Mass Spectrometry: Performance Enhancing Drugs
- Page ID
- 287615
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Purpose: The objective of this module is to ensure that the students have a thorough understanding of the functions, components, capabilities and limitations of using mass spectrometry as a detection system. This will be developed principally through the requirements of doing steroid analysis of urine samples.
Learning Outcomes:
By the end of the assignment students will be able to:
- Identify the individual components of the MS and their respective functions.
- Describe how ions of different m/z are distinguished in a MS.
- Compare and contrast the capabilities of TOF and quadrupole MS detectors.
- Compare, and explain molecular fragmentation patterns.
- Use isotopic abundances and molecular fragmentation as a means of analyte identification.
In order to obtain any meaningful information from a separation of compounds a detector must be coupled to the separation instrument. One of the most powerful detectors is a mass spectrometer (MS). This detector converts compounds into gas phase ions, which are then sorted and quantified on the basis of their mass. As the analytes eluting from a GC separation are already in the gas phase, the MS can be coupled very easily to the GC.
The MS detector is composed of multiple components in order to effectively analyze the gas phase ions. This section will guide you through the structure and function of the various components of the MS detector. In all MS detectors, the final stage of the ion detection process is to quantify the number of ions into an electronic signal. This is typically done with an electron multiplier that is able to convert the impact of an ion with the detector into an electrical signal. However, this detector is effectively “dumb”, it can do little more than quantify the number of ions that hit it. What makes the MS detector so powerful is how it can both make gas phase ions and determine which ones are reaching the detector at a given point in time.
- The gas molecules eluting from the GC do not have a native charge. How might the MS impart a charge on those molecules? Why do the gas molecules need to be an ion in order to be detected?
- Why does the MS need a vacuum pump in order to operate properly?
- Will the carrier gas molecules from the GC result in interfering signals in the MS?
- The gas exiting the GC is directed into a vacuum chamber where it is subsequently ionized. As the gas will inherently diffuse in all directions, and the gas molecules of similar charge will repel one-another, how can the greatest number of gas phase ions be directed towards the detector at the end of the MS?
- When a collection of gas molecules enters the MS from the GC and they are ionized and propelled towards the detector, do all the ions reach the detector at the same time? Explain the reasoning behind your answer.
- What principle advantages does a MS detector provide to a GC separation as compared to a flame ionization or thermal conductivity detector?
- Using the components listed below, draw a block diagram of a GC-MS instrument. Clearly indicate which parts are connected to each other.
- Column
- Computer
- Detector
- Helium Tank
- Injection Port
- Ion Optics
- Ion Source
- Mass Selector
- Oven
- Transfer Line
- Vacuum Pump
Of the individual components of a MS, the mass selector is arguably the most important. This portion of the instrument allows for the differentiation of the various ions entering the MS, and regulates which ions reach the detector. The simplest mass selector is a time of flight (TOF) selector. This type of mass selector discriminates between ions of different mass based on the time at which they reach the detector. The following questions will explore how a TOF mass selector operates.
- Testosterone (T) 288.42 g/mol, and nandrolone (N) 274.40 g/mol, are both injected into a TOF mass selector at the same time. Both compounds are in the gas phase and ionized; each compound lost an electron resulting in two radical cations T+• and N+•. The gas phase ions are propelled towards the detector by applying a positive charge to the electrode next to the injection port, causing the ions to be repelled (fly) towards the detector. Which of the two ions, T+• or N+• will reach the detector first?
- In the example above both testosterone and nandrolone were singly charged. What would you expect to occur if testosterone lost a second electron, and thus had a +2 charge? When would it reach the detector relative to the nandrolone cation (N+•)? Would the detector assign it the same molecular weight as it did in the problem above?
- As the testosterone and nandrolone ions in the problems above have significantly different masses they should be relatively easy to differentiate in a TOF mass selector. For ions with more comparable molecular weights such as testosterone (288.4244 g/mol) and Norfludiazepam (288.704 g/mol), what factors in the design of the TOF could improve the resolution (mass discrimination) of the mass selector?
- An unknown compound is ionized and sent down a TOF mass selector to a detector what information might be ascertained from this measurement?
- With a GC connected directly to a TOF-MS instrument there will be a continuous flow of gas into the ionization system. If these ions are continuously sent towards the detector how will this impact the determination of analyte mass to charge ratios (m/z)? How might the introduction of ions from the GC be modified to improve the mass analysis by the TOF-MS?
The TOF system is one of many mass selection methods available for MS instruments. Another common instrument is the quadrupole mass analyzer (often shortened to “quad”). This mass analyzer uses four parallel cylindrical rods to differentiate between ions of different masses. By varying the electrical potential (DC), and radio frequency (RF) applied to the cylinders, ions with specific m/z values can be guided through the length of the quadrupole. Ions of different m/z than the selected value will not have a stable flight path, and will be unable to exit the orifice at the end of the quadrupole.
- Quadrupole mass analyzers can be operated in two ways: scanning and selected ion monitoring (SIM). In scanning mode, the DC and RF voltages are altered throughout the analysis to allow all m/z values to pass through the quadrupole during the scan period. For SIM analysis, the DC and RF voltages are fixed to allow only a specific m/z to pass through. What advantages and disadvantages can you see with each of these analysis approaches?
- Given the small size and ease of operation of quadrupole mass analyzers it is possible to place several of them in series. In this manner, the ions exiting one quad can enter the next. Can you envision any benefits to designing an instrument with multiple sequential quads?
The identification of a compound in a MS can occasionally be done solely on the basis of the measured molecular weight for the ion. However, with many compounds having identical molecular weights, such as disaccharides with molecular formulas of C12H22O11, molecular weight alone is not sufficient to identify a compound.
- How can the combination of a separation technique, such as LC or GC, and a MS aid in the identification of a compound?
Though the combination of a separation technique and the molecular weight information from a MS can help differentiate between molecules with identical molecular weights, it still may not be sufficient in all cases. Certainly the question can arise as to whether or not compounds were properly resolved in the separation stage of the analysis. This is particularly problematic for steroid analysis, both for health and anti-doping purposes, where differentiating testosterone, epitestosterone, dehydroepiandrosterone, and testrolone (all with a molecular formula of C19H28O2) would be crucial.
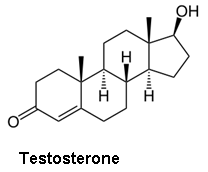
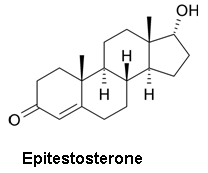
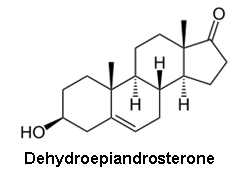
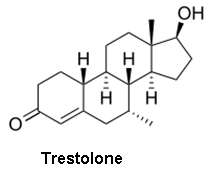
- Epitestosterone - an inactive, naturally occurring epimer of testosterone
- Dehydroepiandrosterone - an endogenous compound, active as a neurosteroid
- Trestolone - a synthetic compound with potential as a male contraceptive
Fortunately the ionization process in MS provides a solution for the differentiation of such compounds. When the molecules are ionized, not all the molecules remain intact, a percentage of the molecules will fragment, forming smaller molecular weight ions. The extent of the fragmentation depends on the structure of the analyte, as well as the amount of energy transferred through the ionization process. Electron ionization (EI), where the analyte molecules collide with high kinetic energy electrons, is considered a hard ionization technique, meaning that the formation of many fragment ions is likely. Softer ionization techniques, such as chemical ionization (CI), or electrospray ionization (ESI), do not form as many fragment ions in the ionization process.
Below is the EI mass spectrum for pure (non-derivatized) testosterone. Use it, along with the structure for testosterone to answer the following questions.
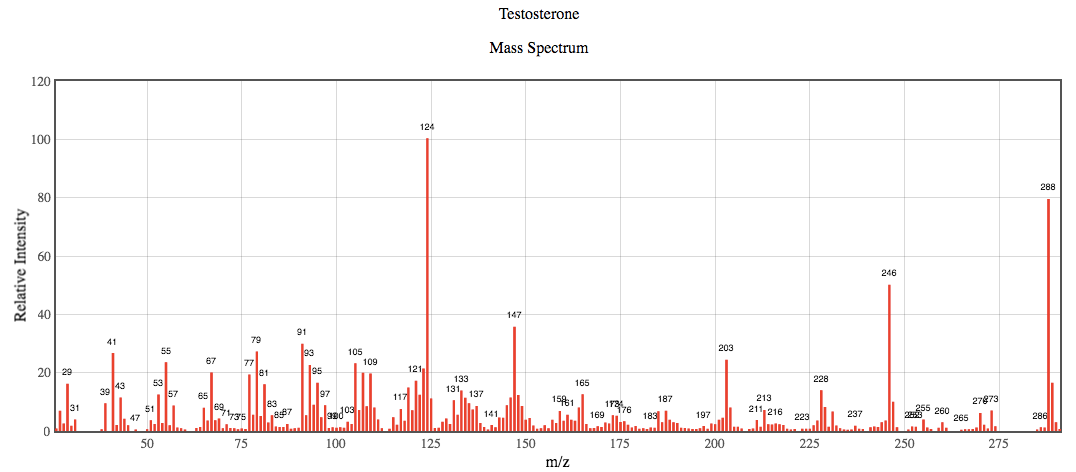
- Testosterone has a molecular weight of 288.4 g/mol, why are there peaks in the mass spectrum at values of 289 and 290 m/z?
- When a parent ion undergoes fragmentation the charge of the parent ion is only transferred to one portion of the fragments, the daughter ion. What happens to the remaining portion(s) of the parent ion?
- Why is the most abundant ion in the mass spectrum for testosterone not the 288 m/z value?
- Determine the m/z ratios for the testosterone fragments indicated by the figure below?
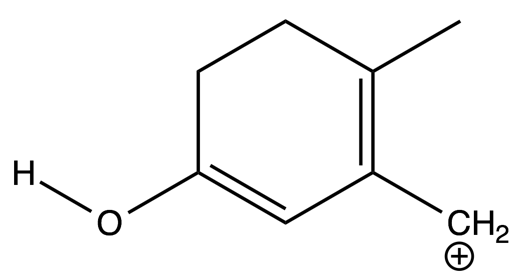
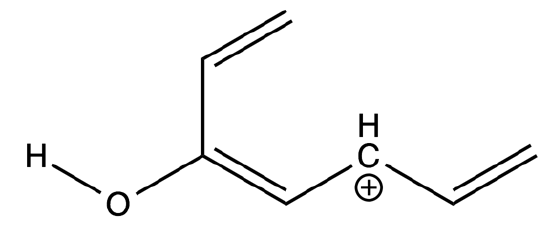
- Indicate on the structure of testosterone where the above fragment ions likely originated.
- How might the mass spectrum for testosterone differ if the molecule was subjected to a softer form of ionization, such as CI?
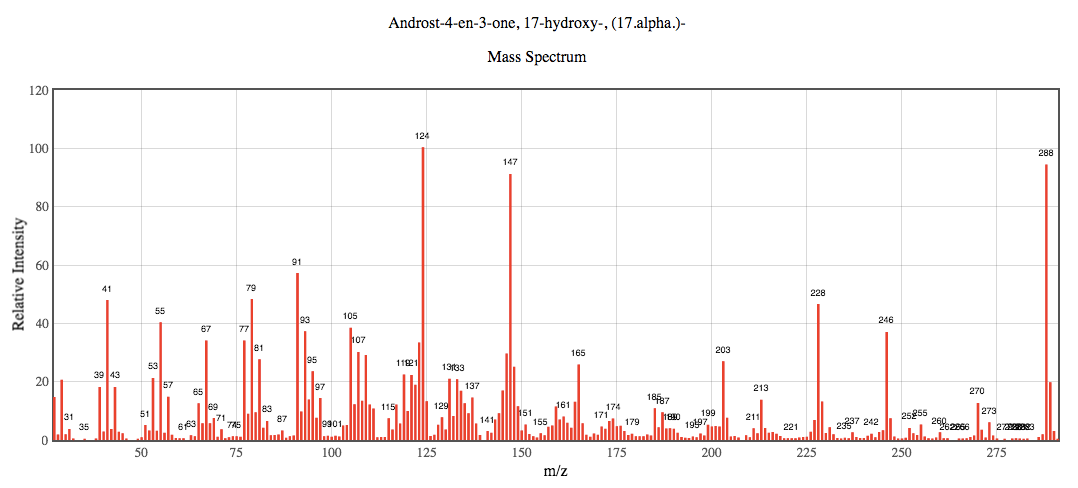
- Below is the EI mass spectrum for epitestosterone. Compare it to the mass spectrum for testosterone. How can the mass spectra for epitestosterone and testosterone be differentiated? Why might this differentiation be possible?
- The analysis of steroids in urine typically involves the derivatization of the steroids with MSTFA. How might this derivatization change the mass spectra obtained for testosterone?
Identifying the abuse of testosterone in male athletes can be very challenging, as they naturally produce testosterone. Fortunately the ratio of 13C and 12C can be used to differentiate the sources of testosterone. Due to the differences in endogenous synthesis, versus chemical synthesis of testosterone, the chemical synthesis produces a testosterone compound with a higher 13C/12C ratio.
- How would you expect the testosterone mass spectrum to change if the testosterone sample was a mixture of natural and synthetic testosterone?
In question 14 you were asked how you might combine multiple quadrupole mass analyzers in a single instrument. Due to the manner in which quadrupoles handle the ions, they have a great potential for analyzing the fragmentation patterns of ions. The most common configuration is three quadrupoles in a row, a “triple quad”. In this configuration the first and third quadrupoles are used for mass analysis, and the second (middle) quadrupole acts as a collision chamber. The collision chamber is used to induce further fragmentation of any ions that enter that quadrupole.
- Considering the two modes of operation for quadrupole mass analyzers (SIM and full scan) how might these two analysis modes be combined in a triple quad analysis? How will the information obtained from each combination differ?
- Using an instrument like a GC-MS/MS do you think it would be possible to identify a novel (previously unknown) synthetic steroid in a urine sample? How might you go about identifying such a compound from the MS/MS data?
Contributors and Attributions
- Chris Harrison, San Diego University (charrison@mail.sdsu.edu)
- Sourced from the Analytical Sciences Digital Library


