3.14: The Carboxylic Acid Derivatives
- Page ID
- 107509
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)learning objectives
- name acid halides, anhydrides, esters, amides, nitriles, and dicarboxylic acids using IUPAC (systematic) and selected common name nomenclature
- draw the structure of acid halides, anhydrides, esters, amides, and nitriles from IUPAC (systematic) and selected common names
Note: Nomenclature of thioesters and phosphoesters is also discussed. Ask the professor if this information is required for your course.
Introduction
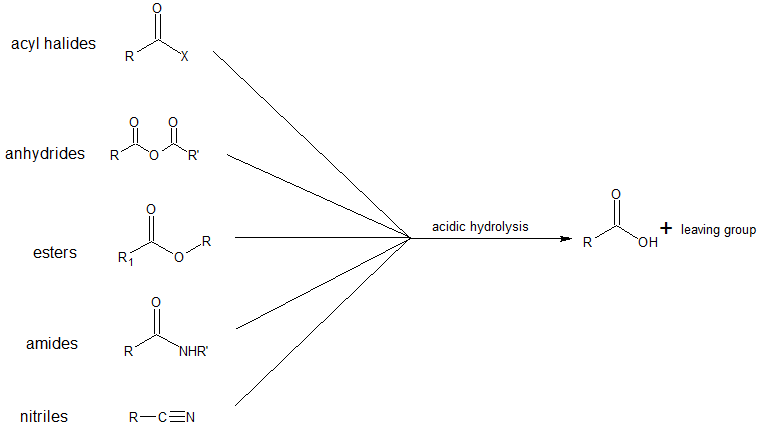
The important classes of organic compounds known as alcohols, phenols, ethers, amines and halides consist of alkyl and/or aryl groups bonded to hydroxyl, alkoxyl, amino and halo substituents respectively. If these same functional groups are attached to an acyl group (RCO–) their properties are substantially changed, and they are designated as carboxylic acid derivatives. Carboxylic acids have a hydroxyl group bonded to an acyl group, and their functional derivatives are prepared by replacement of the hydroxyl group with substituents, such as halo, alkoxyl, amino and acyloxy. The carboxylic acid derivatives can all be hydrolyzed to carboxylic acids. The specific reaction conditions are discussed in the corresponding chapter later in this text, however, the shared pattern of chemical reactivity is summarized in the diagram below.
IUPAC Nomenclature - One Pattern, so Many Variations
Apply the IUPAC nomenclature format to carboxylic acid derivatives as summarized below using the suffix or substituent names listed in the table. Some students find esters challenging to name. Carboxylates can be described as independent ions, but require a contain to form compounds. It can be helpful to think of esters as "alkylated carboxylates": identify and name the carboxylate, this name is preceded by the alkyl group branch on the carboxyl oxygen.
| Functional Group | Structure | Suffix Name | Substituent Name |
| carboxylic acid | 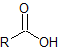 |
-oic acid | carboxy- |
| carboxylate |  |
-oate | see above |
| ester | 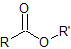 |
-oate | alkoxycarbonyl- |
| dicarboxylic acid | 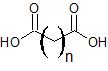 |
-dioic acid | not applicable |
| acyl halide | 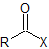 |
-oyl halide | not applicable |
|
anhydride |
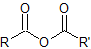 |
-anhydride |
not applicable |
| amide | 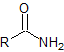 |
-amide | amido- |
| nitrile | 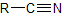 |
-nitrile | cyano- |
Common names
Most common names were derived from older systems of nomenclature that some may argue were "not systematic at all". However, it is helpful to note that the older systems of nomenclature were often based on shared structural features and/or chemical reactivity. Understanding the older nomenclature systems can offer insights into chemical reactivity and structural patterns. There are some common names that are so prevalent, they need to be memorized. Common names frequently exist when the group bonded to the carbonyl carbon is a methyl group (indicated with "acet" or "acetyl" in the common name) or a hydrogen atom (indicated with "formyl" or "form"). For example CH3C≡N is ethanenitrile (or acetonitrile) and HCONH2 is methanamide (or formamide).
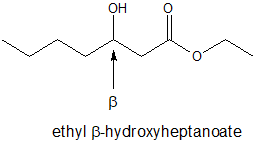
The common names for compounds with carbonyl groups often use Greek letters to specify the carbon position relative to the carbonyl carbon.
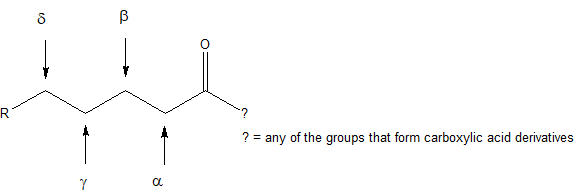
Example
In this text, we will learn about the patterns of reactivity for compounds with for beta-hydroxy carbonyl structures like the one shown below.
Nomenclature of acid halides
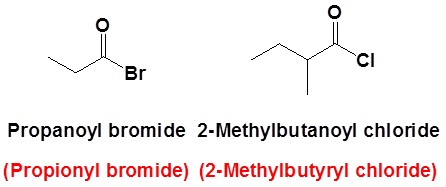
The nomenclature of acid halides starts with the name of the corresponding carboxylic acid. The –ic acid ending is removed and replaced with the ending -yl followed by the name of the halogen with an –ide ending. This is true for both common and IUPAC nomenclature. The carbonyl carbon is given the #1 location number. It is not necessary to include the location number in the name because it is assumed that the functional group will be on the end of the parent chain.
Example
Nomenclature of Anhydrides
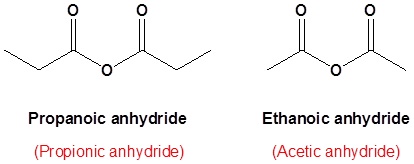
The acid anhydride functional group results when two carboxylic acids combine and lose water (anhydride = without water). Symmetrical acid anhydrides are named like carboxylic acids except the ending -acid is replaced with -anhydride. This is true for both the IUPAC and Common nomenclature.
Symmetrical anhydrides
A symmetrical anhydride is a carboxylic acid anhydride that has the following general structural formula.
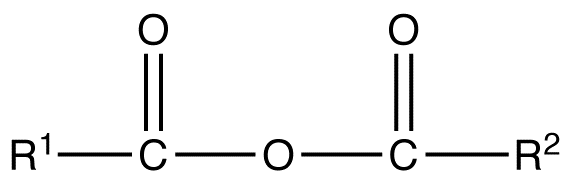
where R1=R2= hydrogen atoms, alkyl groups, aryl groups
Unsymmetrical Anhydrides
A mixed or unsymmetrical anhydride is a carboxylic acid anhydride that has the following general structural formula.
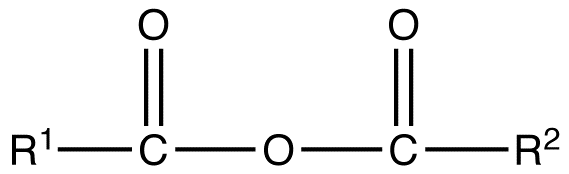
where R1≠R2, but are hydrogen atoms, alkyl groups, aryl groups. When naming unsymmetrical acid anhydrides, name both using alkanoic general method and then put the two names alphabetically. Hence, first name each component and alphabetically arranged them followed by spaces and then the word anhydride.
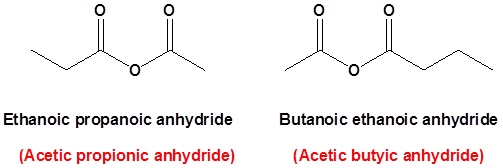
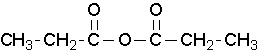
propanoic anhydride
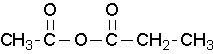
ethanoic propanoic anhydride
Exercises
1. Draw the bond-line structure for benzoic anhydride.
Solution:
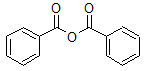
2. What is the common name for the compound below?

Solution: acetic benzoic anhydride
Common anhydride names to know
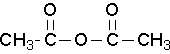
acetic anhydride (Try to name this anhydride by the proper name. J )
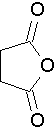
succinic anhydride (Try to name this anhydride by the proper name. J )
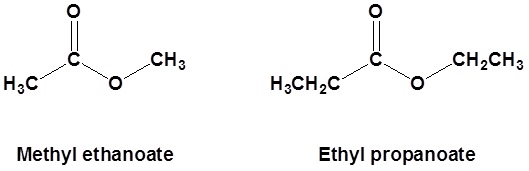
Nomenclature of Esters
Esters are made from a carboxylic acid and an alcohol.
Esters are named as if the alkyl chain from the alcohol is a substituent. No number is assigned to this alkyl chain. This is followed by the name of the parent chain form the carboxylic acid part of the ester with an –e remove and replaced with the ending –oate.
Example