1.4K: Reflux
- Page ID
- 93228
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Overview of Reflux
A reflux setup (Figure 1.58) allows for liquid to boil and condense, with the condensed liquid returning to the original flask. A reflux setup is analogous to a distillation, with the main difference being the vertical placement of the condenser. The liquid remains at the boiling point of the solvent (or solution) during active reflux.

A reflux apparatus allows for facile heating of a solution, but without the loss of solvent that would result from heating in an open vessel. In a reflux setup, solvent vapors are trapped by the condenser, and the concentration of reactants remains constant throughout the process.
The main purpose of refluxing a solution is to heat a solution in a controlled manner at a constant temperature. For example, imagine that you want to heat a solution to \(60^\text{o} \text{C}\) for one hour in order to conduct a chemical reaction. It would be difficult to maintain a warm water bath at \(60^\text{o} \text{C}\) without special equipment, and it would require regular monitoring. However, if methanol was the solvent, the solution could be heated to reflux, and it would maintain its temperature without regular maintenance at the boiling point of methanol \(\left( 65^\text{o} \text{C} \right)\). True, \(65^\text{o} \text{C}\) is not \(60^\text{o} \text{C}\) and if the specific temperature were crucial to the reaction, then specialized heating equipment would be necessary. But often the boiling point of the solvent is chosen as the reaction temperature because of its practicality.
Step-by-Step Procedures

- Pour the solution to be refluxed into a round bottomed flask, and clamp it to the ring stand or latticework with an extension clamp (Figure 1.59a). The flask should be no more than half full.
- Add a stir bar or few boiling stones for bump prevention. Boiling stones should not be used when refluxing concentrated solutions of sulfuric or phosphoric acid, as they will colorize the solution. For example, when a stir bar is used for bump prevention with concentrated sulfuric acid, the solution remains colorless (Figure 1.59b). When the same reaction is conducted using a boiling stone, the solution darkens during heating (Figure 1.59c) and eventually turns the entire solution a deep purple-brown color (Figure 1.59d).
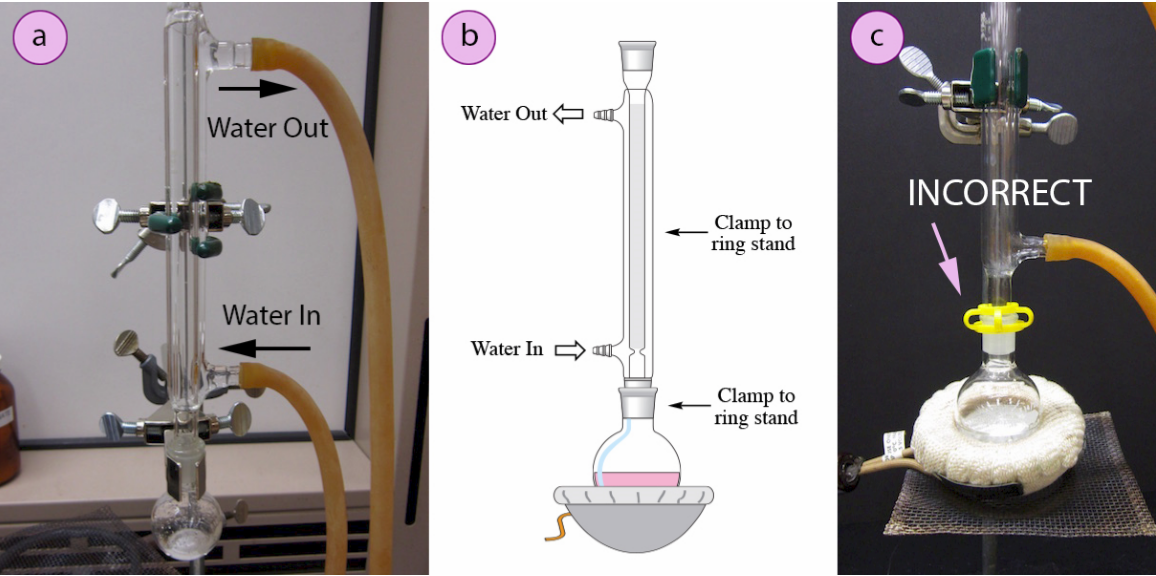
- Place rubber hoses on a condenser (wet the ends first to allow them to slide on), then attach the condenser vertically to the round bottomed flask. If using a tall condenser, clamp the condenser to the ring stand or latticework (Figure 1.60a). Be sure the condenser fits snugly into the flask. Safety note: if the pieces are not properly connected and flammable vapors escape, they may be ignited by the heat source. Do not connect the round bottomed flask and condenser with a plastic clip, as shown in Figure 1.60c. Plastic clips can sometimes fail (especially when they are heated), and this setup does not allow for the flask to be reliably removed from the heat source at the end of the reflux.
- Connect the hose on the lower arm of the condenser to the water faucet and allow the hose on the upper arm to drain to the sink (Figure 1.60b). It is important that water goes in the bottom of the condenser and out the top (so water flows against gravity) or else the condenser will be ineffective as it will not fill completely.

- If multiple solutions will be refluxed at the same time (e.g. if many students are performing a reflux side by side), the hoses from each reflux setup can be connected in series (Figure 1.62). To accomplish this, the upper arm of "Setup A" which normally drains to the sink is instead connected to the lower arm of "Setup B." The upper arm of Setup B then drains to the sink. Connecting apparatuses in series minimizes the use of water, as water exiting one condenser enters the next. Several reflux setups can be connected in series, and the water flow should be monitored to ensure that all setups are adequately cooled.

Figure 1.62: Connecting reflux condensers in series.
- Begin circulating a steady stream of water through the hoses (not so strong that the hose flops around from the high water pressure). Check again that the pieces of glassware securely fit together, then position the heat source under the flask. Turn on the stirring plate if using a stir bar.
- If using a heating mantle, hold it in place with an adjustable platform (e.g. a wire mesh / ring clamp). Allow a few inches below the mantle so when the reaction is complete, the mantle can be lowered and the flask cooled. If the heating mantle is not a perfect fit for the size of the round bottomed flask, surround the flask with sand to create a better contact (Figure 1.63a).
- If using a sand bath, bury the flask in the sand so that sand is at least as high as the level of liquid in the flask (Figure 1.63b).
- If the setup will eventually be left unattended for a long period of time (e.g. overnight), tighten copper wire over the hose attachments to the condenser to prevent changes in water pressure from causing them to pop off.


- If the heat source was preheated (optional), the solution should begin to boil within five minutes. If it does not, increase the rate of heating. The appropriate heating rate occurs when the solution is vigorously boiling and a "reflux ring" is seen roughly one-third of the way up the condenser. A "reflux ring" is the upper limit of where hot vapors are actively condensing. With some solutions (e.g. aqueous solution), the reflux ring is obvious with easily visible droplets in the condenser (Figures 1.64 a+b). With other solutions (e.g. many organic solvents) the reflux ring is subtler, but can be seen with close observation (Figure 1.64c). Subtle movement may be seen in the condenser as liquid drips down the sides of the condenser, or background objects may appear distorted from refraction of light through the condensing liquid (in Figure 1.64d the ring stand pole is distorted).
- If following a procedure in which you are to reflux for a certain time period (e.g. "reflux for one hour"), the time period should begin when the solution is not just boiling but actively refluxing in the bottom third of the condenser.
- The heat should be turned down if the reflux ring climbs to half-way up the condenser or higher, or else vapors could escape the flask.
- After the reflux is complete, turn off the heat source and remove the flask from the heat by either lifting the reflux apparatus up, or dropping the heat source down (Figure 1.65a).

Do not turn off the water flowing through the condenser until the solution is only warm to the touch. After a few minutes of air cooling, the round bottomed flask can be immersed in a tap water bath to accelerate the cooling process (Figure 1.65b).
Summary
 |
 |
 |
 |
|
Pour liquid into the flask along with a stir bar or boiling stones. Use an extension clamp on the round bottomed flask to connect to the ring stand or latticework. Attach the condenser, and connect the hoses so that water travels against gravity (cooling water comes into the bottom and drains out the top). Be sure there is a secure connection between the round bottomed flask and condenser, as vapors escaping this joint have the potential to catch on fire. |
Circulate water through the condenser, then begin heating the flask (by using a heating mantle, sand, water, or oil bath). Use an adjustable platform so the heat can be lowered and removed at the end of the reflux, or if something unexpected occurs. |
Heat so that the "reflux ring" is seen in the lower third of the condenser. Turn down the heat if the refluxing vapors reach higher than halfway up the condenser. |
At the end of the reflux period, lower the heat source from the flask or raise the apparatus. Keep circulating water in the condenser until the flask is just warm to the touch. After air cooling somewhat, the flask can be quickly cooled by immersing in a container of tap water. |
Table 1.9: Procedural summary for reflux.


