5.10: Non-medical Applications of Nuclear Science
- Page ID
- 85499
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Appreciate how radiation is used in agriculture and industry.
- Realize various isotopes and wave radiation are used as tracers and sanitizers.
- Distinguish between millimeter and back scatter technologies uses by the Transportation Security Administration (TSA).
- Know examples of foods that are irradiated in the United States.
Tracer Applications
Radioactive isotopes are effective tracers because their radioactivity is easy to detect. A tracer is a substance that can be used to follow the pathway of that substance through some structure. For instance, leaks in underground water pipes can be discovered by running some tritium-containing water through the pipes and then using a Geiger counter to locate any radioactive tritium subsequently present in the ground around the pipes. (Recall that tritium is a radioactive isotope of hydrogen.)
Tracers are used in diverse ways to study the mechanisms of chemical reactions in plants and animals. These include labeling fertilizers in studies of nutrient uptake by plants and crop growth, investigations of digestive and milk-producing processes in cows, and studies on the growth and metabolism of animals and plants.
For example, the radioisotope C-14 was used to elucidate the details of how photosynthesis occurs. The overall reaction is:
\[\ce{6CO2}(g)+\ce{6H2O}(l)⟶\ce{C6H12O6}(s)+\ce{6O2}(g), \nonumber \]
The process is much more complex, proceeding through a series of steps in which various organic compounds are produced. In studies of the pathway of this reaction, plants were exposed to CO2 containing a high concentration of \(\ce{^{14}_6C}\). At regular intervals, the plants were analyzed to determine which organic compounds contained carbon-14 and how much of each compound was present. From the time sequence in which the compounds appeared and the amount of each present at given time intervals, scientists learned more about the pathway of the reaction.
Airport Applications
Commercial airports use x-rays and CT imaging to items contained in luggage. An x-ray machine can produce images of hard and soft materials. Based upon relative densities, an x-ray image will display a different color for a different type of material. For example, foods and organic materials will appear orange on an x-ray scan. Harder materials like thicker plastics will appear green. Lastly, metals and extremely thick materials will produce a bluish color when scanned. Click on this video for how technology for scanning bags has changed in the last five years.

American airports once used x-rays to scan human bodies for weapons. Up through 2013, back scatter technology was utilized to give very detailed body images. Due to the "naked body" pictures, this type of technology was banned in American airports and has now been replaced with millimeter wave generators. These machines produce cartoon like images and expose the body to high energy radio waves.

Irradiation of Food, Mail, and Water
Co-60 (gamma source) and x-rays are used to irradiate many foods in the United States. Ionizing radiation can be used to kill food-borne illnesses like salmonella and e coli. Irradiating food can also prolong shelf-life, delay ripening, and destroy insects. In addition, gamma/x-ray can sterilize foods making refrigeration unnecessary. Often, sterilized food is served to hospital patients who have impaired immune systems.
In the United States,irradiation of food is regulated by the FDA (Food and Drug Administration). Foods that have been irradiated must display the symbol called the Radura Figure \(\PageIndex{3}\) on their packaging. Some foods that could display this symbol are: meats (beef, pork, chicken), shrimp, lobster, fruits, vegetables, shellfish, and spices. Contrary to the belief of some people, irradiation of food does not make the food itself radioactive.

Ultraviolet (UVC) radiation has been used in the purification of drinking water. The peak wavelength for this process is around 222 nm. By using UVC, many viruses, bacteria, and other pathogens can be annihilated in air, water, and nonporous materials. UVC radiation is even powerful enough to destroy the protein coating of the SARS-Corona Virus. For more information on UVC and sanitation in water, click here.
In the fall of 2001, anthrax-laced letters were sent to various news agencies and two United States senators. Anthrax is an infectious disease caused by bacteria. It exists naturally in some soils and can be isolated in a laboratory. After September 11, 2011, this substance was used as a biochemical weapon. This particular incident killed five people and injured twenty-two, other individuals. Anthrax produces ulcers on the skin and causes severe breathing difficulties. Today, only members of the United States military receive vaccines for anthrax.

Figure \(\PageIndex{4}\): Anthrax-laced letter that was sent to Senator Tom Daschle. (Public Domain; FBI via Wikipedia)
To combat this form of bioterrorism, the United States Postal Service (USPS) and the Federal Bureau of Investigation (FBI) installed x-ray generators to irradiate suspicious-looking mail sent to some governmental facilities. X-rays will kill the majority of these bacteria and some viruses as well. Once irradiated, mail (packages and letters) might change in color, texture, and smell. This ionizing radiation chemically alters the composition of the mail's paper component. The x-rays do not leave the mail radioactive.
Smoke Detectors
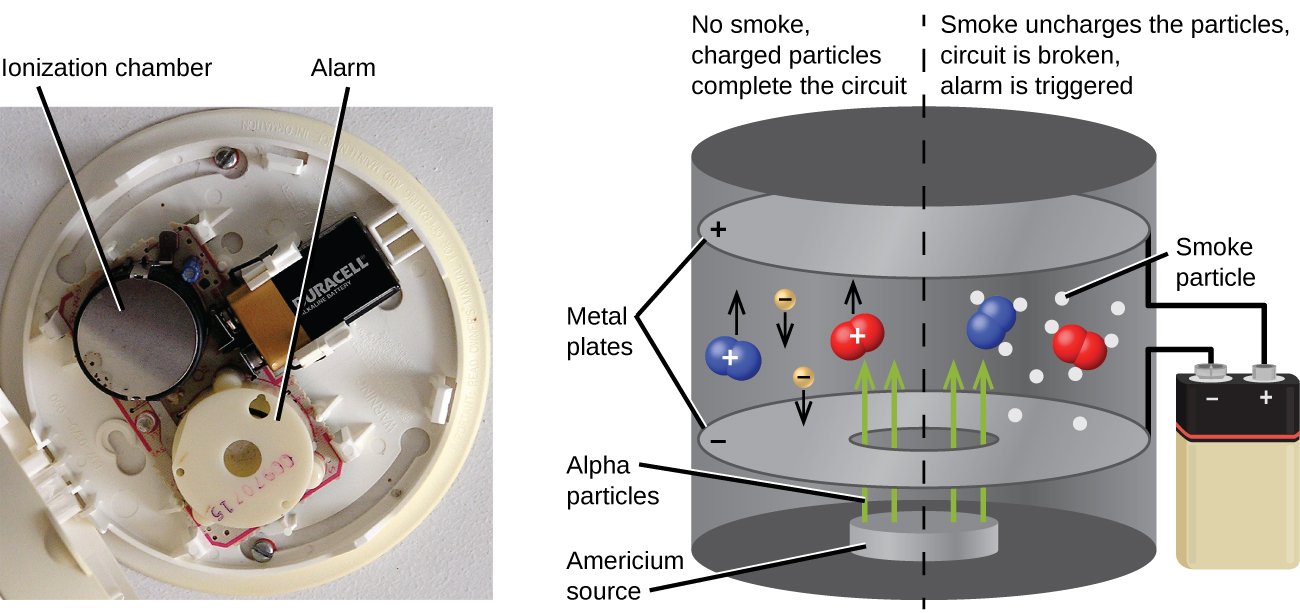
Americium-241, an α emitter with a half-life of 458 years, is used in tiny amounts in ionization-type smoke detectors (Figure \(\PageIndex{5}\)). The α emissions from Am-241 ionize the air between two electrode plates in the ionizing chamber. A battery supplies a potential that causes movement of the ions, thus creating a small electric current. When smoke enters the chamber, the movement of the ions is impeded, reducing the conductivity of the air. This causes a marked drop in the current, triggering an alarm.
References
- Emma Gibney (Furman University)


