8.6: What is aromaticity and how do we recognize aromatic systems?
- Page ID
- 359140
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)
To recap, benzene is uniquely stable and it is relatively difficult to get it to react, despite the high electron density in the ring. It is significantly more stable than cyclohexadiene which is conjugated, but only contains two bonds. There is clearly something special about a conjugated ring system above and beyond simple conjugation properties.

If we examine other systems, we can begin to find the parameters that govern the property of aromaticity. We can begin by looking at some other cyclic, conjugated systems. Many fused six-membered rings (for example: naphthalene and anthracene) are aromatic. However, cyclobutadiene and cyclooctatetraene are not aromatic and have quite different properties.
By investigating many cyclic conjugated systems, it has been possible to identify the factors that lead to aromaticity.
These are known as Huckel’sRule: Aromatic compounds are planar, cyclic, conjugated, and have \(4 n + 2 \(\pi\) electrons in the \(\pi\) electron cloud. Benzene is the archetypal aromatic compound: it is planar (all carbons \(\mathrm{sp}^{2}\) hybridized), cyclic (obviously) conjugated (apparent alternating single and double bonds), and it has \(6 \pi\) electrons (\(n=1\)). If we look at the bonding within benzene, we see that overlapping \(\mathrm{p}\) orbitals form a ring of pi electron density above and below the sigma \(C-C\) framework. More generally, aromatic compounds must be planar, cyclic, and conjugated so that the p orbitals overlap to form this continuous ring of electron density. So why do aromatic compounds have 4n+2 π electrons? The answer involves molecular orbital theory.
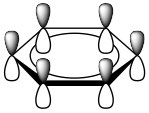


As previously noted, a consideration of the number and type of atomic orbitals that contribute to the bonding system enables \(\mathrm{MO}\) theory to predict the molecular orbitals that span the whole molecule. In benzene, we have six \(\mathrm{p}\) atomic orbitals and, therefore, expect that they combine to give six molecular orbitals as shown here. Note that there are three bonding and three antibonding \(\mathrm{MO}\)s and, since there are only six electrons in the system, we get a total of three bonds.
This type of analysis can be done for any cyclic conjugated system. While it is too complex here to go into the mathematical underpinnings, there is a relatively simple way to determine the relative energies of the \(\mathrm{MO}\)s. The approach requires that you inscribe the cyclic system into a circle, with one corner of the ring at the bottom. The places where the corners meet the ring are represent the relative energies of the \(\mathrm{MO}\)s, as shown below. This arrangement is the origin of the \(4 n + 2\) rule as we will see.



So, for example, if we consider cyclobutadiene (which has \(4 n \pi\) electrons), we see that there are 4 \(\mathrm{MO}\)s and 4 electrons. However, two of those electrons are in non-stabilized orbitals AND are uncoupled because the orbitals are of the same energy. Remember Hund’s rule: electrons occupy orbitals singly until they have to start doubling up.
This means that if cyclobutadiene were aromatic, it would be highly unstable because it would contain two unpaired electrons—it would exist as a diradical. The presence of unpaired electrons generally makes compounds very reactive. In fact, cyclobutadiene is not aromatic and instead exists as two isolated double bonds by lengthening the single bonds. Even so, it is still unstable and does not exist above \(-78^{\circ}\mathrm{C}\).
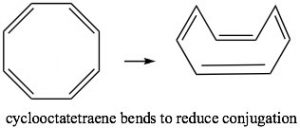
Cyclooctatetraene also has \(4 n \pi\) electrons (where \(n=2\)), and again we see the problem is that there are a pair of degenerate (same energy) \(\mathrm{MO}\)s, where the last two π electrons are located. Again, this is highly destabilizing and, to avoid this electron configuration, cyclooctatetraene actually bends so the double bonds are not conjugated with each other. The origin of the \(n+2\) rule, then, has to do with the arrangement of \(\mathrm{MO}\)s. Any cyclic conjugated compound with \(4 n \pi\) electrons will not be able to take up a stable conjugated arrangement because it will involve the highly unstable diradical.

