4.4: Predicting Ion Charges
- Page ID
- 85155
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Recall how to locate valence electrons for all atoms (except transition and inner transition metals).
- Realize that metals will lose electrons to form cations and nonmetals gain electrons to form anions.
- Predict charges (using the modified Bohr model, Z < 20) for the following groups of elements: alkali, alkaline and halogens.
- Predict charges (using the modified Bohr model) for the following elements: phosphorus, nitrogen, sulfur, oxygen, and aluminum.
- Memorize charges for silver and zinc
- Label your periodic table with predicted charges from Bohr model.
Earlier, we located the valence electrons for elements Z < 20 by drawing modified Bohr structures. We can obtain these values quicker by referring to the roman numeral numbers above each family on the periodic table. The total number of valence electrons for an atom can vary between one and eight. If an element is located on the left side of the table (metal) and has less than three valence electrons, it will lose its valence in order to become stable and achieve an octet. In contrast, elements on the right side of the table (nonmetals) will gain up to eight electrons to achieve octet status.

Cations
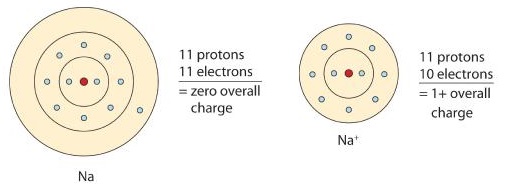
A neutral sodium atom is likely to achieve an octet in its outermost shell by losing its one valence electron.
\[\ce{Na \rightarrow Na^{+} + e^{-}} \nonumber \]
The cation produced in this way, Na+, is called the sodium ion to distinguish it from the element. The outermost shell of the sodium ion is the second electron shell, which has eight electrons in it. The octet rule has been satisfied. Figure \(\PageIndex{1}\) is a graphical depiction of this process.
While they are atoms, most metals are visible. You are aware that lead is a gray, solid metal.

When lead becomes a cation, it will not be visible and can become water soluble. If lead ions enter a drinking water system, one would not be able to detect them visually. For years, lead pipes were installed to transport water to consumers in many countries including the United States. If corrosive water were to enter a lead-based pipe, it would produce soluble lead ions. These charged species could be ingested unknowingly by residents of the community. Watch the video below and note how lead has affected the city of Flint, Michigan.
Follow-up questions
- Why was the drinking water source changed from Lake Huron to Flint River?
- Did the Flint River contain lead? If not, how did the lead enter the drinking water?
- How did the residents describe their drinking water? Please realize the discoloration of the water is due to excess iron and not lead.
- How can lead damage the body? Who does it affect the most?
- What could have been added to the water before releasing it to the pipes? Why did you think officials didn't add this chemical?
- What were all the contaminants this video mentioned?
- How did boiling the water help and hurt the residents of Flint, Michigan?
Anions
Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. When these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons. Negatively charged ions are called anions. Most nonmetals become anions when they make ionic compounds.
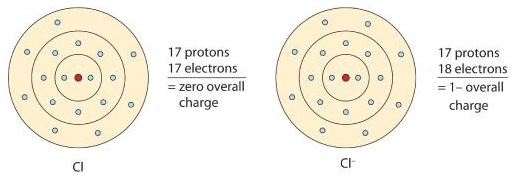
A neutral chlorine atom has seven electrons in its outermost shell. Only one more electron is needed to achieve an octet in chlorine’s valence shell. (In table salt, this electron comes from the sodium atom.)
\[\ce{e^{-} +Cl -> Cl^{-}} \nonumber \]
In this case, the ion has the same outermost shell as the original atom, but now that shell has eight electrons in it. Once again, the octet rule has been satisfied. The resulting anion, Cl−, is called the chloride ion; note the slight change in the suffix (-ide instead of -ine) to create the name of this anion. Figure \(\PageIndex{2}\) is a graphical depiction of this process.
The names for positive and negative ions are pronounced CAT-eye-ons and ANN-eye-ons, respectively.
Like cations, anions could be come soluble in a liquid. This would make the negatively charged species invisible to the naked eye. In this example, we will compare the element chlorine (which exists as Cl2) to the chlorine ion (Cl-). Chlorine is a toxic,yellow, and corrosive gas. This element was one of the first chemical weapons and was used during WWI. As an ion, chloride is used to disinfect water supplies (pools and drinking water). This colorless ion can still be corrosive, but not to the degree that chlorine gas would be.

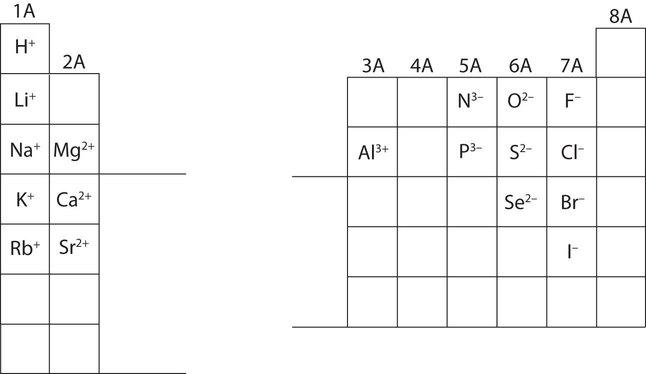
In many cases, elements that belong to the same group (vertical column) on the periodic table form ions with the same charge because they have the same number of valence electrons. Thus, the periodic table becomes a tool for remembering the charges on many ions. For example, all ions made from alkali metals, the first column on the periodic table, have a 1+ charge. Ions made from alkaline earth metals, the second group on the periodic table, have a 2+ charge. On the other side of the periodic table, the next-to-last column, the halogens, form ions having a 1− charge. Figure \(\PageIndex{3}\) shows how the charge on many ions can be predicted by the location of an element on the periodic table.
Will the species below lose or gain electrons?
- aluminium
- halogens
- metals in general
- silver
- oxygen
- Answer a
-
Aluminum is a metal that will always lose three electrons.
- Answer b
-
The halogens all have seven valence electrons. Each one of these elements wants to gain one electron to achieve an octet.
- Answer c
-
Metals will always lose electrons to form cations.
- Answer d
-
Silver is a metal that will always lose one electron. Commit this to memory!
- Answer e
-
Oxygen is a nonmetal that has six valence electrons. It will gain two electrons to achieve an octet.
Contributors and Attributions
Henry Agnew (UC Davis)
Isabella Quiros (Furman University)


