11.1: Introduction to Distribution
- Page ID
- 317863
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Introduction to Distribution
| So far, we have described the absorption of substances into the body. Now we will focus on what happens next to substances in the body after they are absorbed. |
Figure \(\PageIndex{1}\). The processes of toxicokinetics |
Distribution Defined
Distribution is the process in which an absorbed chemical moves away from the site of absorption to other areas of the body. In this section, we will answer the following questions:
- How do chemicals move through the body?
- Does distribution vary with the route of exposure?
- Is a chemical distributed evenly to all organs or tissues?
- How fast is a chemical distributed?
- Why do some chemicals stay in the body for a long time while others are eliminated quickly?
Body Fluids
When a chemical is absorbed, it passes through cell linings of the absorbing organ (skin, lung, or gastrointestinal tract) into the interstitial fluid (fluid surrounding cells) of that organ.
The other body fluids are intracellular fluid (fluid inside cells) and blood plasma. However, the body fluids are not isolated but represent one large pool. The interstitial and intracellular fluids, in contrast to fast moving blood, remain in place with certain components (for example, water and electrolytes) moving slowly into and out of cells. A chemical, while immersed in the interstitial fluid, is not mechanically transported the way it is in blood. Table 1 lists the approximate percentage of body weight each of these body fluids comprise.

A toxicant can leave the interstitial fluid by entering:
- Local tissue cells.
- Blood capillaries and the blood circulatory system.
- The lymphatic system.
Blood Plasma
If the toxicant gains entrance into the blood plasma, it travels along with the blood, either in a bound or unbound form. Blood moves rapidly through the body via the cardiovascular circulatory system. In contrast, lymph (fluid) moves slowly through the lymphatic system. The major distribution of an absorbed chemical is by blood with only minor distribution by lymph. Since virtually all tissues have a blood supply, all organs and tissues of the body are potentially exposed to the absorbed chemical.
Distribution of a chemical to body cells and tissues requires that the toxicant penetrate a series of cell membranes. It must first penetrate the cells of the capillaries (small blood vessels) and later the cells of the target organs. The factors pertaining to passage across membranes apply to these other cell membranes as well. For example, important factors include the concentration gradient; molecular weight; lipid solubility; and polarity, with the smaller, nonpolar toxicants in high concentrations being most likely to gain entrance.
The distribution of a xenobiotic can be affected by whether it binds to plasma protein. Some toxicants may bind to these plasma proteins (especially albumin), which removes the toxicant from potential cell interaction. Within the circulating blood, the non-bound (free) portion is in equilibrium with the bound portion. However, only the free substance is available to pass through the capillary membranes. Those substances that are extensively bound are limited in terms of equilibrium and distribution throughout the body. Protein binding in the plasma greatly affects distribution, prolongs the half-life within the body, and affects the dose threshold for toxicity.
The plasma level of a xenobiotic is important since it generally reflects the concentration of the toxicant at the site of action. The passive diffusion of the toxicant into or out of these body fluids will be determined mainly by the toxicant's concentration gradient.
Volume of Distribution (written as V subscript D - see example in formula below)
The apparent volume of distribution (VD) is the total volume of body fluids in which a toxicant is distributed. The VD is expressed in liters.
If a toxicant is distributed only in the plasma fluid, the plasma concentration will remain high and a low VD results; however, if a toxicant is distributed in all sites (blood plasma, interstitial, and intracellular fluids) there is greater dilution in plasma concentration and a higher VD will result. Binding in effect reduces the concentration of free toxicants in the plasma or VD. Toxicants that undergo rapid storage, biotransformation, or elimination further affect the VD. Toxicologists determine the VD of a toxicant in order to know how extensively a toxicant is distributed in the body fluids. The volume of distribution can be calculated by the formula:
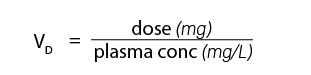
The volume of distribution may provide useful estimates as to how extensive the toxicant is distributed in the body. For example, a very high apparent VD may indicate that the toxicant has distributed to a particular tissue or storage area such as adipose tissue. In addition, the body burden for a toxicant can be estimated from knowledge of the VD by using the formula:
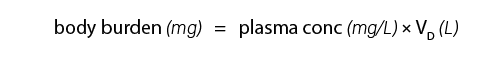
Once a chemical is in the blood stream:
- It may be excreted.
- It may be stored.
- It may be biotransformed into different chemicals (metabolites).
- Its metabolites may be excreted or stored.
- The chemical or its metabolites may interact or bind with cellular components.
Most chemicals undergo some biotransformation. The degree with which various chemicals are biotransformed and the degree with which the parent chemical and its metabolites are stored or excreted vary with the nature of the exposure (dose level, frequency, and route of exposure).
1) When an ingested toxicant is absorbed, it passes through the cells lining the GI tract into the:
a) Intracellular fluid
b) Gastric fluid
c) Interstitial fluid
- Answer
-
Interstitial fluid - This is the correct answer.
When a chemical is absorbed it passes through cell linings of the absorbing organ (in this case, the gastrointestinal tract) into the interstitial fluid (fluid surrounding cells) within that organ.
2) The apparent volume of distribution (VD) represents the:
a) Total volume of body fluids in which a toxicant is distributed
b) Amount of blood plasma in which a toxicant is dissolved
c) Amount of interstitial fluid that contains a toxicant
- Answer
-
Total volume of body fluids in which a toxicant is distributed - This is the correct answer.
The apparent volume of distribution (VD) represents the total volume of body fluids in which a toxicant is distributed. It consists of the interstitial fluid, intracellular fluid, and the blood plasma. Soon after absorption, a toxicant may be distributed to all three types of fluids, although the concentrations may be quite different. Rarely will a toxicant be distributed to only one type of fluid.