20.17 Polymers
- Page ID
- 206405
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Most of these materials are made in the same basic way. The starting materials or monomers are relatively simple molecules—usually carbon compounds derived from petroleum—which can be persuaded to link up with each other in order to form a long chain of repeating units called a polymer. If we think of the monomer as a bead, then the polymer corresponds to a string of beads. Polymers are usually classified into two types: addition polymers and condensation polymers, according to the kind of reaction by which they are made.
Addition Polymers
Addition polymers are usually made from a monomer containing a double bond. We can think of the double bond as "opening out" in order to participate in two new single bonds in the following way:
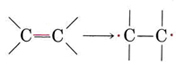
Thus, if ethene is heated at moderate temperature and pressure in the presence of an appropriate catalyst, it polymerizes:

| Monomer | Nonsystematic Name | Polymer | Some Typical Uses |
|---|---|---|---|
| Ethylene | Polyethylene | Film for packaging and bags, toys, bottles, coatings | |
| Propylene | Polypropylene | Milk cartons, rope, outdoor carpeting | |
| Styrene | Polystyrene | Transparent containers, plastic glasses, refrigerators, styrofoam | |
| Vinyl chloride | Polyvinyl chloride, PVC | Pipe and tubing, raincoats, curtains, phonograph records, luggage, floor tiles | |
| Acrylonitrile | Polyacrylonitrile (Orlon, Acrilan) | Textiles, ruga | |
| Tetrafluoroethylene | Teflon | Nonstick pan coatings, bearings, gaskets |
The result is the familiar waxy plastic called polyethylene, which at a molecular level consists of a collection of long-chain alkane molecules, most of which contain tens of thousands of carbon atoms. There is only an occasional short branch chain.
Polyethylene is currently manufactured on a very large scale, larger than any other polymer, and is used for making plastic bags, cheap bottles, toys, etc. Many of its properties are what we would expect from its molecular composition. The fact that it is a mixture of molecules each of slightly different chain length (and hence slightly different melting point) explains why it softens over a range of temperatures rather than having a single melting point. Because the molecules are only held together by London forces, this melting and softening occurs at a rather low temperature. (Some of the cheaper varieties of polyethylene with shorter chains and more branch chains will even soften in boiling water.) The same weak London forces explain why polyethylene is soft and easy to scratch and why it is not very ‘strong mechanically.'
The table above lists some other well-known addition polymers and also some of their uses. You can probably find at least one example of each of them in your home. Except for Teflon, all these polymers derive from a monomer of the form
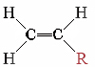
The resulting polymer thus has the general form
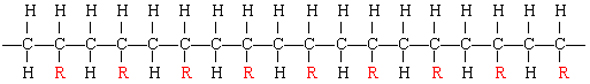
By varying the nature of the R group, the physical properties of the polymer can be controlled rather precisely.
Condensation Polymers
When addition polymers are formed, no by-products result. Formation of a condensation polymer, on the other hand, produces H2O, HCl, or some other simple molecule which escapes as a gas. A familiar example of a condensation polymer is nylon, which is obtained from the reaction of two monomers
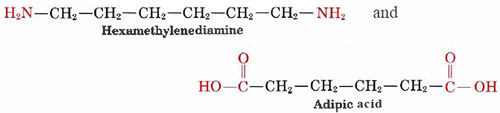
These two molecules can link up with each other because each contains a reactive functional group, either an amine or a carboxylic acid which react to form an amide linkage. They combine as follows:
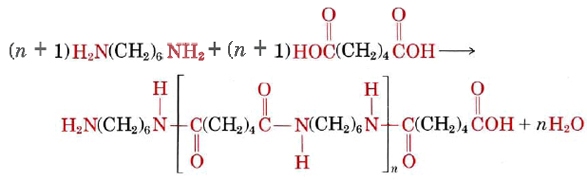
Below is a video of the reaction to form nylon. This reaction is slightly modified from the one described above, as adipoyl chloride, not adipic acid, is used as a reactant. Thus HCl, not H2O is produced. This also means that the chain terminates in an acid chloride, rather than the carboxylic acid shown above. Note that an amide linkage is still formed.
A solution of adipoyl chloride in cyclohexane is poured on top of an aqueous solution of 1,6-diaminohexane in a beaker. Nylon (6,6) polyamide is formed at the interface of the two immiscible liquids and is carefully drawn from the solution and placed on a glass rod. The rod is then spun, and the Nylon (6,6) polyamide is spun onto the rod.
Well-known condensation polymers other than nylon are Dacron, Bakelite, melamine, and Mylar. Nylon makes extremely strong threads and fibers because its long-chain molecules have stronger intermolecular forces than the London forces of polyethylene. Each N—H group in a nylon chain can hydrogen bond to the O of a C=O group in a neighboring chain, as shown below. Therefore the chains cannot slide past one another easily.
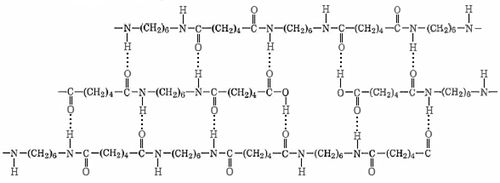
If you pull on both ends of a nylon thread, for example, it will only stretch slightly. After that it will strongly resist breaking because a large number of hydrogen bonds are holding overlapping chains together. The same is not true of a polyethylene thread in which only London forces attract overlapping chains together, and this is one reason that polyethylene is not used to make thread.
Contributors
Ed Vitz (Kutztown University), John W. Moore (UW-Madison), Justin Shorb (Hope College), Xavier Prat-Resina (University of Minnesota Rochester), Tim Wendorff, and Adam Hahn.
Contributors
Ed Vitz (Kutztown University), John W. Moore (UW-Madison), Justin Shorb (Hope College), Xavier Prat-Resina (University of Minnesota Rochester), Tim Wendorff, and Adam Hahn.

