Cholinergic Drugs I - Nicotinic and Muscarinic Receptors
- Page ID
- 89060
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Neurotransmitters released from nerve terminals bind to specific receptors, which are specialized macromolecules embedded in the cell membrane. The binding action initiates a series of specific biochemical reactions in the target cell that produce a physiological response. These effects can be modified by various drugs that act as agonists or antagonists.
The autonomic system consists of two major divisions: the Sympathetic Nervous System and the Parasympathetic Nervous System. These often function in antagonistic ways. A signal is transmitted from the spinal cord to peripheral areas through two successive neurons. The first neuron (preganglionic), which originates in the spinal cord, will synapse with the second neuron (postganglionic) in a ganglion. Parasympathetic ganglia tend to lie close to or within the organs or tissues that their neurons innervate, whereas sympathetic ganglia lie at a more distant site from their target organs. Both systems have associated sensory fibers that send feedback information into the central nervous system regarding the functional condition of target tissues.
The significant difference between the two systems is that their postganglionic fibers secrete different neurotransmitters. Those of the parasympathetic system secrete acetylcholine (ACh), hence the name cholinergic, whereas the postganglionic fibers secrete norepinephrine (NE), hence the name adrenergic. The preganglionic fibers of both systems secrete ACh; therefore, both preganglionic fibers are cholinergic. Motor neurons which are not part of the autonomic nervous system also release acetylcholine (see Figure 1).
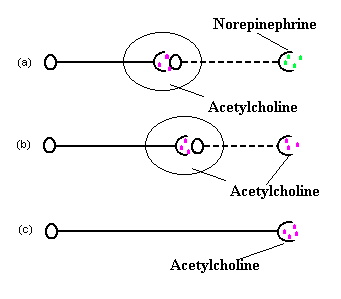
Figure 1. (a) Preganglionic neurons (solid line) of the sympathitic division of the autonomic nervous system release acetycholine at their synapses with postganglionic neurons (dashed line). Although exceptions occur, the postganglionic neurons release mainly norepinephrine at their function with effectors. (b) Pregangionic neurons (solid line) of the parasympathetic division of the autonomic nervous system release acetycholine at their synapses with postganglionic neurons (dashed line), and the postgangionic neurons also release acetycholine at their effectors. (c) Somatic efferent neurons release acetylcholine at their junctions with skeletal muscles.S.K.Anderson
Acetylcholine acts on more than one type of receptor. Henry Dale, a British physiologist working in London in 1914, found that two foreign substances, nicotine and muscarine, could each mimic some, but not all, of the parasympathetic effects of acetylcholine. It was found that Nicotine stimulates receptors on skeletal muscle and sympathetic and parasympathetic postganglionic neurons, however, muscarine stimulates receptor sites located only at the junction between postganglionic parasympathetic neurons and the target organ. Dale therefore classified the many actions of acetylcholine into nicotinic effects and muscarinic effects. It has subsequently become clear that there are two distinct types of acetylcholine receptors affected by either muscarine or nicotine. To restate this again, nicotinic receptors cause sympathetic postganglionic neurons and parasympathetic postganglionic neurons to fire and release their chemicals and skeletal muscle to contract. Muscarinic receptors are associated mainly with parasympathetic functions and stimulates receptors located in peripheral tissues (e.g., glands, smooth muscle). Acetylcholine activates all of these sites.
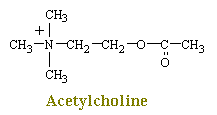
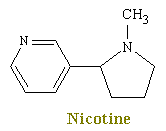
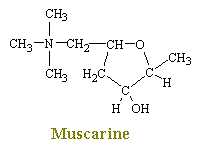
Advanced biochemical techniques have now shown a more fundamental difference in the two types of cholinergic receptors. The nicotinic receptor is a channel protein that, upon binding by acetylcholine, opens to allow diffusion of cations. The muscarinic receptor, on the other hand, is a membrane protein; upon stimulation by neurotransmitter, it causes the opening of ion channels indirectly, through a second messenger. For this reason, the action of a muscarinic synapse is relatively slow. Muscarinic receptors predominate at higher levels of the central nervous system, while nicotinic receptors, which are much faster acting, are more prevalent at neurons of the spinal cord and at neuromuscular junctions in skeletal muscle.
A cholinergic drug is any of various drugs that inhibit, enhance, or mimic the action of the neurotransmitter acetylcholine within the body. Acetylcholine stimulation of the parasympathetic nervous system helps contract smooth muscles, dilate blood vessels, increase secretions, and slow the heart rate. Some cholinergic drugs, such as muscarine, pilocarpine, and arecoline, mimic the activity of acetylcholine in stimulating the parasympathetic nervous system. These drugs, however, have few therapeutic uses. Other cholinergic drugs, such as atropine and scopolamine, inhibit the action of acetylcholine and thus suppress all the actions of the parasympathetic nervous system. These drugs help dry up such bodily secretions as saliva and mucus and relax smooth-muscle walls. They are used therapeutically to relieve spasms of the smooth-muscle walls of the intestines, to relieve bronchial spasms, to diminish salivation and bronchial secretions during anesthesia, and to dilate the pupil during ophthalmological procedures.
Nicotine
Nicotine is an organic compound that is the principal alkaloid of tobacco. Nicotine occurs throughout the tobacco plant and especially in the leaves. The compound constitutes about 5 percent of the plant by weight. Both the tobacco plant (Nicotiana tabacum) and the compound are named for Jean Nicot, a French ambassador to Portugal, who sent tobacco seeds to Paris in 1550.
Crude nicotine was known by 1571, and the compound was obtained in purified form in 1828; the correct molecular formula was established in 1843, and the first laboratory synthesis was reported in 1904. Nicotine is one of the few liquid alkaloids. In its pure state it is a colorless, volatile base (pKa -8.5) with an oily consistency, but when exposed to light or air, it acquires a brown color and gives off a strong odor of tobacco.

The complex and often unpredictable changes that occur in the body after administration of nicotine are due not only to its actions on a variety of neuroeffector and chemosensitive sites but also to the fact that the alkaloid has both stimulant and depressant phases of action. The ultimate response of any one system represents the summation of the several different and opposing effects of nicotine. For example, the drug can increase the heart rate by excitation of sympathetic cardiac ganglia, and it can slow down the heart rate by stimulation of parasympathetic cardiac ganglia. In addition, the effects of the drug on the chemoreceptors of the carotid and aortic bodies and on medullary centers influence heart rate, as do also the cardiovascular compensatory reflexes resulting from changes in blood pressure caused by nicotine. Finally, nicotine causes a discharge of epinephrine from the adrenal medulla, and this hormone accelerates cardiac rate and raises blood pressure.
Nicotine is unique in its biphasic effects. In the medulla, small doses of nicotine evoke the discharge of catacholamines, and in larger doses prevent their release in response to splanic nerve stimulation. Its biphasic effect causes a stimulant effect when inhaled in short puffs, but when smoked in deep drags it can have a tranquilizing effect. This is why smoking can feel invigorating at some times and can seem to block stressful stimuli at others.
Nicotine markedly stimulates the central nervous system (CNS). Appropriate doses produce tremors in both man and laboratory animals; with somewhat larger dose, the tremor is followed by convulsions. The excitation of respiration is a prominent action of nicotine; although large doses act directly on the medulla oblongata, smaller doses augment respiration reflexly by excitation of the chemoreceptors of the carotid and aortic bodies. Stimulation of the CNA is followed by depression, and death usually results from failure of respiration due to both central analysis and peripheral blockade of muscles of respiration. Nicotine also causes vomiting by central and peripheral actions. The central component of the vomiting response is due to stimulation of the chemoreceptor trigger zone is in the medulla.oblongata. In addition, nicotine activates vagal and spinal afferent nerves that from the sensory input of the reflex pathways involved in the act of vomiting.
Although acetylcholine causes vasodilation and a decrease in heart rate, when administered intravenously to the dog, nicotine characteristically produces an increase in heart rate and blood pressure. This is because in general, the cardiovascular responses to nicotine are due to stimulation of the sympathetic ganglia and the adrenal medulla, together with the discharge of catacholamines from sympathetic nerve endings.
Nicotine is commercially obtained from tobacco scraps and is used as an insecticide and as a veterinary vermifuge (wormer). Nitric acid or other oxidizing agents convert nicotine to nicotinic acid, or niacin, which is used as a food supplement.
Medicinal Uses
| General Uses |
Tabacco
| Tabacco Info | |
| Tabacco Death | |
| Cigarettes - Tar, Nicotine, and Carbon Monoxide Content |
Nicotine Addiction
| Link 1 | |
| Link 2* |
Miscellaneous
| How deadly is nicotine? (don't take this site too seriously) | |
| Nicotine Dosages |
Muscarine
Muscarine, and alkaloid obtained from the poisonous mushroom Amanita Muscaria, produces the effects predictable from stimulation of postgangiolinc parasympathetic fibers. The symptoms usually occur within 15-30 minutes of ingestion or injection, and are focused on the involuntary nervous system. The muscarinic alkaloids stimulate the smooth muscle and therby increase motility; large doses cause spasm and severe diarrhea. The bronchial musculature is also stimulated, causing asmatic-like attacks. Excessive salivation, sweating, tears, lactation (in pregnant women), plus severe vomiting also occur. The most prominent cardiovascular effects are the a marked fall in the blood pressure and a slowing or temporarily cessation of the heart. Victims normally recover within 24 hours, but severe cases may result in death due to respiratory failure. All effects of muscarine-like drugs are prevented by the alkaloid atropine. Furthermore, neither atropine-like nor muscarine-like drugs show effects at the neuromuscular junction.

Although muscarine and muscarine like alkaloids are of great value as pharmacological tools, present clinical use is largely restricted. Since evidence is beginning to accumulate that there are distinct subtypes of muscarinic receptors, there has been a renewed interest in synthetic analogs that may enhance the tissue selectivity of muscarinic agonists.

