Penicillin
- Page ID
- 421
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)The penicillins were the first antibiotics discovered as natural products from the mold Penicillium.
Introduction
In 1928, Sir Alexander Fleming, professor of bacteriology at St. Mary's Hospital in London, was culturing Staphylococcus aureus. He noticed zones of inhibition where mold spores were growing. He named the mold Penicillium rubrum. It was determined that a secretion of the mold was effective against Gram-positive bacteria.
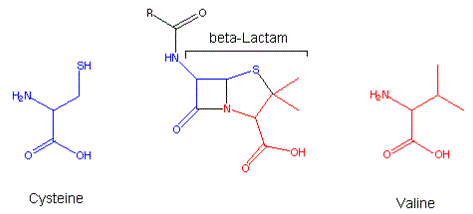
Figure 1: Beta Lactam Structure
Penicillins as well as cephalosporins are called beta-lactam antibiotics and are characterized by three fundamental structural requirements: the fused beta-lactam structure (shown in the blue and red rings, a free carboxyl acid group (shown in red bottom right), and one or more substituted amino acid side chains (shown in black). The lactam structure can also be viewed as the covalent bonding of pieces of two amino acids - cysteine (blue) and valine (red).
Penicillin-G where R = an ethyl pheny group, is the most potent of all penicillin derivatives. It has several shortcomings and is effective only against gram-positive bacteria. It may be broken down in the stomach by gastric acids and is poorly and irregularly absorbed into the blood stream. In addition many disease producing staphylococci are able to produce an enzyme capable of inactivating penicillin-G. Various semisynthetic derivatives have been produced which overcome these shortcomings.
Powerful electron-attracting groups attached to the amino acid side chain such as in phenethicillin prevent acid attack. A bulky group attached to the amino acid side chain provides steric hindrance which interferes with the enzyme attachment which would deactivate the pencillins i.e. methicillin. Refer to Table 2 for the structures. Finally if the polar character is increased as in ampicillin or carbenicillin, there is a greater activity against Gram-negative bacteria.
Penicillin Mode of Action
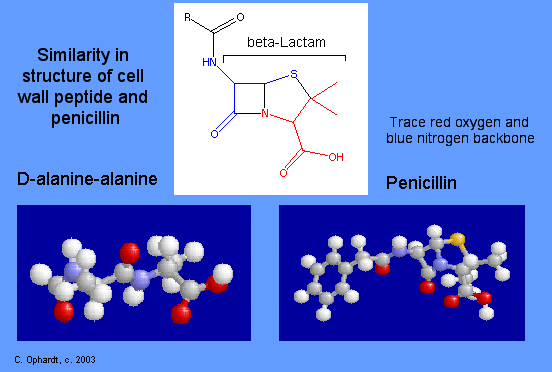
All penicillin derivatives produce their bacteriocidal effects by inhibition of bacterial cell wall synthesis. Specifically, the cross linking of peptides on the mucosaccharide chains is prevented. If cell walls are improperly made cell walls allow water to flow into the cell causing it to burst. Resemblances between a segment of penicillin structure and the backbone of a peptide chain have been used to explain the mechanism of action of beta-lactam antibiotics. The structures of a beta-lactam antibiotic and a peptide are shown on the left for comparison. Follow the trace of the red oxygens and blue nitrogen atoms.
Gram-positive bacteria possess a thick cell wall composed of a cellulose-like structural sugar polymer covalently bound to short peptide units in layers.The polysaccharide portion of the peptidoglycan structure is made of repeating units of N-acetylglucosamine linked b-1,4 to N-acetylmuramic acid (NAG-NAM). The peptide varies, but begins with L-Ala and ends with D-Ala. In the middle is a dibasic amino acid, diaminopimelate (DAP). DAP (orange) provides a linkage to the D-Ala (gray) residue on an adjacent peptide.
The bacterial cell wall synthesis is completed when a cross link between two peptide chains attached to polysaccharide backbones is formed. The cross linking is catalyzed by the enzyme transpeptidase. First the terminal alanine from each peptide is hydrolyzed and secondly one alanine is joined to lysine through an amide bond.
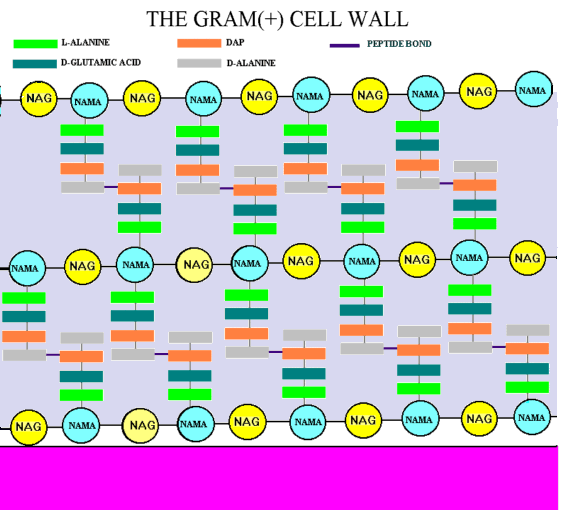
Peptidoglycan image courtesy of the University of Texas-Houston Medical School
Penicillin binds at the active site of the transpeptidase enzyme that cross-links the peptidoglycan strands. It does this by mimicking the D-alanyl-D-alanine residues that would normally bind to this site. Penicillin irreversibly inhibits the enzyme transpeptidase by reacting with a serine residue in the transpeptidase. This reaction is irreversible and so the growth of the bacterial cell wall is inhibited. Since mammal cells do not have the same type of cell walls, penicillin specifically inhibits only bacterial cell wall synthesis.
Bacterial Resistance
As early as the 1940s, bacteria began to combat the effectiveness of penicillin. Penicillinases (or beta-lactamases) are enzymes produced by structurally susceptable bacteria which renders penicillin useless by hydrolysing the peptide bond in the beta-lactam ring of the nucleus. Penicillinase is a response of bacterial adaptation to its adverse environment, namely the presence of a substance which inhibits its growth. Many other antibiotics are also rendered ineffective because of this same type of resistance.
Severe Allergic Shock
It is estimated that between 300-500 people die each year from penicillin-induced anaphylaxis, a severe allergic shock reaction to penicillin. In afflicted individuals, the beta-lactam ring binds to serum proteins, initiating an IgE-mediated inflammatory response. Penicillin and ala-ala peptide - Chime in new window
Cephalosporins
Cephalosporins are the second major group of beta-lactam antibiotics. They differ from penicillins by having the beta-lactam ring as a 6 member ring. The other difference, which is more significant from a medicinal chemistry stand point, is the existence of a functional group (R) at position 3 of the fused ring system. This now allows for molecular variations to effect changes in properties by diversifying the groups at position 3.
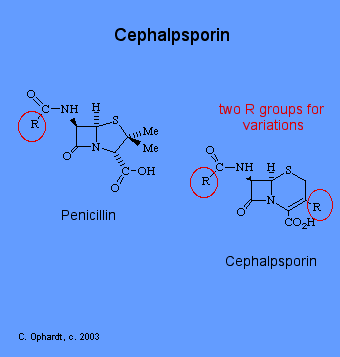
The first member of the newer series of beta-lactams was isolated in 1956 from extracts of Cephalosporium acremonium, a sewer fungus. Like penicillin, cephalosporins are valuable because of their low toxicity and their broad spectrum of action against various diseases. In this way, cephalosporin is very similar to penicillin. Cephalosporins are one of the most widely used antibiotics, and economically speaking, has about 29% of the antibiotic market. The cephalosporins are possibly the single most important group of antibiotics today and are equal in importance to penicillin.
The structure and mode of action of the cephalosporins are similar to that of penicillin. They affect bacterial growth by inhibiting cell wall synthesis, in Gram-positive and negative bacteria. Some brand names include: cefachlor, cefadroxil, cefoxitin, ceftriaxone. Cephalexin

