7: The Grignard Reaction (Experiment)
- Page ID
- 126802
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)In 1912, the Nobel Prize in Chemistry was awarded to Victor Grignard for his work with the reaction that is now named after him. This reaction is valuable for its ability to form carbon-carbon bonds. The Grignard reagents are formed from the reaction of an alkyl halide with magnesium metal in anhydrous ether. The carbon atom of the Grignard reagent can function as both a strong base and a strong nucleophile. However, due to the high reactivity of these Grignard reagents, great care must be taken to keep the reactions as free from air and water as possible.
Reaction Scheme

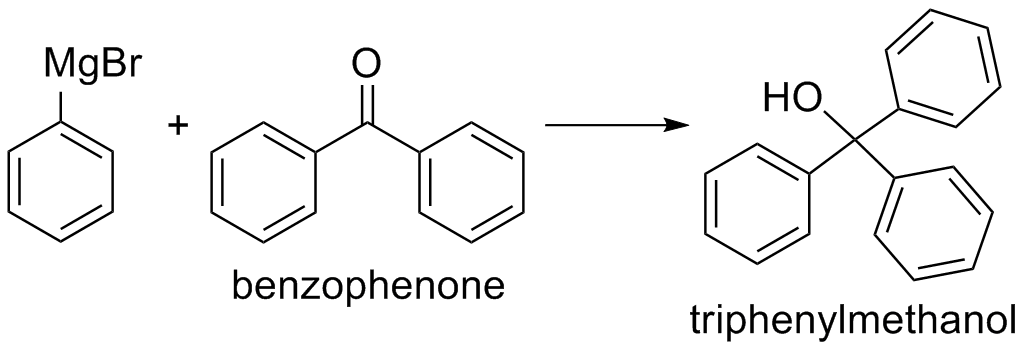
Biphenyl, which is formed from a coupling reaction between unreacted bromobenzene and Grignard reagent, is often observed as a major impurity in this reaction. Formation of the side product is favored by high concentrations of bromobenzene and increased reaction temperature. Biphenyl is yellowish in color and can be separated from the triphenylmethanol product.
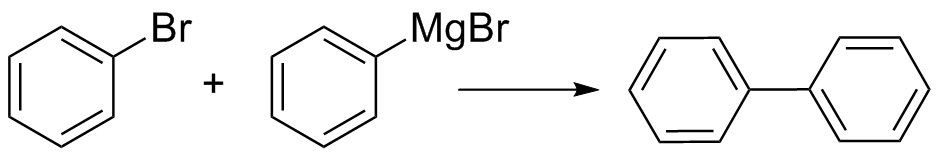
Objective
Experiment 7 is a group experiment. You will work in a small group to carry out variations of the Grignard reaction with phenyl magnesium bromide and benzophenone to form triphenylmethanol and to answer the focus questions below.
Focus Questions
- What conditions favor the formation of the biphenyl side product?
- Does using iodine as an additive improve the Grignard reaction?
Procedure
Equipment preparation
A significant amount of water will adhere to the surface of glassware. Therefore all glassware used in the Grignard reaction must be scrupulously cleaned and dried. In preparation wash all glassware, rinse with acetone, and place dry glassware in the oven overnight. Glassware to be placed in the oven includes the Claisen head adapter, 5 mL conical vial, magnetic spin vein, stir rod or spatula and 3 disposable dram vials. Note disposable dram vials need not be washed, but should be rinsed with acetone.
Just prior to your experiment carefully remove dry glassware from the oven, assemble and immediately cap it with a septum. If the rubber septum in your kit has been punctured multiple times obtain a replacement septum from your GSI. The hard side of the septum is the solvent resistant Teflon side and should be place downward. Stopper the disposable dram vials with corks. Allow all glassware to cool to room temperature before proceeding with the experiment. Keep the vessel capped throughout the course of the experiment unless you need to add something to it.
Experiment 7 is a two week experiment. At the end of the first session be sure to re-clean and replace your glassware in the oven so as to ensure that it is dry for the second session. With careful planning each group member should be able to carry out 2 complete Grignard experiments.
Preparation of Grignard reagent
Obtain 3 mL of anhydrous diethyl ether from your GSI in one of the oven dried dram vials. Weigh magnesium powder (50 mg, 2 mmol) and add it to your reaction vessel. Using a 1.0 mL syringe inserted through the septum add 0.5 mL of anhydrous diethyl ether to the reaction vessel. In a separate oven dried vial, add bromobenzene (330 mg, 2.1 mmol) and 0.7 mL of anhydrous diethyl ether and transfer 0.1 mL of this solution into the reaction tube also using the 1.0 mL syringe inserted through the septum. If the reaction doesn’t start quickly, remove the septum and crush the magnesium with a dry stirring rod. Replace the septum, making sure to release pressure as necessary using a needle to vent as necessary. The solution should turn cloudy and start to boil, indicating that the reaction has started. To prevent the ether from boiling away, wrap a damp paper towel around neck of the Claisen adapter. Over several minutes, slowly add the remaining bromobenzene/ether solution, making sure that the reaction does not boil too vigorously. It is important to control the addition of bromobenzene to avoid the rapid evolution of heat. After boiling slows, add a magnetic stir bar to the reaction tube and stir.
The reaction is complete when none of the magnesium remains. If the level of the ether has decreased, add more. To prevent decomposition of the Grignard reagent it will be used immediately in the solution that it is in; there is no isolation or purification of the phenylmagnesium bromide.
Formation of triphenylmethanol
In an oven dried vial, dissolve benzophenone (364 mg, 2 mmol) in 1 mL of anhydrous ether. With a dry 1 mL syringe, slowly add the solution to the reaction vessel containing the Grignard reagent, thus maintaining a gentle reflux. Rinse the benzophenone vial with a small amount of anhydrous ether and add this to the reaction tube also. Vent the reaction vessel with a needle as necessary to release pressure. Allow the reaction mixture to stand at room temperature and observe a color change. The reaction is complete when the red color disappears.
When the reaction is complete, cool the tube in ice and add to it drop wise (with stirring) 2 mL of 3M HCl. It may be necessary to transfer the reaction mixture to a larger container before completing the addition. As the acid is added the triphenylmethanol will separate from solution as a white precipitate. Add additional ether to re-dissolve all of the precipitate, it need not be anhydrous. Remove the aqueous layer using a pipette and add an equal volume of saturated aqueous sodium chloride solution (brine) to the diethyl ether layer. Remove the aqueous layer and dry the ether layer by adding anhydrous calcium chloride until the drying agent no longer clumps. Carefully shake or swirl the vial occasionally over 5-10 minutes. Remove the ether from the reaction tube and place it in a tared, dry reaction tube. Use additional ether to wash the drying agent and combine all of the ether extracts. Evaporate the ether in a hood by blowing nitrogen or air onto the solution. After all of the solvent has been removed, determine the weight of the crude product.
There will also be some pale yellow-colored biphenyl present from the reaction of bromobenzene and phenyl magnesium bromide. In order to remove the biphenyl, add 0.5 mL of petroleum ether to the residue, cool in an ice bath briefly while stirring and remove the solvent (process is called trituration). The remaining residue (the crude triphenylmethanol) should then be recrystallized from 2-propanol. Determine the weight, melting point, and percent yield of the triphenylmethanol. Dichloromethane is a good TLC solvent for comparing triphenylmethanol and biphenyl.
Alternate procedure focus question 2
Before starting the reaction place the magnesium powder and an iodine crystal in the oven dried reaction vessel . Gently warm the vessel on your hot plate until evidence of purple iodine vapor is observed. Allow the vessel to cool and proceed as above.
Post-lab Questions
- Why is biphenyl soluble in petroleum ether, but triphenylmethanol is not?
- What other ketones are available in the lab, that could be used in the Grignard reaction?
- What would you do differently if you could modify the design of the experiment?
- Was your hypothesis correct? What results either supported or refuted your hypothesis?


