2.4: Hofmann’s Rule and Zaitsev’s Rule
- Page ID
- 21831
In reactions like Hofmann’s Exhaustive Methylation – Elimination reactions, the least substituted olefin is generally formed as a major product. This is called the Hofmann’s Rule. All such reactions bear charged leaving groups like –NR3+ or –SR2+ and involve strong bases. The Zaitsev’s Rule (or Saytzeff rule) draws our attention to the alternate possibility. On elimination of HX, the more stable olefin is obtained (Fig 2.3.1). The apparent contradiction in this set of rules is easily resolved through a critical look at the mechanisms involved in these two sets of reaction conditions.
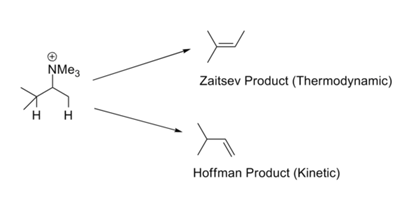
There could be two reasons for such preferences. Ingold (1960) and Bunnett (1969) suggested that a positively charged leaving group increases the acidity of the β-protons. A substituent at the β-position could hyperconjugatively decrease the acidity of the β-proton. Consequently, a terminal methyl group (this has no alkyl substituent) is more acidic than the internal methine proton (bearing at least one alkyl substituent). When the leaving group is a halogen, the mechanism shifts towards E1. Under these conditions, the stability of the developing double bond becomes important and this leads to the thermodynamically more stable product. The school of H.C Brown had suggested (1956) that steric factors govern such elimination reactions. The charged leaving groups are large compared to neutral leaving groups.
The larger leaving groups like –NR3+ and –SR2+ give more Hoffmann product than smaller groups like halogens. The bulkiness of the base also increases the Hoffmann product at the cost of the Zaitsav product. The situation appears to be more complex. When the base strength was increased without increasing the bulk at the reaction site (X-C6H4-O¯), the Hoffmann product increased at the cost of the Zaitsav product (Froemsdorf (1966,67)). This suggests an E1cB mechanism, where the acidity of the β proton is important. Thus the mechanism (and therefore the products composition) could be altered by factors such as the size of the leaving group, size of the base, nature of the leaving group and the strength of the base.


