C and has a molar mass of 78.1 g/mol. What is its molecular formula?
- Dichloroethane, a compound that is often used for dry cleaning, contains carbon, hydrogen, and chlorine. It has a molar mass of 99 g/mol. Analysis of a sample shows that it contains 24.3% carbon and 4.1% hydrogen. What is its molecular formula?
- Determine the empirical and molecular formula for chrysotile asbestos. Chrysotile has the following percent composition: 28.03% Mg, 21.60% Si, 1.16% H, and 49.21% O. The molar mass for chrysotile is 520.8 g/mol.
- A major textile dye manufacturer developed a new yellow dye. The dye has a percent composition of 75.95% C, 17.72% N, and 6.33% H by mass with a molar mass of about 240 g/mol. Determine the molecular formula of the dye.
[reveal-answer q=”265420″]Show Selected Answers[/reveal-answer]
[hidden-answer a=”265420″]
2. The empirical formulas can be found as follows:
- The percent of an element in a compound indicates the percent by mass. The mass of an element in a 100.0-g sample of a compound is equal in grams to the percent of that element in the sample; hence, 100.0 g of the sample contains 15.8 g of C and 84.2 g of S. The relative number of moles of C and S atoms in the compound can be obtained by converting grams to moles as shown.
Step 1:
Step 2:- The empirical formula is CS2.
- Step 1:
Step 2:- The empirical formula is CH2O.
5. To determine the empirical formula, a relationship between percent composition and atom composition must be established. The percent composition of each element in a compound can be found either by dividing its mass by the total mass of compound or by dividing the molar mass of that element as it appears in the formula (atomic mass times the number of times the element appears in the formula) by the formula mass of the compound. From this latter perspective, the percent composition of an element can be converted into a mass by assuming that we start with a 100-g sample. Then, multiplying the percentage times 100 g gives the mass in grams of that component. Division of each mass by its respective atomic mass gives the relative ratio of atoms in the formula. From the numbers so obtained, the whole-number ratio of elements in the compound can be found by dividing each ratio by the number representing the smallest ratio. Generally, this process can be done in two simple steps (a third step is needed if the ratios are not whole numbers).
Step 1: Divide each element’s percentage (converted to grams) by its atomic mass:
This operation established the relative ration of carbon to hydrogen in the formula.
Step 2: To establish a whole-number ratio of carbon to hydrogen, divide each factor by the smallest factor. In this case, both factors are essentially equal; thus the ration of atoms is 1 to 1:
The empirical formula is CH.
Since the molecular mass of the compound is 78.1 amu, some integer times the sum of the mass of 1C and 1H in atomic mass units (12.011 amu + 1.00794 amu = 13.019 amu) must be equal to 78.1 amu. To find this number, divide 78.1 amu by 13.019 amu:
The molecular formula is (CH)6 = C6H6.
7. The formulas can be found as follows:
(2)(Mg1.5Si1H1.5O4) = Mg3Si2H3O8 (empirical formula), empirical mass of 260.1 g/unit
so (2)(Mg3Si2H3O8) = Mg6Si4H6O16
8. Assume 100.0 g; the percentages of the elements are then the same as their mass in grams. Divide each mass by the molar mass to find the number of moles.
Step 1:
Step 2: Divide each by the smallest number. The answers are 5C, 1N, and 5H. The empirical formula is C5H5N, which has a molar mass of 79.10 g/mol. To find the actual molecular formula, divide 240, the molar mass of the compound, by 79.10 to obtain 3. So the formula is three times the empirical formula, or C15H15N3.
[/hidden-answer]
Glossary
empirical formula mass: sum of average atomic masses for all atoms represented in an empirical formula
percent composition: percentage by mass of the various elements in a compound
- Chemistry. Provided by: OpenStax College. Located at: http://openstaxcollege.org. License: CC BY: Attribution. License Terms: Download for free at https://openstaxcollege.org/textbooks/chemistry/get
- How to Calculate Percent Composition, Empirical Formulas and Molecular Formulas - Chemistry. Authored by: Mr. Causey. Located at: https://youtu.be/mdNYDMoQ6As. License: All Rights Reserved. License Terms: Standard YouTube License


