Introduction
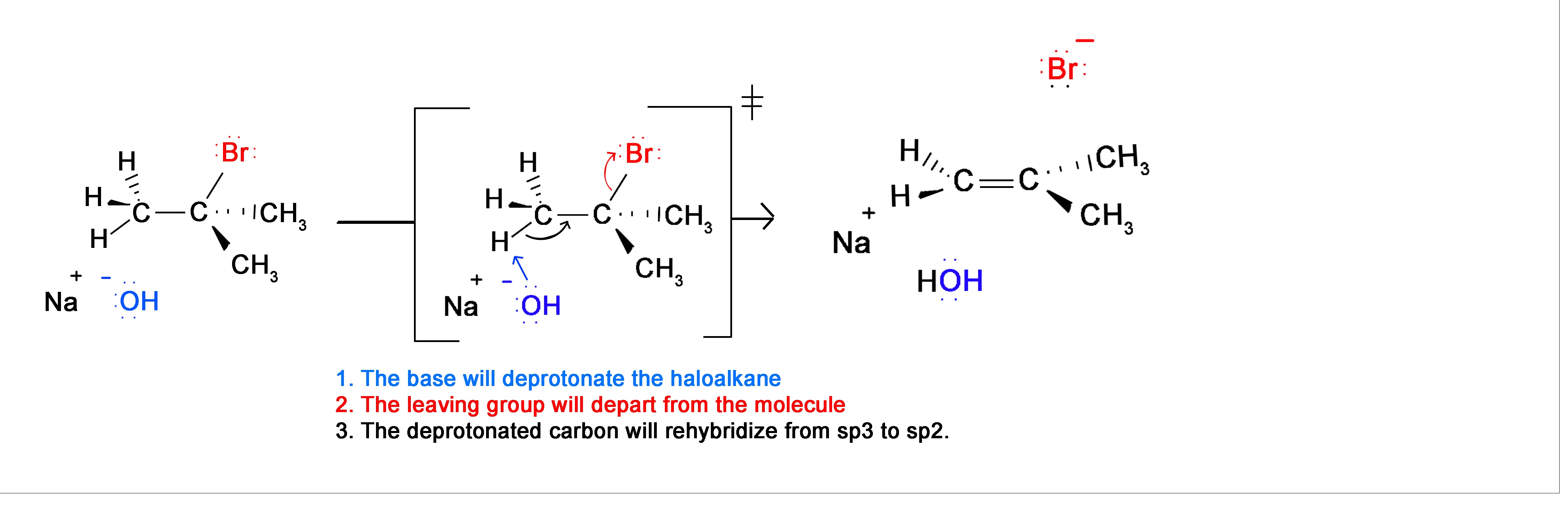
To synthesize alkynes from dihaloalkanes we use dehydrohalogenation. The majority of these reactions take place using alkoxide bases (other strong bases can also be used) with high temperatures. This combination results in the majority of the product being from the E2 mechanism. Recall that the E2 mechanism is a concerted reaction (occurs in 1 step). However, in this 1 step there are 3 different changes in the molecule. This is the reaction between 2-bromo-2-methylpropane and sodium hydroxide.
Now, if we apply this concept using 2 halides on vicinal or geminal carbons, the E2 reaction will take place twice resulting in the formation of 2 pi bonds and an alkyne as shown in the examples below where the strong base is symbolized B-.
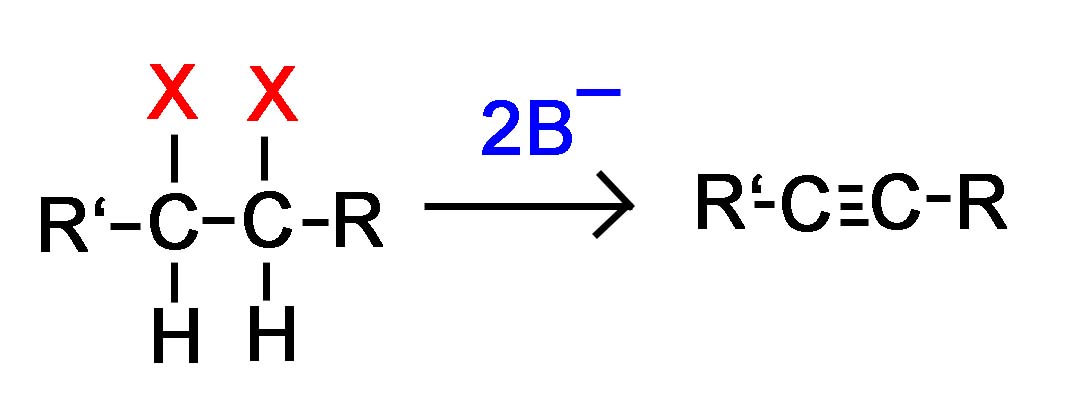
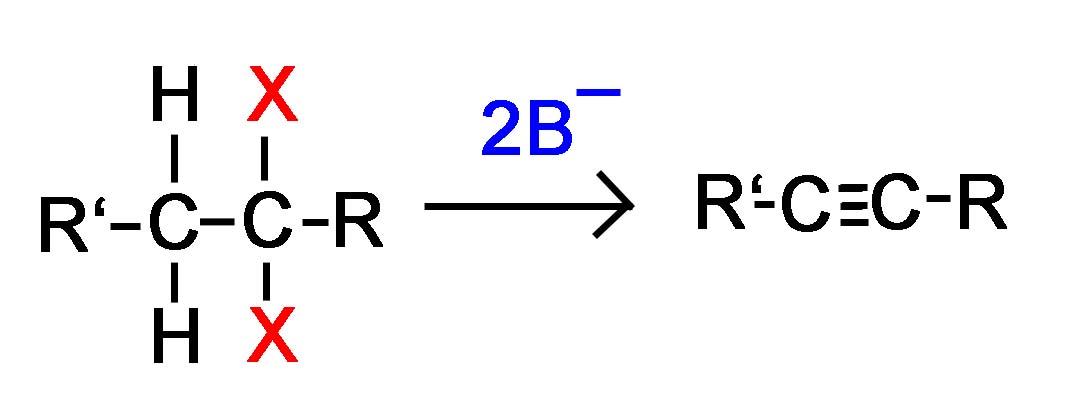
Double E2 of a Vicinal Dihalide Double E2 of a Geminal Dihalilde
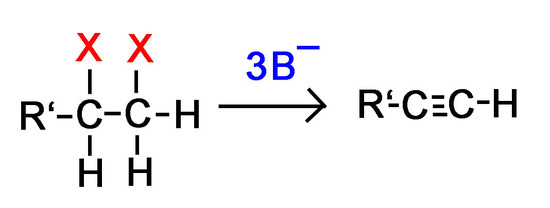
It is important to note that the reaction of terminal haloalkanes requires 3 equivalents of base instead of 2 because of the relative acidity of alkynes that is discussed in a later section of this chapter.
The mechanism of a reaction between 2,3-dibromopentane with sodium amide in liquid ammonia is shown below where liquid ammonia is not part of the reaction, but is used as a solvent.
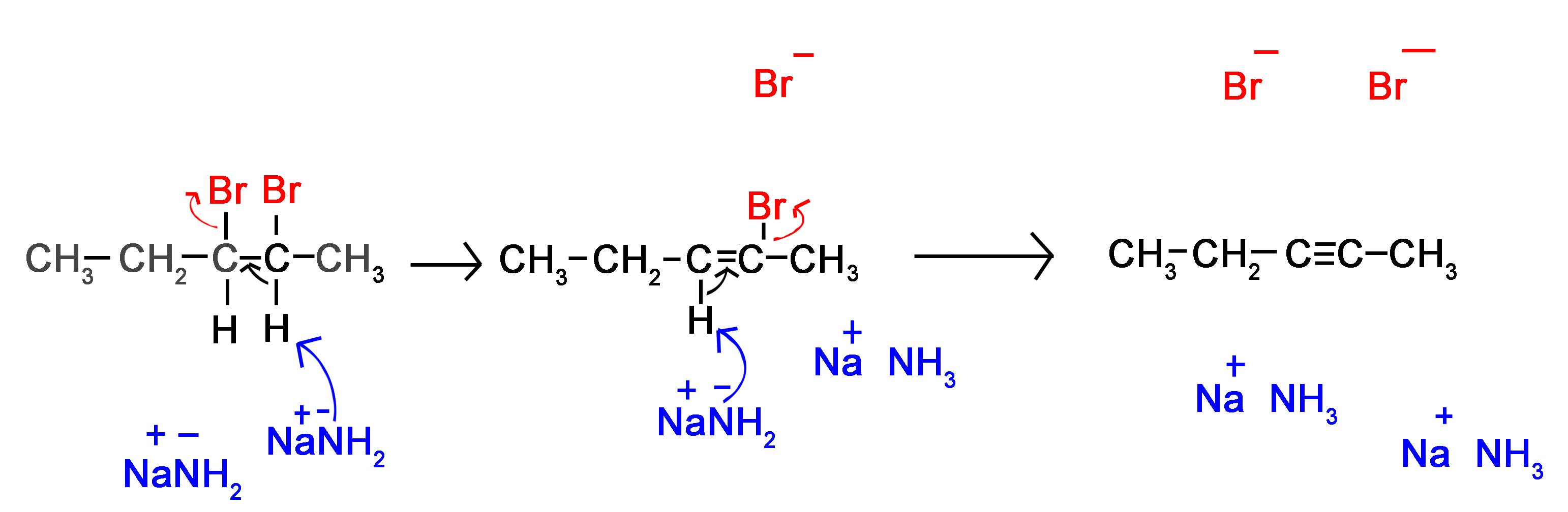
Notice the intermediate of the alkyne synthesis. It is stereospecifically in its anti form. Because the second proton and halogen are pulled off the molecule this is unimportant to the synthesis of alkynes.
Preparation of Alkynes from Alkenes
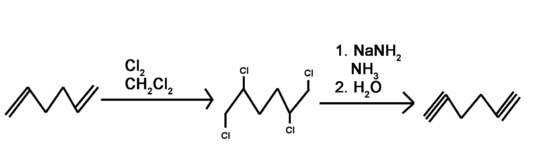
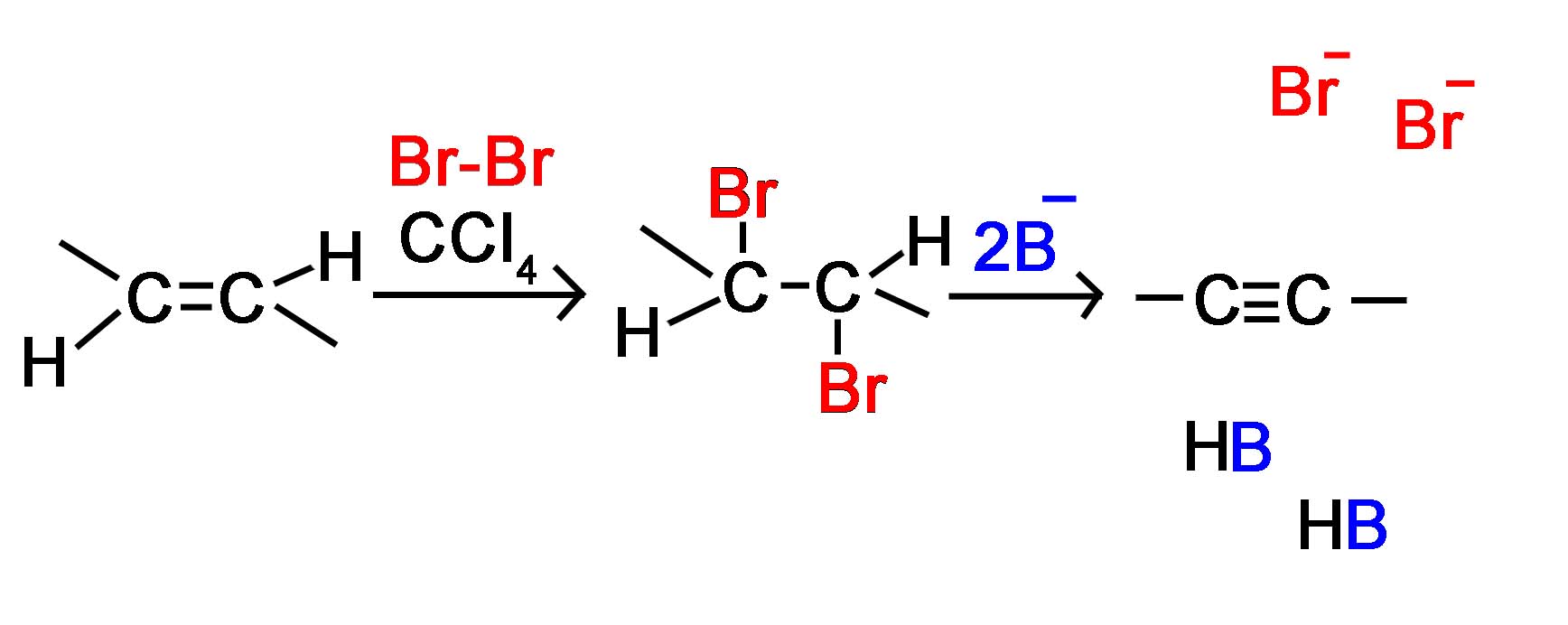
Lastly, we will briefly look at how to prepare alkynes from alkenes. This is a simple process using first halogenation of the alkene bond to form the dihaloalkane, and next, using the double elimination process form the alkyne.
This first process is gone over in much greater detail in the page on halogenation of an alkene. In general, chlorine or bromine is used with an inert halogenated solvent like chloromethane to create a vicinal dihalide from an alkene. The vicinal dihalide formed is then reacted with a strong base and heated to produce an alkyne. The two-step reaction pathway is shown below.
In The Lab
Due to the strong base and high temperatures needed for this reaction to take place, the triple bond may change positions. An example of this is when reactants that should form a terminal alkyne, form a 2-alkyne instead. The use of NaNH2 in liquid NH3 is used in order to prevent this from happening due to its lower reacting temperature. Even so, most chemists will prefer to use nucleophilic substitution instead of elimination when trying to form a terminal alkyne.
Exercise \(\PageIndex{1}\)
Question 1: Why would we need 3 bases for every terminal dihaloalkane instead of 2 in order to form an alkyne?
Question 2: What are the major products of the following reactions:
a.) 1,2-Dibromopentane with sodium amide in liquid ammonia
b.) 1-Pentene first with Br2 and chloromethane, followed by sodium ethoxide (Na+ -O-CH2CH3)
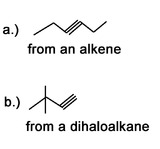
Question 3: What would be good starting molecules for the synthesis of the following molecules:
Question 4: Use a 6 carbon diene to synthesize a 6 carbon molecule with 2 terminal alkynes.
- Answer
-
Answer 1: Remember that hydrogen atoms on terminal alkynes make the alkyne acidic. One of the base molecules will pull off the terminal hydrogen instead of one of the halides like we want.
Answer 2:
a.) 1-Pentyne
b.) 1-Pentyne
Answer 3:
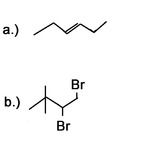
Answer 4: Bromine or chlorine can be used with different inert solvents for the halogenation. This can be done using many different bases. Liquid ammonia is used as a solvent and needs to be followed by an aqueous work-up.