Molecules (and extended solids) are built from atoms that form chemical bonds. Theories of bonding seek to explain why molecules and solids form, what their structures are, why some are more stable than others, and how they react. As we will learn in Chapter 2, quantum mechanics gives us the most realistic picture of chemical bonding via molecular orbital (MO) theory. However, the MO description of bonding is conceptually difficult and mathematically intensive. This chapter will review less rigorous (but still useful) models such as Lewis dot structures and valence shell electron-pair repulsion (VSEPR) theory. When combined with a qualitative quantum mechanical description of bonding through the concepts of orbital hybridization and resonance, these simple models can help us understand a great deal about the structures, stabilities, and reactions of inorganic molecules.
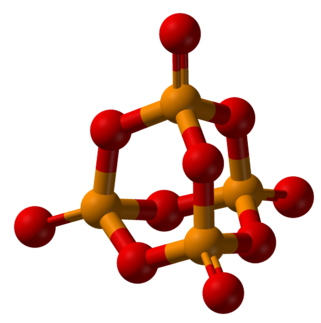
Ball-and-stick representation of chemical bonding in the molecules P4O10 and P4S10.
The theory of chemical bonding has a long history, dating back to ancient Greece and the atomists Democritus, Leucippus, and the Epicureans. They postulated the existence of immutable atoms moving through the void, and envisioned the physical properties of materials as arising from the kinds and shapes of atoms. In his epic poem De rerum natura (On the Nature of Things), the Roman poet Lucretius (c. 99 BC – c. 50 BC), drawing on his Epicurean beliefs, describes some atoms and chemical bonding in the following way:
What seems to us the hardened and condensed
Must be of atoms among themselves more hooked,
Be held compacted deep within, as 'twere
By branch-like atoms -- of which sort the chief
Are diamond stones, despisers of all blows
And stalwart flint and strength of solid iron
And brazen bars, which, budging hard in locks,
Do grate and scream. But what are liquid, formed
Of fluid body, they indeed must be
Of elements more smooth and round -- because
Their globules severally will not cohere.
Lucretius' poem is enjoyable reading and contains some remarkable insights into the microscopic world, given the tools available at the time. Modern analytical methods show that he was off base with his ideas about hooks and spheres, however. We will revisit the nature of chemical bonding in the substances Lucretius mentions (diamond, silicates, iron, brass, and water) in the context of modern chemical theories to understand why they have the special properties they do.


