2.3B: Uses of TLC
- Page ID
- 93518
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)TLC is a common technique in the organic chemistry laboratory because it can give quick and useful information about the purity of a sample and whether or not a reaction in progress is complete. When low polarity solvents are used, a TLC plate can be complete in less than 5 minutes.
Assessing Purity
One of the uses of TLC is to assess the purity of a sample. In Figure 2.7 are TLC plates of acetophenone and cinnamaldehyde: samples that were diluted from their reagent bottle, run, and visualized with UV light. Acetophenone appeared as only one spot on the TLC plate, indicating the reagent is likely pure. Conversely, cinnamaldehyde is unquestionably impure as its TLC showed two large spots, and had a few fainter spots as well. Aldehydes are prone to air oxidation, and it is common for aldehydes to be found in their reagent bottles alongside with their corresponding carboxylic acids. TLC is one method that can be used to determine how much an aldehyde has degraded.

Assessing Reaction Progress
Use of a Co-Spot
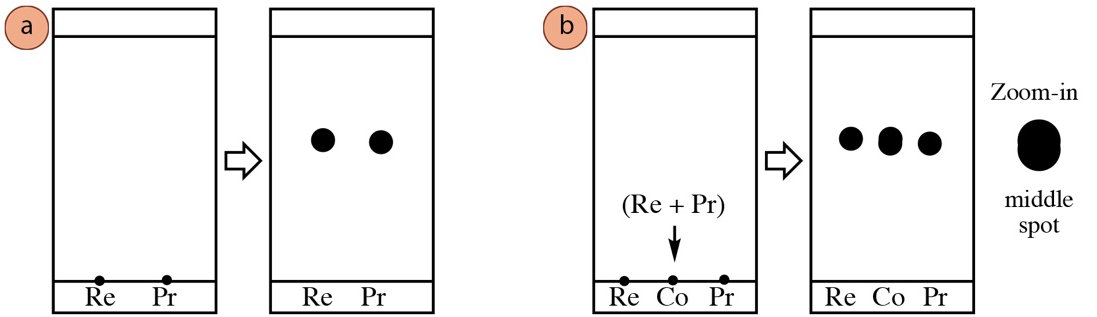
TLC can be used to analyze a chemical reaction, for example to determine if the reactants have been consumed and a new product has formed. A pure sample of the reactant can be spotted in one lane of a TLC, and the product mixture in another lane. Often the central lane is used for reference, where both reactant and product mixture are spotted over top of one another, in what is called the "co-spot."
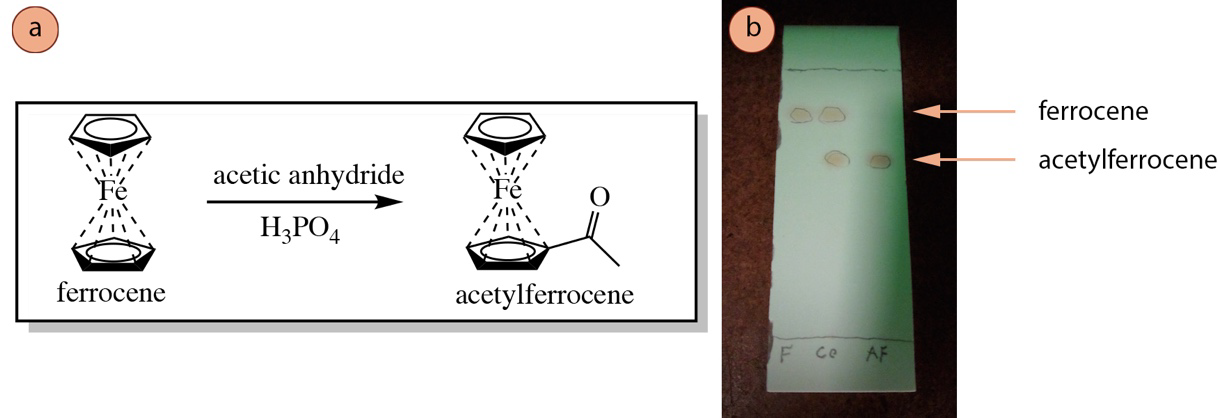
For example, the reaction shown in Figure 2.8a is analyzed by TLC in Figure 2.8b. In the first lane of Figure 2.8b (labeled F) is spotted a pure sample of the reactant ferrocene. In the last lane (labeled AF) is spotted the product mixture, which is assumed to be acetylferrocene. In the central lane (labeled co) is spotted both pure ferrocene and the product mixture. The right-most "AF" lane shows that the reaction appears to be a success: the higher spot of ferrocene is absent (meaning it has been consumed), and a new product spot is present. More tests would have to be done to confirm that the lower spot is the expected product of acetylferrocene, but the TLC results look promising.
The co-spot is used for reference, and can be useful in interpreting certain situations. For example, at times the solvent may run with a slight diagonal, causing identical components to elute to slightly different heights. The co-spot can be useful in tracking these variations. It can also be helpful in identifying spots as different if they have similar \(R_f\) values (a feature difficult to be certain of in Figure 2.9a). Two compounds with slightly different \(R_f\) values eluted over top of one another in the co-spot lane may produce a spot with an elongated shape (Figure 2.9b), making it more obvious they are different compounds. Furthermore, a cospot can be helpful in deciphering a TLC plate if the reaction solvent persists in the product mixture lane (for example if the solvent has a relatively high boiling point like DMF or DMSO), such that the residual solvent has an effect on the component's \(R_f\) values.
Monitoring a Reaction by TLC
TLC can be used to monitor the progress of a reaction. This method is often used in research, and one such journal article reporting its usage is shown in Figure 2.10.
.png?revision=1&size=bestfit&width=407&height=495)
To use TLC in this manner, three lanes are spotted on a TLC plate: one for the limiting reactant, one for the co-spot, and one for the reaction mixture. The goal is to note the disappearance of the limiting reactant in the reaction mixture lane and the appearance of a new product spot. When the limiting reactant has completely disappeared, the chemist deduces that the reaction is complete, and can then be "worked up".
To demonstrate how TLC can be used to monitor a reaction, the transesterification reaction in Figure 2.11 was studied over time (Figure 2.12). In the first lane of each TLC plate (marked "BA") was spotted a dilute sample of the reactant benzyl acetate, while in the third lane of each was spotted the reaction mixture (marked "Pr") at different times. In the central lane (marked "Co" for the co-spot), both benzyl acetate and the reaction mixture were delivered over top of one another.
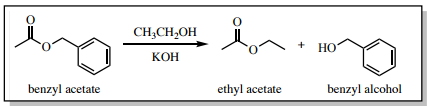
Figure 2.12 shows the progress of the reaction over time. In Figure 2.12a, with the third lane representing the reaction mixture after one minute of mixing, a faint spot near the middle of the plate corresponds to unreacted benzyl acetate. A new spot appears below it, representing the benzyl alcohol product. Over time (Figure 2.12b-e), the top benzyl acetate spot disappears in the reaction mixture lane, and the lower benzyl alcohol spot intensifies. It is apparent from the TLC plates that the reaction was nearing completion at 10 minutes, and was complete at 20 minutes. The TLC demonstrates that the reaction mixture could be worked up after 20 minutes of mixing.
.png?revision=1&size=bestfit&width=1104&height=344)
Obtaining an Aliquot of a Reaction in Progress
To monitor a reaction's progress by TLC, an "aliquot" (or tiny sample) of the reaction mixture is necessary.
If the reaction is run at room temperature or with only mild heating, and the concentration of reactants is conducive to TLC, a capillary spotter can be directly inserted into the flask where the reaction is taking place (Figure 2.13a). A long spotter is ideal if one is available. The aliquot can then be directly spotted on the TLC plate.
If the sample is expected to be UV active, it is a good idea to view the TLC plate under UV light before eluting the plate. If the sample spot is not visible before elution it will not be visible afterwards, as compounds diffuse during elution.
If the sample is expected to be UV active, and only a faint hint of material is seen on the baseline, the material can be deposited multiple times before elution (Figure 2.13b): deliver a small spot of sample on the baseline, and let it fully dry before delivering another spot over top of the first. If the spots are not allowed to dry in between applications, the spot will be too large. Check the plate under UV light again, and if necessary spot more times.
It is important to fully allow a spot to dry on the TLC plate before placement in the TLC chamber. In Figure 2.13c the spot for the reaction mixture (labeled Pr) was not fully dry before elution, and the ethanol solvent likely affected the appearance of the lower \(R_f\) spot. In Figure 2.13d the sample was allowed to fully dry before elution, and the lower \(R_f\) spot was more distinct. Furthermore, residual ethanol was likely the reason why the compound spotted in the reagent lane (labeled "BA") had a different \(R_f\) than it did in the central co-spot lane ("Co").
.png?revision=1&size=bestfit&width=1101&height=328)
To obtain an aliquot of a refluxing solution, briefly remove the condenser and insert a spotter into the reaction mixture (Figure 2.13e). Immediately re-connect the condenser and adjust the clamps while holding the aliquot. Alternatively, lift the flask from the heat source to temporarily cease the reflux before inserting the spotter. The sample may be able to be spotted directly on the TLC plate, but if too concentrated it can be first diluted by running an appropriate solvent (e.g. acetone) through the pipette and into a small vial.


