Stereochemistry of Amino Acids
- Page ID
- 2262
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)With the exception of glycine, all the 19 other common amino acids have a uniquely different functional group on the central tetrahedral alpha carbon (i.e. \(C_{\alpha}\)). The \(C_{\alpha}\) is termed "chiral" to indicate there are four different constituents and that the Ca is asymmetric. Since the \(C_{\alpha}\) is asymmetric there exists two possible, non-superimposable, mirror images of the amino acids:
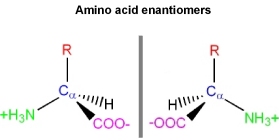
How are these two uniquely different structures in the figure ago distinguished?
- Answer
-
Based off the stereochemistyr at the \(C_{\alpha}\).
The D, L system
Glyceraldehyde contains a chiral carbon, and therefore, there are two enantiomers of this molecule. One is labeled the "L" form, and the other the "D" form. This is the frame of reference used to describe amino acid enantiomers as being either the "L" or "D" form
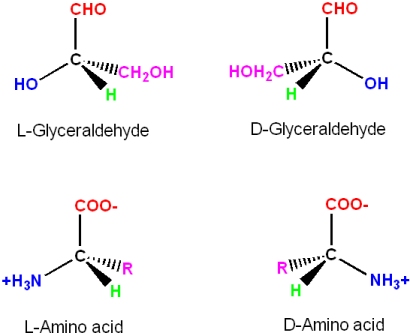
Even though the two enantiomers would seem to be essentially equivalent to each other, all common amino acids are found in the "L" enantiomer in living systems. When looking down the H-C, a bond towards the \(C_{\alpha}\) there is a mnemonic to identify the L-enantiomer of amino acids (note: in this view the three functional groups are pointing away from you, and not towards you; the H atom is omitted for clarity - but it would be in front of the C)
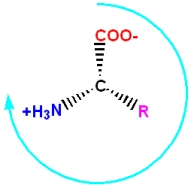
Starting with the carbonyl functional group, and going clockwise around the \(C_{\alpha}\) of the L-enantiomer, the three functional groups spell out the word CORN. If you follow the above instructions, it will spell out CONR (a silly, meaningless word) for the D-enantiomer
Optical Activity
Enantiomeric molecules have an optical property known as optical activity - the ability to rotate the plane of plane polarized light. Clockwise rotation is known as "dextrorotatory" behavior and counterclockwise rotation is known as "levorotatory" behavior.

All common amino acids are the L-enantiomer (i.e. their \(C_{\alpha}\) chiral center is the L-enantiomer), based on the structural comparison with L-glyceraldehyde. However, not all L-amino acids are Levorotatory, some are actually Dextrorotatory with regard to their optical activity. To (attempt) to avoid confusion, the optical activities are given as (+) for dextrorotatory, and (-) for levorotatory
- L(+)-alanine (this is the L-enantiomer and it is dextrorotatory)
- L(-)-serine (this is the L-enantiomer and it is levorotatory)
Multiple chiral centers
- Molecules with N chiral centers can exist in 2N isomeric structures
- Isomers that differ in configuration at only one chiral center are called diastereomers
The R,S system of naming chiral centers
A relative ranking of the "priority" of various functional groups is given as:
\[\ce{SH > OH > NH2 > COOH > CHO > CH2OH > CH3 > H}\]
- A chiral center has four different functional groups. Identify the functional group with the lowest priority
- View the chiral center down the bond from the chiral center to the lowest priority atom
- don't confuse this with the CORN mnemonic method of identifying the L-amino acid chirality by viewing from the H to the Ca )
- Assign priorities to the three other functional groups connected to the chiral center, using the above ranking
- If the priorities of these other groups goes in a clockwise rotation, the chirality is "R". If the priorities of these other groups goes counterclockwise, the chirality is "S". (Note that this assignment has nothing to do with optical activity, and is not using L-glyceraldehyde as a reference molecule)

Spectroscopic properties of amino acids
This refers to the ability of amino acids to absorb or emit electromagnetic energy at different wavelengths (i.e. energies)
- No amino acids absorb light in the visible spectrum (i.e. they are "colorless").
- If proteins have color (e.g. hemoglobin is red) it is because they contain a bound, non-protein atom, ion or molecule; iron in this case)
- All amino acids absorb in the infrared region (longer wavelengths, weaker energy than visible light)
- Some amino acids absorb in the ultraviolet spectrum (shorter wavelengths, higher energy than visible light)
- Absorption occurs as electrons rise to higher energy states
- Electrons in aromatic ring structures absorb in the u.v. spectrum. Such structures comprise the side chains of
- tryptophan, tyrosine and phenylalanine.
Separation and analysis of amino acid mixtures
The 20 common amino acids differ from one another in several important ways. Here are just two:
- Mass. The smallest amino acid (glycine) has a mass of 57 Da (i.e. g/mol), and the largest (tryptophan) has a mass of 186 Da
- Isoelectric point (pH at which the amino acid has a neutral charge). This is a function of all ionizable groups on the amino acid, including the amino and carboxyl functional groups in addition to any ionizable group on the side chain.
|
Amino Acid |
Mass (Da) |
Isoelectric Point |
Amino Acid |
Mass (Da) |
Isoelectric Point |
|---|---|---|---|---|---|
|
Aspartic Acid |
114.11 |
2.98 |
Isoleucine |
113.16 |
6.038 |
|
Glutamic Acid |
129.12 |
3.08 |
Glycine |
57.05 |
6.064 |
|
Cysteine |
103.15 |
5.02 |
Alanine |
71.09 |
6.107 |
|
Tyrosine |
163.18 |
5.63 |
Proline |
97.12 |
6.3 |
|
Serine |
87.08 |
5.68 |
Histidine |
137.14 |
7.64 |
|
Methionine |
131.19 |
5.74 |
Lysine |
128.17 |
9.47 |
|
Tryptophan |
186.12 |
5.88 |
Arginine |
156.19 |
10.76 |
|
Phenylalanine |
147.18 |
5.91 |
Threonine |
101.11 |
- |
|
Valine |
99.14 |
6.002 |
Asparagine |
115.09 |
- |
|
Leucine |
113.16 |
6.036 |
Glutamine |
128.14 |
- |
We can use these differences in physical properties to fractionate complex mixtures of amino acids into individual amino acids
- In looking at the isoelectric point of the different amino acids it seems that they will have different partial charges at a given pH.
- For example, at pH 6.0 some will be negatively charged, and some positively charged.
- For those that are negatively charged, some will be slightly negative, and others strongly negative. Similarly, for those that are positively charged, some will be slightly positive, and others strongly positive
- The charge differences of the amino acids means that they will have different affinities for other cationic or anionic charges
References
- The Merck Index, Merck & Co. Inc., Nahway, N.J., 11(1989); CRC Handbook of Chem.& Phys., Cleveland, Ohio, 58(1977)

