2.1: Drawing organic structures
- Page ID
- 523122
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Lewis structure
The molecular formula tells the elemental composition of a compound. For example, \(\ce{C5H10O2}\) is a molecular formula. Multiple Lewis structures, representing different compounds, may have the same formula.
The compounds that have the same molecular formula but different structures are isomers of each other.
Lewis structure, also called Kekulé structure or a fully expanded formula, shows bonds between atoms as lines and lone pairs as dots. For example, pentanoic acid, 2-methyl butanoic acid, and 2,2-dimethylpropanoic acid are isomers having the same molecular formula \(\ce{C5H10O2}\), as shown in Figure \(\PageIndex{1}\)
 Pentanoic acid
Pentanoic acid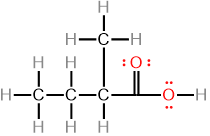 2-Methylbutanoic acid
2-Methylbutanoic acid 2,2-Dimethylpropanoic acid
2,2-Dimethylpropanoic acidMajor drawbacks of Lewis structures are (i) they are not practical for large molecules, and (ii) they do not accurately represent the bond angles.
Condensed formula
The condensed formula simplifies the Lewis structure.
- A condensed formula is written as a sequence of central atoms in the parent chain: each central atom is followed by its peripheral atoms other than the atoms of the parent chain.
- A branch is shown either connected to the corresponding atom of the chain by a line or
- grouped in central brackets written next to the corresponding central atom, like peripheral atoms.
- Lone pairs are often omitted, but may be shown when needed. It is understood that with zero formal charge, \(\ce{C}\) has no none pair, \(\ce{N}\) has one, and \(\ce{O}\) has two.
For example, the condensed formula of pentonic acid (Figure \(\PageIndex{1}\)) is \(\ce{CH3CH2CH2CH2COOH}\), 2-methylbutanoic acid is \(\ce{CH3CH2\!\!\!\!\!\!\!\!\!\overset{\overset{\huge \;\;\;\;\; CH{\Large _3}}|}{C}\!\!\!\!\!\!\!\!\!HCOOH}\) or \(\ce{CH3CH2CH(CH3)COOH}\), and 2,2-dimethylpropanoic acid is \(\ce{CH3\!\!\!\!\!\!\!\!\!\overset{\overset{\huge \;\;\;\;\; CH{\Large _3}}|}{\underset{\underset{\huge \;\;\;\;\; CH{\Large _3}}|}{C}}\!\!\!\!\!\!\!\!\!COOH}\) or \(\ce{CH3C(CH3)2COOH}\). Note that the repeat \(\ce{H3C{-}}\) groups in the last example are placed in the same center bracket with a subscript indicating the repeat number. The same can be applied to the repeat \(\ce{CH2}\) groups. For example, the condensed formula of pentonic acid may be written as \(\ce{CH3(CH2)3COOH}\).
Small variations in the condensed formulas are acceptable if needed, as long as they represent the same structure. For example, the condensed formula of 2,2-dimethylpropane may be written \(\ce{CH3C(CH3)2COOH}\) or as \(\ce{(CH3)3CCOOH}\). Similarly, some sections of the condensed formula may be shown in expanded form for emphasis, if needed. For example, the condensed form of pentanoic acid may be written as \(\ce{CH3(CH2)3-\overset{\!\!\overset{\huge {\:O}}|\!\!|\,}{C}-OH}\) showing more details of carbon-to-oxygen bonds.
Skeletal formula
The skeletal formula is based on the fact that \(\ce{C-C}\) bonds in the parent chain of carbons can be placed on the plane of a page as a zig-zag line, as shown by the models of pentanoic acid, 2-methyl butanoic acid and 2,2-dimethylpropanoic in Figure \(\PageIndex{2}\).
 Pentanoic acid
Pentanoic acid 2-Methylbutanoic acid
2-Methylbutanoic acid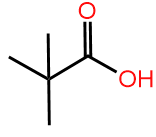 2,2-Dimethylpropanoic acid
2,2-Dimethylpropanoic acidHow to draw a skeletal formula?
- Omit \(\ce{C}\) and \(\ce{H}\) from the model of the molecule, keeping the \(\ce{C-C}\) bonds as zig-zag lines, while maintaining heteroatoms, and hydrogens on the heteroatoms, results in the skeletal structure of the compound.
- If there is any \(sp\) hybridized atom, show bonds around it at 180°, because the zig-zag line accommodates \(sp^3\) and \(sp^2\) hybridized atoms, but \(sp\) hybridized atoms have a linear geometry around them.
Examples of the first rule are the skeletal structures of pentanoic acid, 2-methylbutanoic acid, and 2,2-dimethylpropanoic acid shown in Figure \(\PageIndex{2}\). Examples of rule 2 are 2-hexyne, having two \(sp\) carbons (a triple bond), along with 2-hexene, having two \(sp^2\) carbons (a double bond), and hexane, having all \(sp^3\) atoms are shown in Figure \(\PageIndex{3}\). Note that four atoms on and around the triple bond are in a linear geometry.
 2-Hexyne
2-Hexyne 2-Hexene
2-Hexene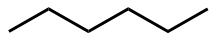 Hexane
HexaneRecall that the tetrahedral geometry of four bonds on a carbon is two Vs, perpendicular to each other and joined at the vertex. If one V is placed in the plane of the page, one bond of the other V is protruding outward to the viewer, and the other is recessing away from the viewer. The third and the fourth bonds on a tetrahedral carbon are shown as solid lines in the skeletal drawing of 2-methylbutanoic acid, and 2,2-dimethylpropanoic acid in Figure \(\PageIndex{2}\). This drawing does not distinguish whether the third or the fourth bond shown is protruding outward or recessing inward. If needed, it is shown as a solid wedge for the bond protruding outward or a hashed wedge for a bond recessing inward. For example, there are two isomers of 2-methylbutanoic acid: one with the 2-methyl bond protruding outward and the othe with 2-methyl recessing inwords when the molecule is drawn left to right with \(\ce{ -\overset{\!\!\overset{\huge {\:O}}|\!\!|\,}{C}-OH}\) on the right. This is distinguished in the skeletal drawing by converting the relevant portion of the drawing into a perspective drawing with the use of solid or hashed wedges, as shown in Figure \(\PageIndex{4}\). The solid or hashed wedges are not used in the skeletal drawing of 2,2-dimethylbutanoic acid because both bonds are the same.
 (2R)-2-Methylbutanoic acid
(2R)-2-Methylbutanoic acid (2S)-2-Methylbutanoic acid
(2S)-2-Methylbutanoic acid 2,2-Dimethylbutanoic acid
2,2-Dimethylbutanoic acidVariations in the structural formula
A single bond is a \(\sigma\) bond symmetrical around the bond axis, allowing rotation of one bonded atom from 0° to 360° relative to the other in most cases. Different structures obtained by rotating around a single bond are not different compounds; they are called conformations of the same compound. Different conformations have different energies and exist in equilibrium with each other, where the most stable conformation is the most predominant in the mixture. The zig-zag line often used in skeletal structures to represent \(\ce{C-C}\) chain, as in Figure \(\PageIndex{5}\)a, is usually the most stable conformation. However, it can be drawn in multiple ways that may result from rotation around any single bond. For example, n-hexane shown in Figure \(\PageIndex{5}\)a is rotated around the middle \(\ce{C-C}\) bond to acquire new confirmation shown in Figure \(\PageIndex{5}\)b and rotated further along the next \(\ce{C-C}\) to acquire another confirmation shown in Figure \(\PageIndex{5}\)c. All three structural formulas are shown in Figures \(\PageIndex{5}\)a, b, and c are correct skeletal structures representing the same molecule.
a)





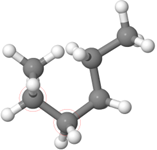
Lone pairs are usually omitted but may be shown if needed. Similarly, a hybrid of skeletal formula and condensed or fully expanded formula is also used if required. For example, Figure \(\PageIndex{5}\)a shows the skeletal formula of pentanoic acid ((\(\ce{CH3CH2CH2CH2COOH}\), its variations shown in Figure \(\PageIndex{5}\)b and Figure \(\PageIndex{5}\)c are also acceptable and may be used if needed.
 a)
a) b)
b) c)
c)How to interpret a skeletal formula
- If an element symbol is not shown at the end or bend of a line in the skeletal structure, it is a \(\ce{C}\).
- Since \(\ce{C}\) has four bonds, \(\ce{H}\) on the \(\ce{C}\) = 4 - number of lines on the \(\ce{C}\).
- Heteroatoms and \(\ce{H}\) on them, if present, are shown in the skeletal formula.
- Lone pairs, if present, can be figured out from the formal charge and the number of bonds on an atom. The following is a summary of number of bonds, formal charges, and nubmer of lone pairs for elements commonly found in organic molecules: \(\ce{-\overset{|}{\underset{|}{C}}-}\), \(\ce{-\overset{+}{\underset{|}{C}}-}\), \(\ce{-\!\!\!\overset{\!\huge{\;\;\;\cdot\cdot\,\small -}}{\underset{|}{C}}\!\!\!-}\), \(\ce{-\overset{\!\huge{\;\cdot\cdot\,}}{\underset{|}{N}}-}\), \(\ce{-\!\!\overset{|}{\underset{|}{\;\;N^+}}\!\!-}\), \(\ce{-\!\!\!\overset{\!\huge{\;\;\;\cdot\cdot\,\small -}}{\underset{\!\huge\:\cdot\cdot}{N}}\!\!\!-}\), \(\ce{-\overset{\!\huge\:\cdot\:\!\cdot}{\underset{\!\huge\:\cdot\:\!\cdot}{O}}-}\), \(\ce{-\!\!\!\overset{\!\huge{\;\;\;\cdot\cdot\,\small +}}{\underset{|}{O}}\!\!\!-}\), \(\ce{-\!\!\!\overset{\!\huge{\;\;\;\cdot\cdot\,\small -}}{\underset{\!\huge\:\cdot\cdot}{O}}\!\!\!\!\Large:}\), \(\ce{-\overset{\huge{\cdot\cdot}}{\underset{\!\huge\:\cdot\cdot}{X}}\!\Large:}\) (where \(\ce{X}\) is a halogen \(\ce{F}\), \(\ce{Cl}\), \(\ce{Br}\), or \(\ce{I}\)),\(\ce{-H}\), and \(\ce{\overset{\!\huge{\;\;\;\cdot\cdot\,\small -}}{H}}\).
For example, the skeletal formula of the antibiotic amoxicillin is shown in Figure \(\PageIndex{6}\) (left) with \(\ce{C}\) locations labeled by green numbers by rule#1, the numbers are the number of \(\ce{H}\) on the carbon calculated by rule#2 as shown in the middle figure, heteroatoms are already showing on the skeletal formula according to rule#3, and lone pairs on them are determined by rule#4, as shown in the fully expanded formula in \(\PageIndex{6}\) (right).
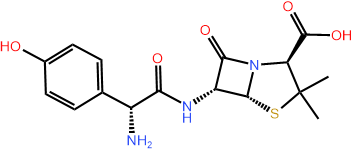

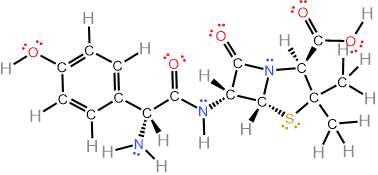
Note that the skeletal drawing of amoxicillin is showing cyclic structures (a six-member ring on the right, and a four-member ring fused with a five-member ring on the left of the formula) and a 3D drawing (perspective drawing) where needed by hashed or solid wedges. Two more examples are shown in Figure \(\PageIndex{7}\) where skeletal structure of retinol, i.e., vitamin A1 needed for vision and several other functions, and of cholesterol that is essential for several functions but also related with cardiovascular diseases, are shown along with their semi-condensed formulas showing all \(\ce{C}\), \(\ce{H}\) and lone pair. Note that the perspective drawing is shown where needed by using solid or hashed wedges.
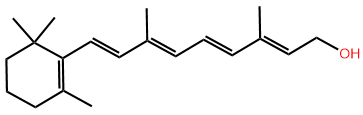
 Retinol
Retinol
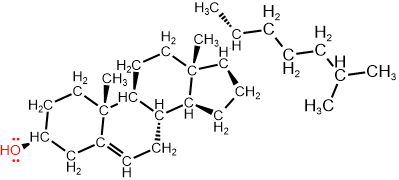 Cholesterol
Cholesterol

