3.7: Types of chiral molecules
- Page ID
- 531786
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Often, an asymmetric carbon is the cause of molecular chirality. There are exceptions: i) some molecules have asymmetric carbons, yet they are achiral, ii) some molecules do not have any chirality center, yet they are chiral, iii) some chiral molecules have chirality centers that are atoms other than carbon. The molecules that are chiral due to the presence of an asymmetric carbon are described in the following. The exceptions are described in between where they are relevant or at the end of this section.
Chiral molecules containing one asymmetric carbon
Compounds containing one chiral center are chiral. They have a stereoisomer called an enantiomer, which is their non-superposable mirror image.
Enantiomers have the same physical and chemical properties, except when they react with or interact with other chiral things. Pure enantiomers have a characteristic specific rotation \([\alpha]^{T}_{\lambda}\) that has a + sign for dextrorotatory and an equal amount but a - sign for levorotatory enantiomer.
For example, hands and gloves are chiral objects. The right-hand glove fits on the right hand, and the left-hand glove fits on the left. Similarly, enantiomer molecules react equally, and when they are produced in a chemical reaction, they are made in equal amounts, except when one of the reagents is chiral. For example, enzymes are chiral reagents and usually react with one of the two enantiomers preferentially or exclusively or produce one of the two enantiomers preferentially or exclusively. Enantiomers of some chiral molecules containing one asymmetric carbon are shown below, along with their IUPAC names and physical properties, which are common for the enantiomer pair. Their differences when they interact with chiral entities are described later.
|
(2S)-2-chlorobutane \([\alpha]^{20}_D\) = -12.9 |
(2R)-2-chlorobutane \([\alpha]^{20}_D\) = +12.9 |
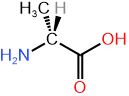
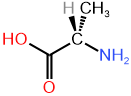
(2R)-2-aminopropanoic acid (D-alanine) \([\alpha]^{20}_D\) (in water)= -2.8 |
(2S)-2-aminopropanoic acid (L-alanine) \([\alpha]^{20}_D\) in water= +2.8 |
(2R)-2,3-dihydroxypropanal (D-glyceraldehyde) \([\alpha]^{25}_D\) = +8.7 |
(2S)-2,3-dihydroxypropanal (L-glyceraldehyde) \([\alpha]^{25}_D\) = -8.7 |
| Densy 0.873 \(\frac{g}{cm^{3}}\), mp -140 \(^o\)C, bp 70 \(^o\)C | Density 1.424 \(\frac{g}{cm^{3}}\), mp 258 \(^o\)C, bp 415 \(^o\)C sublime | Density 1.455 \(\frac{g}{cm^{3}}\), mp 145 \(^o\)C, bp 228 \(^o\)C | |||
Chiral molecules containing more than one asymmetric carbon
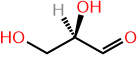
A chirality center has two configurations. Therefore, a molecule having one chirality center has two stereoisomers which are enantiomers of each other. If there are more tahn one chirality centers, each one can have two possible configurations, resulting in \(2^n\) possible stereoisomers, where \(n\) is the number of chiral centers. For example, glyceraldehyde has one chiral center and (\(2^1 = 2\)) two stereoisomers: D-glyceraldehyde and L-glyceraldehyde., as shown in the previous table. Glyceraldehyde is a monosaccharide, i.e., a type of sugar. Erythrose is another monosaccharide that has two chirallity centers and (\(2^2 = 4\)) four stereoisomers, as shown in Figure \(\PageIndex{1}\).
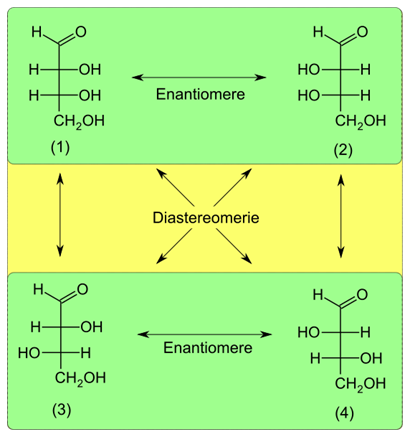
Each chiral compound can have one enantiomer. D-erythrose and L-erythrose are enantiomers, and the other two, i.e., D-threose and L-threose are diastereomers to them. Similarly, D-threose and L-threose are enantiomers of each other and D-erythrose and L-erythrose are their diastereomers, as illustrated in Figure \(\PageIndex{1}\).
Stereoisomers that are not enantiomers are diastereomers of each other.
D-Ribose is another monosaccharide found in DNA, that has four chirality center and (\(2^3 = 8\)) possible stereoisomers, shown in Figure \(\PageIndex{2}\). Each has one enantiomer and six diastereomers. For example, L-ribose is an enantiomer of D-ribose and the other six are their diastereomers.

D/L Stereodescriptors pre-date R/S stereodescriptors in IUPAC nomenclature, but they are still commonly used in biochemistry for naming monosaccharides, which are monomers of carbohydrates, and \(\alpha\)-amino acids, which are monomers of proteins. D/L are designated by relating to the Fisher projection of glyceraldehyde drawing by placing aldehyde \(\ce{-CHO}\) on the top of the vertical line. If the \(\ce{-OH}\) of the penultimate \(\ce{C}\), i.e., second from the bottom end, is pointing towards right, it is designated dexter (D), which in Latin means left); and if it is pointing towards left, it is designate laevus (L) meaning left, as shown in Figure \(\PageIndex{3}\). For other monosaccharides having more than one chirality centers, D/L defines the configuration of penultimate \(\ce{C}\). The configuration of the rest of the chirality centers is implicit in the name, e.g., D-ribose and its isomers shown in Figure \(\PageIndex{2}\). All naturally occuring monosaccharides have D-configuration.
 D-glyceraldehyde
D-glyceraldehyde L-glyceraldehyde
L-glyceraldehyde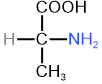 D-alanine
D-alanine L-alanine
L-alanineIn \(\alpha\)-amino acids, \(\alpha ce{C}\) bears \(\ce{-COOH}\) and \(\ce{-NH2}\) groups. In a Fisher projection, \(\ce{-COOH}\) is placed on the top of vertical line and if \(\ce{-NH2}\) is pointing towards right, it is designated D and if pointing towards left, it is designated L, as shown in Figure \(\PageIndex{3}\). All aminoacids present in proteins have L-configuration.
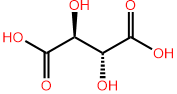
The formula \(2^n\) tells the maximum possible number of stereoisomers of compounds having \(n\) chiral centers. The actual number may be less than the maximum. For example, tartaric acid has two chiral centers and only three stereoisomers: (+) tartaric acid and (-) tartaric acid, which are enantiomers, and meso-tartaric acid, which is achiral, as illustrated in Figure \(\PageIndex{4}\). The third isomer is achiral because it has a symmetry plane that divides it into two equal halves, which are mirror images of each other. Figure \(\PageIndex{5}\) and Figure \(\PageIndex{6}\) are interactive modules of tartaric acid provided as exercises to verify that the enantiomers are nonsuperposable and the meso isomer has a superposable mirror image.
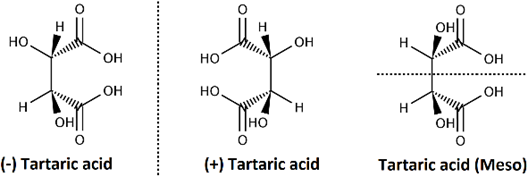
(2S,3S)-2,3-dihydroxybutandioic acid
(2R,3R)-2,3-dihydroxybutandioic acid
(2R,3S)-2,3-dihydroxybutandioic acid
(2R,3S)-2,3-dihydroxybutandioic acid
A meso compound is i) an achiral compound with two or more chiral centers, ii) optically inactive, and iii) has a plane of symmetry that divides it into two mirror-image halves.
Enantiomers have the same physical and chemical properties, except when interacting with chiral objects, chiral reagents, or chiral plane polarized light, where they exhibit different properties. Diastereomers have different physical and chemical properties, as shown in the table below for stereoisomers of tartaric acid: enantiomers (2S,3S)-2,3-dihydoxybutandioic acid and (2R,3R)-2,3-dihydoxybutandioic acid and their diastereomer (2R,3S)-2,3-dihydoxybutandioic acid.
| Stereoisomer |  |
 |
 |
| \([\alpha]^{25}_D\) | -12.0 | +12.0 | 0 |
| IUPAC Name | (2S,3S)-2,3-dihydoxybutandioic acid | (2R,3R)-2,3-dihydoxybutandioic acid | (2R,3S)-2,3-dihydroxybutandioic acid |
| Density (\(\frac{g}{cm^3}\)) | 1.73 | 1.73 | 1.886 |
| Melting point (\(^o\)C) | 169-172 | 169-172 | 165-166 |
| Solubility in water (\(\frac{g}{ml}\)) at 25 \(^o\)C | 1.33 | 1.33 | 1.25 |
| \(pK_{a1}\) (it represent acidity) | 2.89 | 2.89 | 3.22 |
| \(pK_{a2}\) (it represent acidity) | 4.40 | 4.40 | 4.45 |
Disubstituted cycloalkanes usually have two chiral centers and can have four (\(2^2 =4 \)) stereoisomers, i.e., two pairs of enantiomers. If two substituents are different, usually all stereoisomers are observed. For example, 1-chloro-2-methylcyclohexane has four stereoisomers shown in Figure \(\PageIndex{7}\), where the first two are trans enantiomers and the second two are cis enantiomers, which are diastereomers of the first two.
 a) (1S,2R)-1-chloro-2-methylcyclohexane
a) (1S,2R)-1-chloro-2-methylcyclohexane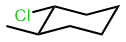 b) a) (1R,2S)-1-chloro-2-methylcyclohexane
b) a) (1R,2S)-1-chloro-2-methylcyclohexane c) (1R,2R)-1-chloro-2-methylcyclohexane
c) (1R,2R)-1-chloro-2-methylcyclohexane d) (1S,2S)-1-chloro-2-methylcyclohexane
d) (1S,2S)-1-chloro-2-methylcyclohexaneIf two substituents in disubstituted cycloalkanes are identical, they usually have three stereoisomers: a pair of enantiomers and a meso form, as illustrated in Figure \(\PageIndex{8}\) with the example of 1,2-dimethylcyclohexane.
 a) (1S,2S)-1,2-dimethylcyclohexane
a) (1S,2S)-1,2-dimethylcyclohexane b) a) (1R,2R)-1,2-dimethylcyclohexane
b) a) (1R,2R)-1,2-dimethylcyclohexane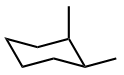 c) (1R,2S)-1,2-dimethylcyclohexane
c) (1R,2S)-1,2-dimethylcyclohexane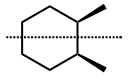 d) (1R2S)-1,2-dimethylcyclohexane
d) (1R2S)-1,2-dimethylcyclohexaneThe enantiomers of trans and cis conformations, Figure \(\PageIndex{7}\) and trans conformation in Figure \(\PageIndex{8}\), may appear identical by flipping the molecule horizontally. It is not so because flipping converts the bonds pointing away from the viewer behind the page to pointing towards the viewer above the page. It is suggested to use the Interactive 3D model of enantiomers of trans-1,2-dimethylcyclohexane presented below to verify that they are nonsuperposable.
(1S,2S)-1,2-dimethylcyclohexane
(1R,2R)-1,2-dimethylcyclohexane
The symmetry plane is not evident in every conformation. It becomes obvious when the most symmetric conformation is drawn. For example, eclipsed conformation of meso tartaric acid in which every group on one stereogenic \(\ce{C}\) is eclipsing the same group on the second stereogenic \(\ce{C}\) is the most symmetric conformation, showing symmetry plane as in Figure \(\PageIndex{4}\). The most symmetric conformation of cycloalkanes is their planar conformation. For example, Figure \(\PageIndex{8}\) d) is the most symmetric conformation of the meso form of 1,2-dimethylcyclohexane, showing the symmetric plane as a dotted line in it.
1,2-Disubstituted cyclohexanes that have an even number of atoms in the ring and identical substituents across the ring are achiral and do not have any chirality center. The plan of symmetry passes through the substituents, as observed in the examples shown below, which are presented in their most symmetric planar conformations. The cis and trans isomers, which are diastereomers, still exist.
 trans-1,4-dichlorocyclohexane
trans-1,4-dichlorocyclohexane cis-1,4-dichlorocyclohexane
cis-1,4-dichlorocyclohexane trans-1,3-dichlorocyclobutane
trans-1,3-dichlorocyclobutane cis-1,3-dichlorocyclobutane
cis-1,3-dichlorocyclobutaneChiral compounds without achirallty center
Chiral allenes
Allenes are conjugated alkenes that have an \(sp\)-\(\ce{C}\) in the middle making a \(\pi\)-bond to an \(sp^2\)-\(\ce{C}\) on one side and a second \(pi\)-bond to an \(sp^2\)-\(\ce{C}\) on the other side. Since the two \(p\)-orbitals of \(sp\)-\(\ce{C}\) are perpendicular to each other, the two \(\pi\)-bonds are perpendicular to each other. Similarly, the two substituents on one \(\sp^2\)-\(\ce{C}\) are in a V shape, perpendicular to the two substituents in a V shape on the other \(\sp^2\)-\(\ce{C}\), as illustrated Figure \(\PageIndex{9}\). If two substituents on each \(sp^2\)-\(ce{C}\) are different, e.g., a and b on one and a' and b' ( a & a' can be the same, and b and b' can be the same), the molecule is chiral. For example, penta-2,3-diene, shown in Figure \(\PageIndex{9}\), is a chiral molecule without any chirality center.
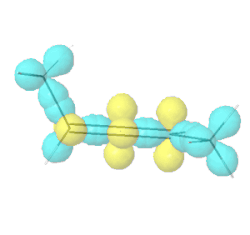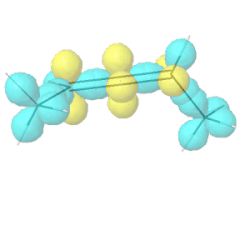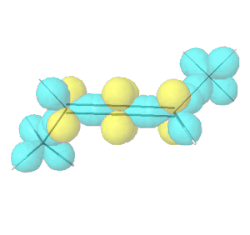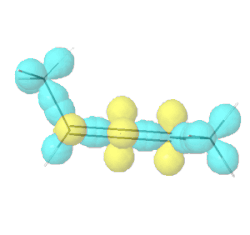 Hybrid atomic orbitals
Hybrid atomic orbitals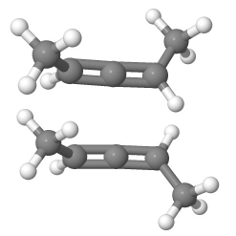 Models of enantiomers
Models of enantiomers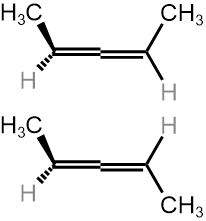 Perspective drawings of enantiomers
Perspective drawings of enantiomersChiral conformations

 Achiral molecules may have chiral conformers. For example, butane has gauche, anti, and eclipsed conformers, as shown on the right margin. The anti and eclipsed are achiral because they possess a plane of symmetry. Still, the gauche is chiral, lacking any plane or point of symmetry. Enantiomers of gauche conformers of butane are illustrated in the figure on the right margin. The gauche enantiomers are practically not separable due to rapid inversion through free rotation around single bonds.
Achiral molecules may have chiral conformers. For example, butane has gauche, anti, and eclipsed conformers, as shown on the right margin. The anti and eclipsed are achiral because they possess a plane of symmetry. Still, the gauche is chiral, lacking any plane or point of symmetry. Enantiomers of gauche conformers of butane are illustrated in the figure on the right margin. The gauche enantiomers are practically not separable due to rapid inversion through free rotation around single bonds.
The separation of enantiomers from their mixture is called the resolution of enantiomers. Molecules that have chiral conformations or structures, but their enantiomers can not be resolved, are considered achiral.
For example, butane is achiral, having two gauche conformers, which are enantiomers, but they cannot be resolved.
Atropisomers
In some molecules, the chiral conformers are locked due to severely restricted rotation around the single bond. The rotation is usually restricted because the geometry and size of the substituents (steric factors) prevent rotation.
The molecules in which chiral conformations are locked due to restricted rotation around single bonds, and as a result, the enantiomers can be resolved, are chiral, and the enantiomers are known as atropisomers.
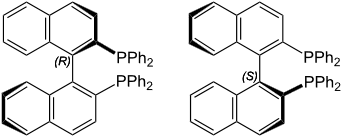
 For example, ([1,1′-Binaphthalene]-2,2′-diyl)bis(diphenylphosphane), commonly known as BINAP, exhibits atropisomerism due to restricted rotation around the single bond between naphthyl groups as shown in the figure on the right margin (copyright: Public domain). It is an example of a chiral compound without a chirality center in it. Its atoropisomers, (R)-BINAP and (S)-BINAP, are resolved and used as ligands to synthesize chiral catalysts, which, in turn, are employed for some enantioselective organic syntheses. 2,2'-Dibromo-6,6'-diiodo-1,1'-biphenyl, shown in the left margin (copyright: public domain), is another example that exhibits atropisomerism.
For example, ([1,1′-Binaphthalene]-2,2′-diyl)bis(diphenylphosphane), commonly known as BINAP, exhibits atropisomerism due to restricted rotation around the single bond between naphthyl groups as shown in the figure on the right margin (copyright: Public domain). It is an example of a chiral compound without a chirality center in it. Its atoropisomers, (R)-BINAP and (S)-BINAP, are resolved and used as ligands to synthesize chiral catalysts, which, in turn, are employed for some enantioselective organic syntheses. 2,2'-Dibromo-6,6'-diiodo-1,1'-biphenyl, shown in the left margin (copyright: public domain), is another example that exhibits atropisomerism.
\(\ce{N}\), \(\ce{P}\), and \(\ce{S}\) Chirality centers
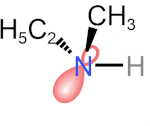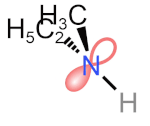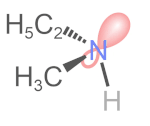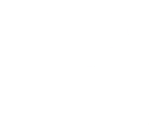 Tetrahedral \(\ce{N}\), \(\ce{P}\), and \(\ce{S}\) are common in organic compounds and can be chiral centers. \(\ce{N}\) in amines is tetrahedral with a lone pair and three other substituents. According to CIP sequence rules, a lone pair is a substituent of the lowest priority. If the other three substituents on the amine nitrogen are different, it is a chirality center. For example, \(\ce{N}\) in ethyl methyl amine is a chirality center, but its (R) and (S) configurations rapidly interconvert at room temperature, as shown in the simulation on the right margin (Copyright: Public domain).
Tetrahedral \(\ce{N}\), \(\ce{P}\), and \(\ce{S}\) are common in organic compounds and can be chiral centers. \(\ce{N}\) in amines is tetrahedral with a lone pair and three other substituents. According to CIP sequence rules, a lone pair is a substituent of the lowest priority. If the other three substituents on the amine nitrogen are different, it is a chirality center. For example, \(\ce{N}\) in ethyl methyl amine is a chirality center, but its (R) and (S) configurations rapidly interconvert at room temperature, as shown in the simulation on the right margin (Copyright: Public domain).
- This process of lone pair of \(\ce{N}\) in an \(sp^3)\) orbital changing to a \(sp^2)\) transition state with lone pair in a \(p\) orbital and then to \(sp^3\) orbital in an inverted configuration is called nitrogen inversion or pyramidal inversion.
- Since the enantiomers of amines with a \(\ce{N}\) chirality center are not resolvable, they are considered achiral molecules.
Quaternary ammonium compounds having four different substituents are chiral. For example, the enantiomers of the quaternary ammonium salt, shown in Figure \(\PageIndex{10}\), have been synthesized and resolved. Since there is no lone pair on \(\ce{N}\) of quaternary ammonium compounds, there is no inversion of configuration.
Phosphines are similar to amines with \(\ce{P}\) in the place of \(\ce{N}\). If three substituents of phosphine are different, it is a chiral center. It undergoes inversion like amines, but it is slower, taking a few hours. Therefore, phosphines with three different substituents are chiral molecules. For example, the enantiomers of chiral methylpropylphenylphosphine, shown in Figure \(\PageIndex{10}\), have been resolved.
The sulfur atom in sulfoxides, sulfones, and sulfates is tetrahedral and chiral if the substituents on the \(\ce{S}\) are all different. The configuration \(\ce{S}\) chirality center is relatively stable up to several days, and the enantiomers can be resolved, making these compounds chiral. A well-known example is coenzyme S-adenosyl methionine, a methylating agent involved in numerous metabolic pathways. \(\ce{S}\) in S-adenosyl methionine has (S) configuration, as shown in Figure \(\PageIndex{10}\),
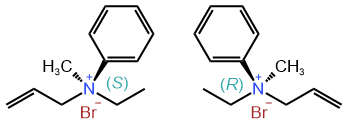
Allyl ethyl methyl phenyl ammonium bromide
 Methylpropylphenylphosphine
Methylpropylphenylphosphine
S-Adenosyl methionine
E/Z Stereodescriptors for double bond systems
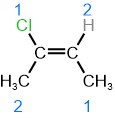
R/S stereodescriptors define the configuration of chiral centers. In the case of polysubstituted cycloalkanes, cis/trans stereodescriptors are used to indicate the relative orientation of two substituents on the same side (cis) or the opposite sides (trans); but have been replaced with R/S stereodescriptors in IUPAC nomenclature. However, cis/trans isomers of alkene and related double bond systems do not have chirality centers. The two stereogenic centers of alkenes usually have one \(\ce{-H}\) and another substituent. Cis/trans usually refers to the relationship of substituents other than \(\ce{-H}\). It becomes confusing when both substituents are other than \(\ce{-H}\). For example, if the structure shown on the right margin is named cis-2-chlorobut-2-ene, it is not clear \(\ce{-Cl}\) on one and \(\ce{-CH3}\) on the other stereogenic carbons are cis or \(\ce{-CH3}\) on one and \(\ce{-CH3}\) on the other are cis. To avoid this confusion, IUPAC has replaced cis/trans with E/Z stereodescriptors for double-bond systems, such as alkenes.
The following steps assign E or Z to the diastereomers of the double bond system.
- Assign priorities 1 and 2 to the two substituents of one stereogenic center using CIP priority, where 1 is higher priority and 2 is lower.
- Do the same to the second stereogenic center.
- If the two high-priority (#1) substituents on the two stereogenic centers are on the same side, assign zusammen (Z), which means together (cis) in German; otherwise, assign entgegen (E), which in German means opposite (trans). The E/Z capital, italicized, preceded by its locant, placed in brackets before the name, and separated by a hyphen from the name.
- If there is more than one double bond systems, the corresponding E and/or Z are preceded by the locant number and separated by a comma.
Figure \(\PageIndex{9}\) provides examples of the applications of the above rules.