RO11. Redox in Organic Carbonyls
- Page ID
- 4348
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)The idea of oxidation states is not normally applied to organic compounds, but it can be useful to do so. When we do, we can gain some insight into certain reactions of organic molecules.
For example,carbon dioxide, CO2, can be thought of as having carbon in an oxidized state. If we apply the usual oxidation state rule, carbon dioxide is overall neutral and contains two oxygens, each with 2- charge. To counter that charge, carbon must be in oxidation state 4+.
On the other hand, methane, CH4, can be thought of as having carbon in a reduced state. If we apply the usual oxidation state rule here, methane is overall neutral but contains four protons. That means the carbon must be in a 4- oxidation state. Of course, the carbon does not behave as if it has a minus four charge. But we will see that this sort of exercise can be useful for book-keeping purposes.
Problem RO11.1.
Assign the formal oxidation state to carbon in the following molecules.
a) methanol, CH3OH b) formaldehyde or methanal, CH2O c) carbonate, CO32- d) hydrogen cyanide, HCN
e) ethane, CH3CH3 f) ethene, CH2CH2 g) ethyne, CHCH
The general trend here is that the more bonds there are to oxygen, the more oxidized is carbon. The more bonds there are to hydrogen, the more reduced is carbon.
Adding a hydrogen nucleophile to a carbonyl electrophile is routinely referred to as a reduction. For example, adding sodium borohydride to methanal would result in reduction to form methanol. Of course, a hydride is really a proton plus two electrons. We could write an equation for the reduction of methanal that looks a lot like the redox reactions we see in a table of standard reduction potentials.
\[ CH_2O + H^- + H^+ \rightarrow CH_3OH \]
or
\[ CH_2O + 2e^- + 2H^+ \rightarrow CH_3OH \]
It stands to reason that the opposite reaction, the conversion of methanol to methanal, is a two electron oxidation.
\[ CH_3OH \rightarrow CH_2O + 2e^- + 2H^+ \]
We know how to accomplish the reduction of methanal, at least on paper. We just add a complex metal hydride, such as lithium aluminum hydride or sodium borohydride, to the carbonyl compound. After an acidic aqueous workup to remove all the lithium and aluminum compounds, we get methanol. For practical reasons, methanol may be difficult to isolate this way, but that's the general idea of the reaction.
How do we accomplish the reverse reaction?
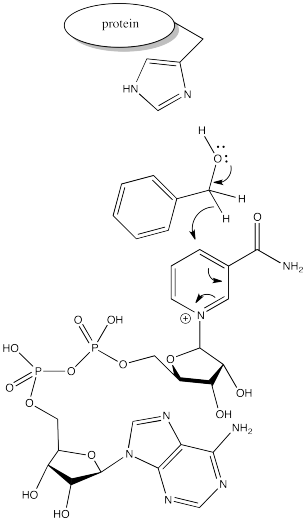
One way would be to provide a hydride acceptor in the reaction, so that we could catch hydride as it comes off the methanol. The most well-known such entity is NAD+, of course. There are biological oxidations that employ NAD+ for this reason.
More generally, the reaction can be accomplished in a number of ways, on paper, by separating out the two tasks involved. We need something to accept the two protons: that's a base. We need something to accept the two electrons: that's an oxidizing agent.
For the latter task, there are a number of high oxidation state transition metal compounds that are quite willing to accept two electrons. One of the most widely employed is Cr(VI), which accepts two electrons to become Cr(IV). A number of other methods are available, having been developed partly to avoid the toxicity of chromium salts, but let's look at the chromium case as an example.
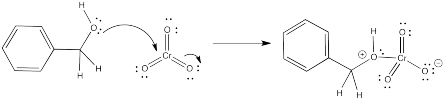
A simple chromium(VI) compound is chromium trioxide. A simple base is pyridine. If we took these two reagents together with benzyl alcohol in a solvent such as dichloromethane, what would happen? OK, you might not want to try this, because chromium trioxide has an alarming capacity to cause spontaneous combustion in organic compounds, but we can do it on paper.
Is chromium trioxide a nucleophile or an electrophile? That Cr(VI) is pretty electrophilic, surely. So what part of the benzyl alcohol is nucleophilic? The oxygen atom. When we mix these things, the oxygen atom would likely coordinate to the chromium.
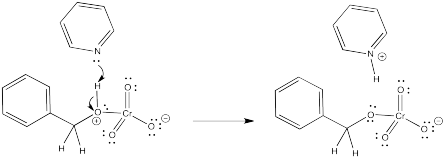
When the oxygen atom coordinates to the chromium, the oxygen gets a positive formal charge. It is now motivated to lose a proton. That's what the pyridine is for.
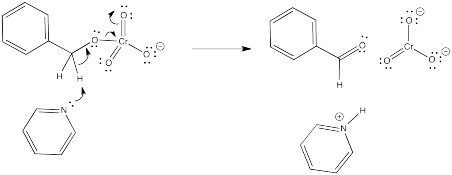
Now we have accomplished one of the goals of the reaction. We have removed a proton from benzyl alcohol. We have one more proton and two electrons left. The second proton will have to come from the carbon attached to the oxygen; that's the place where we need to form a carbonyl.
But wait a minute. You can't take two protons off the same molecule, can you? And certainly not from two atoms that are right next to each other. Doesn't that generate an unstable dianion?
Not this time. The chromium is there to accept two electrons. We won't generate an anion at all, as far as the benzyl alcohol is concerned. It is oxidized to benzaldehyde.
Problem RO11.2.
A completely different outcome to this reaction would be obtained in aqueous solution because of the equilibrium that exists between a carbonyl and the geminal diol (or hydrate) in water. Instead of obtaining an aldehyde, a carboxylic acid would be obtained via a second reduction. Provide a mechanism for this reaction.