Cellulose
- Page ID
- 394
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Polysaccharides are carbohydrate polymers consisting of tens to hundreds to several thousand monosaccharide units. All of the common polysaccharides contain glucose as the monosaccharide unit. Polysaccharides are synthesized by plants, animals, and humans to be stored for food, structural support, or metabolized for energy.
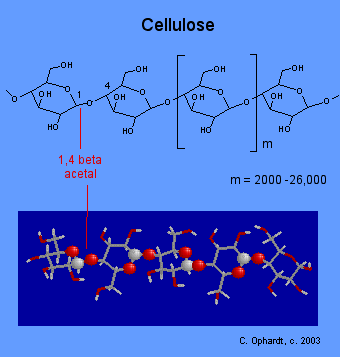
The major component in the rigid cell walls in plants is cellulose. Cellulose is a linear polysaccharide polymer with many glucose monosaccharide units. The acetal linkage is beta which makes it different from starch. This peculiar difference in acetal linkages results in a major difference in digestibility in humans. Humans are unable to digest cellulose because the appropriate enzymes to breakdown the beta acetal linkages are lacking. Indigestible cellulose is the fiber which aids in the smooth working of the intestinal tract.
Animals such as cows, horses, sheep, goats, and termites have symbiotic bacteria in the intestinal tract. These symbiotic bacteria possess the necessary enzymes to digest cellulose in the GI tract. They have the required enzymes for the breakdown or hydrolysis of the cellulose; the animals do not, not even termites, have the correct enzymes. No vertebrate can digest cellulose directly.
Even though we cannot digest cellulose, we find many uses for it including: Wood for building; paper products; cotton, linen, and rayon for clothes; nitrocellulose for explosives; cellulose acetate for films. The structure of cellulose consists of long polymer chains of glucose units connected by a beta acetal linkage. The graphic on the left shows a very small portion of a cellulose chain. All of the monomer units are beta-D-glucose, and all the beta acetal links connect C # 1 of one glucose to C # 4 of the next glucose.
Acetal Functional Group
Carbon # 1 is called the anomeric carbon and is the center of an acetal functional group. A carbon that has two ether oxygens attached is an acetal. The Beta position is defined as the ether oxygen being on the same side of the ring as the C # 6. In the chair structure this results in a horizontal or up projection. This is the same definition as the -OH in a hemiacetal.
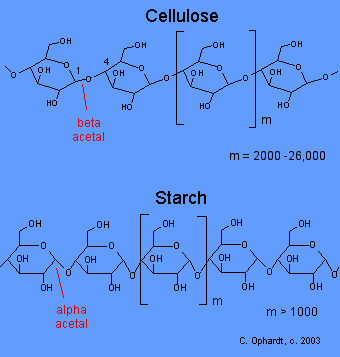
Compare Cellulose and Starch Structures
Cellulose: Beta glucose is the monomer unit in cellulose. As a result of the bond angles in the beta acetal linkage, cellulose is mostly a linear chain. Starch: Alpha glucose is the monomer unit in starch. As a result of the bond angles in the alpha acetal linkage, starch-amylose actually forms a spiral much like a coiled spring.
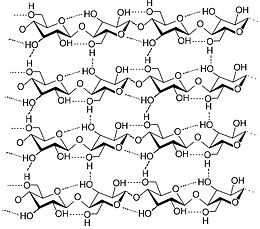
Fiber in the Diet
Dietary fiber is the component in food not broken down by digestive enzymes and secretions of the gastrointestinal tract. This fiber includes hemicelluloses, pectins, gums, mucilages, cellulose, (all carbohydrates) and lignin, the only non-carbohydrate component of dietary fiber. High fiber diets cause increased stool size and may help prevent or cure constipation. Cereal fiber, especially bran, is most effective at increasing stool size while pectin has little effect. Lignin can be constipating.
Fiber may protect against the development of colon cancer, for populations consuming high fiber diets have a low incidence of this disease. The slow transit time (between eating and elimination) associated with a low fiber intake would allow more time for carcinogens present in the colon to initiate cancer. But constipated people do not have a higher incidence of colon cancer than fast eliminators, so fiber's role in colon cancer remains unclear.
Dietary fiber may limit cholesterol absorption by binding bile acids. High fiber diets lower serum cholesterol and may prevent cardiovascular disease. Some fibers, such as pectin and rolled oats, are more effective than others, such as wheat, at lowering serum cholesterol. Dietary fiber is found only in plant foods such as fruits, vegetables, nuts, and grains. Whole wheat bread contains more fiber than white bread and apples contain more fiber than apple juice, which shows that processing food generally removes fiber.
Contributors
- Charles Ophardt, Professor Emeritus, Elmhurst College; Virtual Chembook

