10.4: Elimination Reactions of Alcohols- Dehydration
- Page ID
- 13900
The discussion of alkyl halide reactions noted that 2º and 3º-alkyl halides experience rapid E2 elimination when treated with strong bases such as hydroxide and alkoxides. Alcohols do not undergo such base-induced elimination reactions and are, in fact, often used as solvents for such reactions. This is yet another example of how leaving-group stability influences the rate of a reaction. When an alcohol is treated with sodium hydroxide, the following acid-base equilibrium occurs. Most alcohols are slightly weaker acids than water, so the left side is favored.
R–O–H + Na(+) OH(–)  R–O(–) Na(+) + H–OH
R–O(–) Na(+) + H–OH
The elimination of water from an alcohol is called dehydration. Recalling that water is a much better leaving group than hydroxide ion, it is sensible to use acid-catalysis rather than base-catalysis in such reactions. Four examples of this useful technique are shown below. Note that hydrohalic acids (HX) are not normally used as catalysts because their conjugate bases are good nucleophiles and may create substitution products. The conjugate bases of sulfuric and phosphoric acids are not good nucleophiles, and do not participate in substitution under typical conditions.
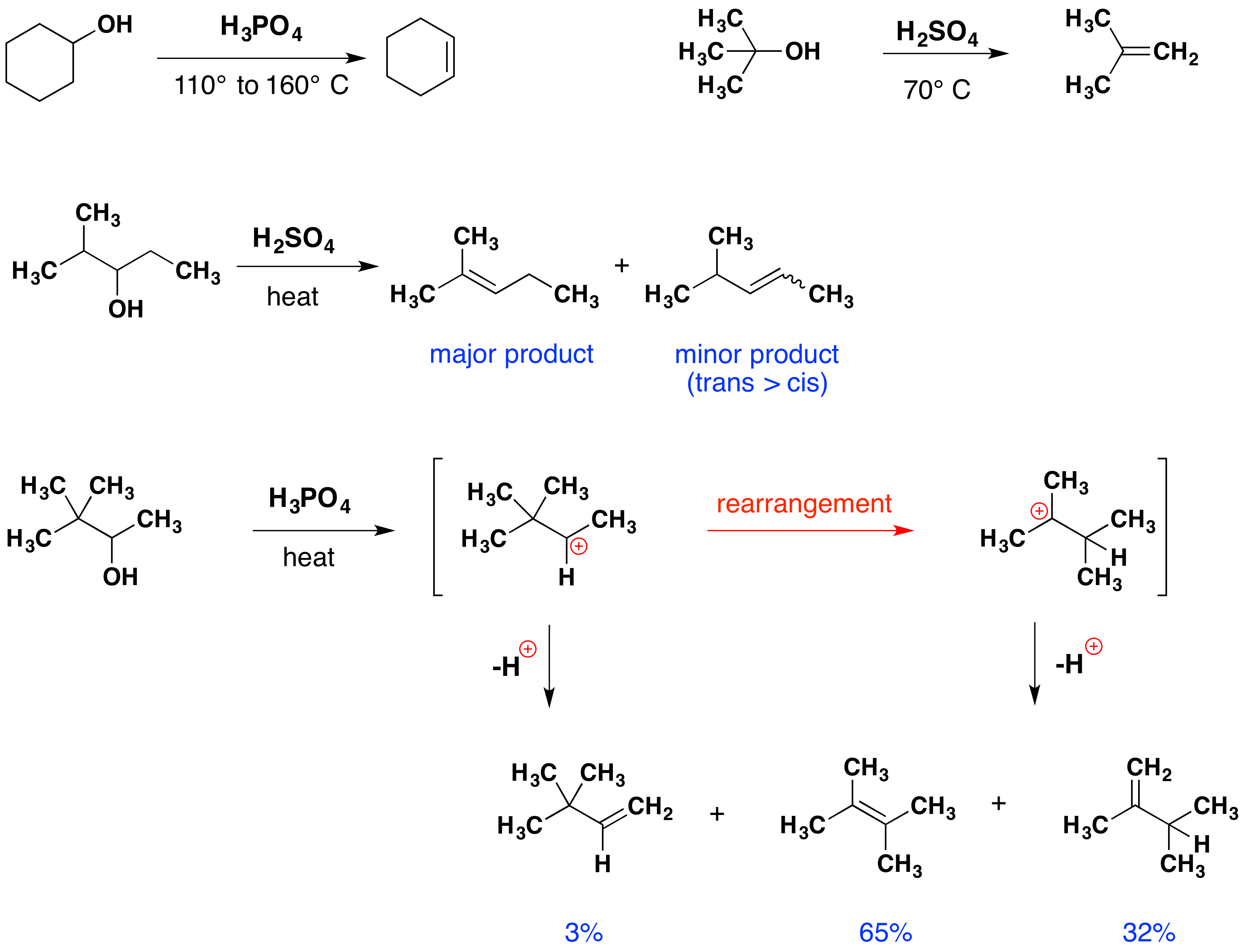
The first two examples (in the top row) are typical, and the more facile elimination of the 3º-alcohol suggests the predominant E1 character for the reaction. This agrees with the tendency of branched 1º and 2º-alcohols to yield rearrangement products, as shown in the last example. The last two reactions also demonstrate that the Zaitsev rule applies to alcohol dehydrations, as well as to alkyl halide eliminations. Therefore, the more highly-substituted double bond isomer is favored among the products.
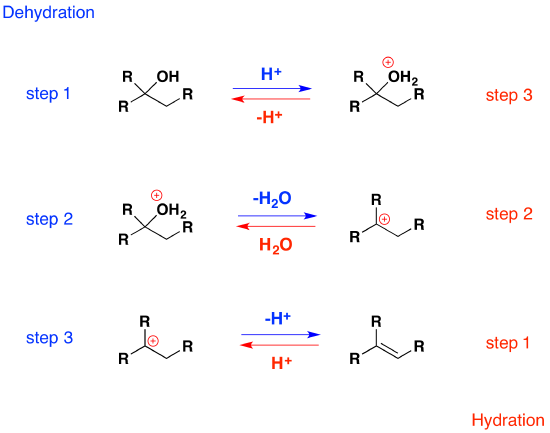
It should be noted that the acid-catalyzed dehydrations discussed here are the reverse of the acid-catalyzed hydration reactions of alkenes. Indeed, for these types of reversible reactions, the laws of thermodynamics require that the mechanism in both directions proceed by the same reaction path. This is known as the principle of microscopic reversibility. To illustrate, the following diagram lists the three steps in each transformation. The dehydration reaction is shown by the blue arrows; the hydration reaction by magenta arrows. The intermediates in these reactions are common to both, and common transition states are involved. This can be seen clearly in the energy diagrams depicted by clicking the button beneath the equations.
Base-induced E2 eliminations of alcohols may be achieved from sulfonate ester derivatives. This has the advantage of avoiding strong acids, which may cause molecular rearrangement or double bond migration in some cases. Because 3º-sulfonate derivatives are sometimes unstable, this procedure is best used with 1º and 2º-mesylates or tosylates. Application of this reaction sequence is shown here for 2-butanol. The Zaitsev rule favors the formation of 2-butene (cis + trans) over 1-butene.
| CH3CH2CH(CH3)–OH | CH3SO2Cl |
CH3CH2CH(CH3)–OSO2CH3 | C2H5O(–)Na(+) |
| CH3CH=CHCH3 + CH3CH2CH=CH2 + CH3SO2O(–) Na(+) + C2H5OH |
The E2 elimination of 3º-alcohols under relatively non-acidic conditions may be accomplished by treatment with phosphorous oxychloride (POCl3) in pyridine. This procedure is also effective with hindered 2º-alcohols, but for unhindered and 1º-alcohols, an SN2 chloride ion substitution of the chlorophosphate intermediate competes with elimination. Examples of these and related reactions are given in the following figure. The first equation shows the dehydration of a 3º-alcohol. The predominance of the non-Zaitsev product (less substituted double bond) is presumed due to steric hindrance of the methylene group hydrogen atoms, which interferes with the approach of the base at that site. The second example shows two elimination procedures applied to the same 2º-alcohol. The first uses the single step POCl3 method, which works well in this case because SN2 substitution is impeded by steric hindrance. The second method is another example in which an intermediate sulfonate ester confers halogen-like reactivity on an alcohol. In every case, the anionic leaving group is the conjugate base of a strong acid.

Pyrolytic syn-Eliminations
Ester derivatives of alcohols may undergo unimolecular syn-elimination upon heating. To see examples of these, Click Here.
Contributors
- William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry

