17.14: Soaps, Detergents, and Micelles
- Page ID
- 16417
This page describes ways of hydrolyzing esters - splitting them into carboxylic acids (or their salts) and alcohols by the action of water, dilute acid or dilute alkali. It starts by looking at the hydrolysis of simple esters like ethyl ethanoate, and goes on to look at hydrolyzing bigger, more complicated ones to make soap.
Technically, hydrolysis is a reaction with water. That is exactly what happens when esters are hydrolyzed by water or by dilute acids such as dilute hydrochloric acid. The alkaline hydrolysis of esters actually involves reaction with hydroxide ions, but the overall result is so similar that it is lumped together with the other two.
Hydrolysis using water or dilute acid
The reaction with pure water is so slow that it is never used. The reaction is catalyzed by dilute acid, and so the ester is heated under reflux with a dilute acid like dilute hydrochloric acid or dilute sulfuric acid. Here are two simple examples of hydrolysis using an acid catalyst.
First, hydrolyzing ethyl ethanoate:

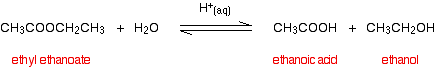
. . . and then hydrolyzing methyl propanoate:


Notice that the reactions are reversible. To make the hydrolysis as complete as possible, you would have to use an excess of water. The water comes from the dilute acid, and so you would mix the ester with an excess of dilute acid.
Hydrolysis using dilute alkali
This is the usual way of hydrolyzing esters. The ester is heated under reflux with a dilute alkali like sodium hydroxide solution. There are two advantages of doing this rather than using a dilute acid. The reactions are one-way rather than reversible, and the products are easier to separate. Taking the same esters as above, but using sodium hydroxide solution rather than a dilute acid:
First, hydrolyzing ethyl ethanoate using sodium hydroxide solution:

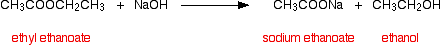
and then hydrolyzing methyl propanoate in the same way:

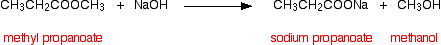
Notice that you get the sodium salt formed rather than the carboxylic acid itself. This mixture is relatively easy to separate. Provided you use an excess of sodium hydroxide solution, there will not be any ester left - so you don't have to worry about that. The alcohol formed can be distilled off. That's easy!
If you want the acid rather than its salt, all you have to do is to add an excess of a strong acid like dilute hydrochloric acid or dilute sulfuric acid to the solution left after the first distillation. If you do this, the mixture is flooded with hydrogen ions. These are picked up by the ethanoate ions (or propanoate ions or whatever) present in the salts to make ethanoic acid (or propanoic acid, etc). Because these are weak acids, once they combine with the hydrogen ions, they tend to stay combined. The carboxylic acid can now be distilled off.
Hydrolyzing complicated esters to make soap
This next bit deals with the alkaline hydrolysis (using sodium hydroxide solution) of the big esters found in animal and vegetable fats and oils. If the large esters present in animal or vegetable fats and oils are heated with concentrated sodium hydroxide solution exactly the same reaction happens as with the simple esters. A salt of a carboxylic acid is formed - in this case, the sodium salt of a big acid such as octadecanoic acid (stearic acid). These salts are the important ingredients of soap - the ones that do the cleaning. An alcohol is also produced - in this case, the more complicated alcohol, propane-1,2,3-triol (glycerol).
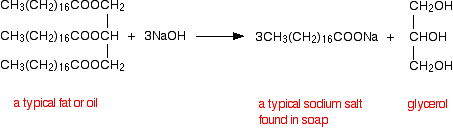
Because of its relationship with soap making, the alkaline hydrolysis of esters is sometimes known as saponification.
Contributors
Jim Clark (Chemguide.co.uk)

