10.9: Functional Groups
- Page ID
- 237768
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Define the following terms: functional group, alkene, alkyne, aromatic, halide, alcohol, ether, thiol, sulfide, amine, carbonyl, aldehyde, ketone, carboxylic acid, ester, amide.
- Classify an organic molecule according to the functional group(s) that it contains.
Sections 10.2 through 10.6 identified and defined the five types of structural notations in which an organic molecule can be symbolized and described the compositional characteristics that can be determined by analyzing the molecular formula and expanded, VSEPR, condensed, and bond-line pictures of a substance. Subsequently, multiple molecular structures were provided and used as reference images from which alternative structural representations were derived. The molecules that are represented by these pictures are largely comprised of carbon/carbon and carbon/hydrogen single bonds and, therefore, are classified as alkanes. As stated in Section 10.1, the types of elements and bonds that are present in an organic molecule dictate not only the physical properties of that substance, but also its chemical reactivity. Therefore, because alkanes contain a limited combination of elements and bonds, both the utility and the reactivity of this class of organic molecules is severely restricted.
Recall that atoms of carbon can form four stable covalent bonds with each other and with many other elements and, consequently, are found in millions of different substances. Thousands of these molecules contain other non-metals, such as nitrogen, N, oxygen, O, phosphorus, P, sulfur, S, and the halogens, X, in addition to the carbon and hydrogen atoms that are present in alkanes. Furthermore, while most of the elements that are found in organic molecules bond to one another by sharing one pair of electrons, carbon atoms are also able to form double and triple bonds, in which two or three pairs of electrons, respectively, are shared between the same set of atoms.
During their investigations of organic molecules, chemists recognized that these bonds and atoms were often combined with one another in 14 specific configurations and, furthermore, that molecules with similar compositions shared common physical properties and reactivity patterns. Therefore, scientists defined 14 terms, which are collectively-known as functional groups, in order to indicate the specific types of bonds and atoms that are present in each of these chemically-significant bonding arrangements. As will be discussed in greater detail in the following paragraphs, five of the functional groups that will be described contain both a double bond and one or more heteroatoms and, therefore, are more chemically-complex than the remaining nine configurations, in which either a multiple bond or a heteroatom is present.
Finally, in addition to a verbal description, the bonds and atoms that are contained in each type of functional group will be indicated in red in a molecular structure that is symbolically-represented using bond-line notation, which, due to its minimalistic format, is the drawing convention that is most commonly-used by organic chemists. As stated in Section 10.6, bond-line notation uses "zig-zag" lines to represent the bonds that exist between consecutive carbon atoms and does not explicitly show the elemental symbols of carbon or hydrogen. Recall that any double or triple bonds that are present in an organic molecule are symbolized by writing an extra "floating" line on a "zig-zag" core structure. Furthermore, any hydrogen atoms that are bonded to heteroatoms are shown using a condensed format.
Hydrocarbon Functional Groups
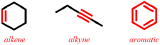
Recall that hydrocarbons are defined as organic molecules that are comprised of only carbon, C, and hydrogen, H, and, therefore, do not contain any other elements. Consequently, since a functional group must contain at least one multiple bond or heteroatom, either a double or a triple bond must be present in a hydrocarbon functional group. An organic molecule that contains one or more carbon/carbon double bonds is classified as an alkene. As shown in the first image in Figure \(\PageIndex{1}\), the second bond of an alkene is indicated by the presence of one additional "floating" line on the main bond-line "zig-zag" pattern. An alkyne must contain one or more carbon/carbon triple bonds and, therefore, are symbolically-represented by adding two additional "floating" lines to a bond-line core structure, as shown in the second image in Figure \(\PageIndex{1}\). The final type of hydrocarbon functional group, an aromatic, contains alternating single and double bonds in a six-carbon cyclic "ring" core. Therefore, while both the first and last pictures that are shown in Figure \(\PageIndex{1}\) both symbolize cyclic organic molecules that contain six carbons, only the final structure, in which an extra "floating" line is present on every other bond, can be classified as an aromatic. The molecule that is represented by the first image does not have enough "floating" bonds to be categorized as an aromatic and, therefore, is classified as an alkene, as stated above.
Heteroatom Functional Groups
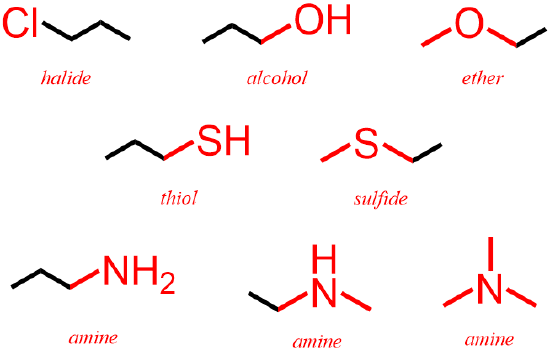
By definition, no heteroatoms are present in alkenes, alkynes, and aromatics, which are functional groups that contain one or more carbon/carbon double or triple bonds. In contrast, halide, alcohol, ether, thiol, sulfide, and amine functional groups, which are comprised of only single bonds, each contain a specific heteroatom, as shown below in Figure \(\PageIndex{2}\). The elemental symbol of a halogen, such as fluorine, F, chlorine, Cl, bromine, Br, or iodine, I, must be explicitly-written in the bond-line structure that represents an organic halide. While an alcohol contains an oxygen atom, O, that is singly-bonded to one carbon and one hydrogen, H, if an oxygen, O, that is present in an organic molecule is singly-bonded to two different carbon atoms, that substance is classified as an ether. Furthermore, because oxygen and sulfur are located in the same group, or column, of the periodic table, these elements usually achieve identical configurations when bonding. Therefore, thiols and sulfides are sulfur-analogs of the alcohol and ether functional groups, respectively, that are defined above. Finally, organic amines contain nitrogen atoms, N, that are singly-bonded to at least one carbon atom. However, unlike the other heteroatom functional groups, the other two atoms that are associated with an amine nitrogen can be either carbons or hydrogens and, therefore, three types of amine structures can be drawn, as shown below in Figure \(\PageIndex{2}\).
Carbonyl Functional Groups
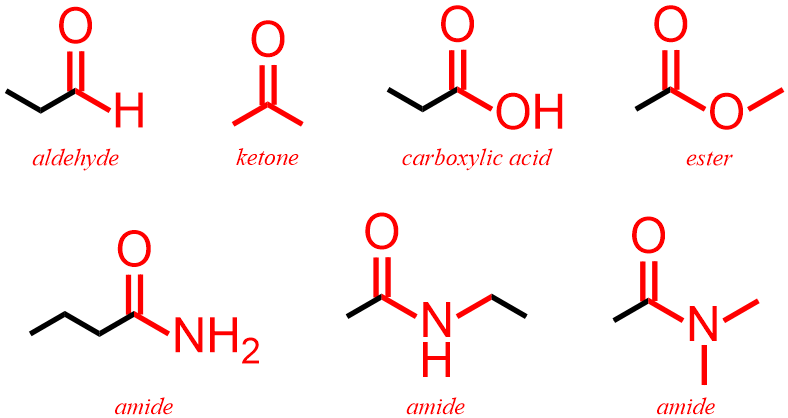
The five remaining functional groups all contain a carbon/oxygen double bond, which is called a carbonyl. As stated previously, atoms of carbon can form four covalent bonds when incorporated into organic molecules. Therefore, since, by definition, the carbon atom that is present in a carbonyl bears two bonds to oxygen, this carbon must also be bonded to two additional atoms, in order to achieve its preferred bonding configuration. As a result, aldehyde, ketone, carboxylic acid, ester, and amide functional groups are defined and distinguished by the types of atoms that are bonded to a carbonyl carbon. In an aldehyde, a carbonyl carbon must be bonded to one hydrogen, H. If, however, that carbon is bonded to two different carbon atoms, the corresponding molecule is classified as a ketone. Carboxylic acid, ester, and amide functional groups contain carbonyls that are directly bonded to alcohol, ether, and amine units, respectively. Therefore, the presence or absence of a carbonyl is used to distinguish between carboxylic acids and alcohols, esters and ethers, and amides and amines. Finally, as described above, the other two atoms that are associated with an amide nitrogen can be either carbons or hydrogens and, therefore, three types of amide structures can be drawn, as shown below in Figure \(\PageIndex{3}\).
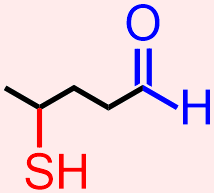
For example, classify the organic molecule that is represented below according to the functional group(s) that it contains.

The molecule that is represented above contains an alkyne, an aromatic, and an amide. The atoms and bonds that are associated with each of these functional groups, a carbon/carbon triple bond, alternating single and double bonds in a six-carbon cyclic "ring" core, and a carbonyl that is directly bonded to an amine unit, respectively, are indicated in red, blue, and green in the structure that is shown below.
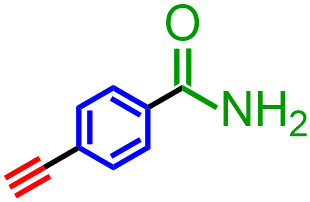
Classify the organic molecule that is represented below according to the functional group(s) that it contains.

- Answer
- The molecule that is represented above contains an alkene, an ether, and an ester. The atoms and bonds that are associated with each of these functional groups, a carbon/carbon double bond, an oxygen that is singly-bonded to two different carbon atoms, and a carbonyl that is directly bonded to an ether unit, respectively, are indicated in red, blue, and green in the structure that is shown below.
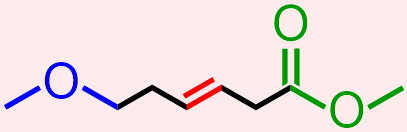
Classify the organic molecule that is represented below according to the functional group(s) that it contains.
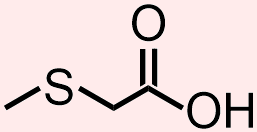
- Answer
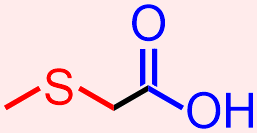
- The molecule that is represented above contains a thiol and a carboxylic acid. The atoms and bonds that are associated with each of these functional groups, a sulfur that is singly-bonded to two differerent carbon atoms and a carbonyl that is directly bonded to an alcohol unit, respectively, are indicated in red and blue in the structure that is shown below.
Classify the organic molecule that is represented below according to the functional group(s) that it contains.
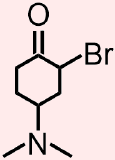
- Answer
- The molecule that is represented above contains a halide, an amine, and a ketone. The atoms and bonds that are associated with each of these functional groups, the elemental symbol of a halogen, Br, a nitrogen that is singly-bonded to at least one carbon atom, and a carbonyl that is bonded to two different carbon atoms, respectively, are indicated in red, blue, and green in the structure that is shown below.
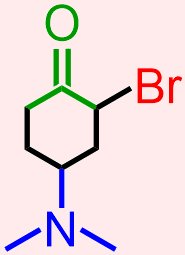
Classify the organic molecule that is represented below according to the functional group(s) that it contains.
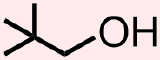
- Answer
- The molecule that is represented above contains an alcohol. The atoms and bonds that are associated with this functional group, an oxygen that is singly-bonded to one carbon and one hydrogen, H, are indicated in red in the structure that is shown below.
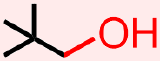
Classify the organic molecule that is represented below according to the functional group(s) that it contains.
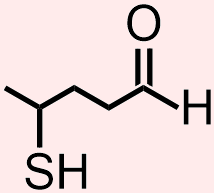
- Answer
- The molecule that is represented above contains a thiol and an aldehyde. The atoms and bonds that are associated with each of these functional groups, a sulfur that is singly-bonded to one carbon and one hydrogen, H, and a carbonyl that is bonded to one hydrogen, H, respectively, are indicated in red and blue in the structure that is shown below.