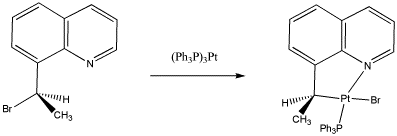OA3. Polar Oxidative Addition
- Page ID
- 4318
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
OA3. Polar Oxidative Addition
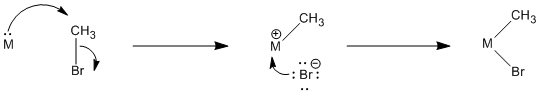
The polar oxidative addition mechanism is very similar to an aliphatic nucleophilic substitution (SN1 or SN2) reaction.
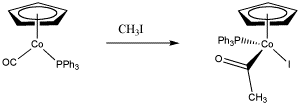
Figure OA3.1. An example of a polar oxidative addition.
In an oxidative addition, the metal can act as a nucleophile in the first step in an SN2 process. In the second step, the liberated halide binds to the metal. That doesn't happen in a normal nucleophilic substitution. In this case, the metal has donated its electrons and is able to accept another pair from the halide.
Figure OA3.2. Mechanistic steps in a polar oxidative addition.
Polar oxidative addition has some requirements similar to a regular SN1 or SN2 reaction:
- Requires good leaving group
- Requires tetrahedral carbon (or a proton) as electrophile
Problem OA3.1.
- What do you think is the most difficult step (i.e. the rate-determining step) for the reaction in Figure OA3.2? Why?
- Suggest the probable rate law for this reaction.
Problem OA3.2.
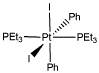
The platinum compound shown below is capable of reductively eliminating a molecule of iodobenzene.
a) Show the products of this reaction.
The starting platinum compound is completely stable in benzene; no reaction occurs in that solvent. However, reductive elimination occurs quickly when the compound is dissolved in methanol instead.
b) Explain why the solvents may play a role in how easily this compound reacts.
The reaction in methanol is inhibited by added iodide salts, such as sodium iodide.
c) Provide a mechanism for the reductive elimination of iodobenzene from the platinum complex, taking into account the solvent dependence and the inhibition by iodide ion.
Problem OA3.3.
For the following reaction,
- Identify the oxidation state at platinum in the reactant and the products.
- Assign stereochemical configuration in the product and the reactant.
- Explain the steresochemistry of the reaction.
Problem OA3.4.
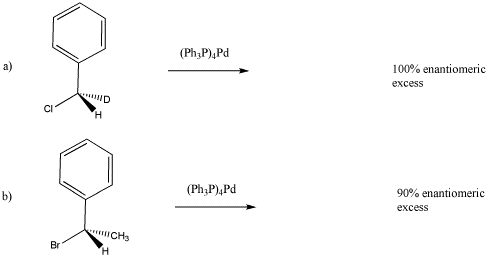
Reaction of the following deuterium-labeled alkyl chloride with tetrakis(triphenylphosphine) palladium produces an enantiomerically pure product (equation a). Draw the expected product.
However, reaction of a very similar alkyl halide produces a compound that is only 90% enantiomerically pure. Draw the major product and explain the reason that there is some racemization.