8.12: Orientation of Electrophilic Additions - Markovnikov's Rule
- Last updated
- Save as PDF
- Page ID
- 227099
Objectives
After completing this section, you should be able to
- use Markovnikov’s rule to predict the product formed when a protic acid, HX, reacts with an alkene.
- identify the protic acid, HX, and the alkene that must be reacted together to produce a given alkyl halide. [Note: Special conditions are needed if an alkyl iodide is to be produced.]
- distinguish among primary, secondary and tertiary carbocations.
Key Terms
Make certain that you can define, and use in context, the key terms below.
- Markovnikov’s rule
- regioselective (regiospecific)
Study Notes
Recall the definitions of primary, secondary and tertiary hydrogen atoms given in Section 3.3. It follows that a “primary carbocation” is a carbocation in which the carbon atom carrying the positive charge is bonded to only one other carbon atom, a “secondary carbocation” is one in which the carbon atom carrying the positive charge is bonded to two other carbon atoms, and so on.
Regioselectivity
If more than one reaction could occur between a set of reactants under the same conditions giving products that are constitutional isomers and if one product forms in greater amounts than the others, the overall reaction is said to be regioselective.
Say three reactions could occur between the hypothetical reactants A and B under the same conditions giving the constitutionally isomeric products C, D, and E.
There are two possibilities:
1. The three products form in equal amounts, i.e., of the total product 33% is C, another 33% D, the remaining 33% E. (These percentages are called relative yields of the products.)
If this is what is observed, the overall reaction between A and B is not regioselective.
2. One product forms in greater amounts than the others. Say, for example, the relative yields of C, D, and E are 25%, 50%, and 25%, respectively.
If this is what is observed, the overall reaction between A and B is regioselective.
eg:
Experimentally, 2 is the major product; 3 is the minor product. Thus, the overall reaction between 1 and HBr is regioselective toward 2.
If more than one reaction could occur between a set of reactants under the same conditions giving products that are constitutional isomers and if only one product is observed, the overall reaction is said to be 100% regioselective or regiospecific.
eg:
The only observed product is 5. (Relative yields of 5 and 6 are 100% and 0%, respectively.) Thus the overall reaction between 4 and HBr is regiospecific toward 5.
Regiospecificity is merely the limiting case of regioselectivity. All regiospecific reactions are regioselective, but not all regioselective reactions are regiospecific.
Addition to unsymmetrical alkenes
During the electrophilic addition of HX to an alkene, the halide (X) could attach to either carbon in the double bond producing two different isomers as products. But, when an unsymmertrically alkyl substituted alkene undergoes an electrophillic addition with HX a single isomer is typically produced. For example, if propene were reacted with HBr, two products could possibly form: 2-bromolpropane and 1-bromopropane. However, 2-bromopropane is produced as the reaction's only product. Reactions are called regiospecific when only one of multiple possible isomers is exclusively formed.
Example \(\PageIndex{1}\)
If HCl adds to an unsymmetrical alkene like propene what will the major product be?
Solution
The regiospecificity of electrophilic additions to alkenes is commonly known as Markovnikov's rule, after the Russian chemist Vladimir Markovnikov who proposed it in 1869. The electrophilic addition of HX to an alkene is said to follow Markovnikov's rule.
Markovnikov's rule: During the electrophilic addition of HX to an alkene, the H adds to the carbon of the double bond with the fewest number of alkyl substitutent. The halide (X) adds to the double bond carbon with the most alkyl substituents. Although Markovnikov's rule has been specifically stated for the electrophilic addition of HX, later in this chapter many more reaction will be shown to also follow Markovnikov's rule.
It is important to point out that Markovnikov's rule only truly applies when there is a difference between the number of alky groups attached to each carbon in the double bond. When both carbon of the double bond have the same degree of alkyl substitution, Markovnikovs's rule becomes void and a mixture of both possible isomers is produced.
To consider an explanation for why Markovnikov's rule holds true, the mechanism of the reaction needs to be considered. As seen in the previous section, the mechanism starts with the addition of H to a carbon in the double bond. This in turn caused the other double bond carbon to become a carbocation intermediate. In the second step of the mechanism the halide ion attacks the carbocation to form a C-X. Because Markovnikov's rule says that the halogen adds to the carbon in the double bond with the most alky substituents, it can be said the carbocation also prefers to form on the carbon with the most alkyl substituents. The reason for this holds the explaination for Markovnikov's rule. To simplify this idea, Markovnikov's rule can be restated in a different form.
Markovnikov's rule: During the electrophilic addition of HX to an alkene, the carbocation intermediate forms on the double bond carbon with the greatest number of alkyl substitutents.
Predicting the Product of an Electrophilic Addition with HX
Overall, during the electrophilic addition of HX to an alkene there are two major changes in the bonding. First, the pi bond of the alkene is broken. Second, a single bond is formed on each carbon that was originally in the double bond. The two single bonds will become attached to and H and an X. If the alkene is unsymmertrically alkyl substituted, Markovnikov's rule is followed and the X will be bonded to the more alky substituted carbon and the H to the less substiuted. If the alkene is symmertrically alkyl substituted a mixture of isomers will be produced in the product.
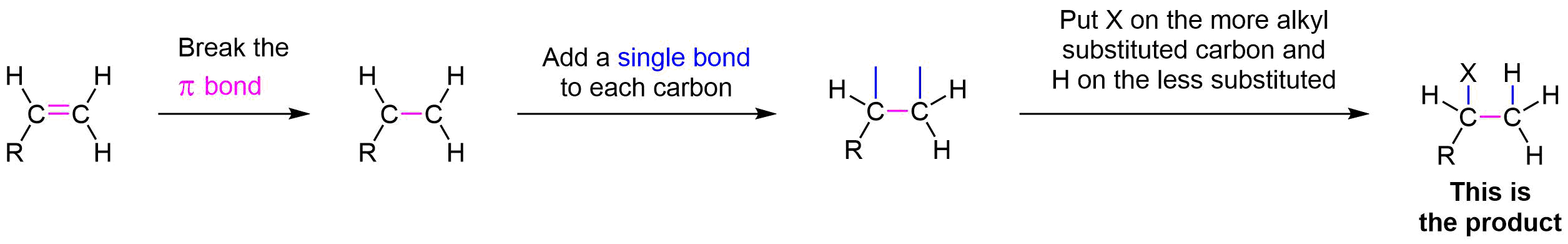
Worked Example\(\PageIndex{1}\)
Please draw the product of the following reaction:
- Answer
-
In answering these types of questions it is always important to first determine which reaction is occurring. Because an alkene is the reactant and HBr is the product this reaction is an electrophilic addition. Overall, the double bond will be broken as H and Br are added. The next step is to determine if Markovnikov's rule needs to be applied. In the reactant's double bond the upper carbon has two alkyl substituents and the lower carbon has only two. Markovnikov's rule says that Br will attach to the upper carbon and H to the lower.
Planning the Synthesis of an Alkyl Halide using Electrophilic Addition
Understanding the starting material and reaction required for the synthesis a specific target molecule is an important concept in organic chemistry. The preferred method for answering these types of questions is to work backwards from the target molecule. Often there will be multiple variations of reactions and starting materials capable synthesizing the target molecule. Further analysis of these variations can draw out strengths and weaknesses and lead to the pathway with the best chance of success.
To create a possible starting material for a given alkyl halide simply reverse the bonding changes expected during an electrophilic addition. Remove the C-X single bond and a C-H bond from an adjacent carbon. Then connect these two carbons with a double bond.
Worked Example \(\PageIndex{1}\)
What alkene would be required to make the following 2-bromopentane using an electrophilic addition?
- Answer
-
Analysis: Possible starting materials can be made by first removing the C-Br bond and an adjacent C-H bond. Then connecting the two carbons with a double bond. Because there are two different locations where an adjacent C-H bond can be removed there will be two possible starting material.
Solution: When comparing the the possible starting materials, #1 will follow Markovnikov's rule and produce 2-bromopentane as the sole product. Starting material #2 is symmertrically alkyl substituted, so Markovnikov's rule does not apply. A mixture of 2-bromopentane and 3-bromopentane would be produced. Between the two possible starting material #1 would be preferred because only one product is produced.
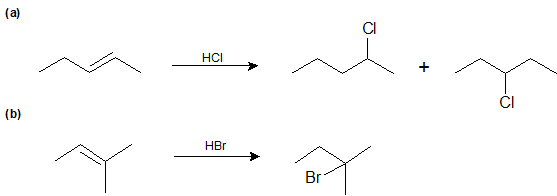
Exercise \(\PageIndex{1}\)
Predict the product(s) for the following reactions:
- Answer
-
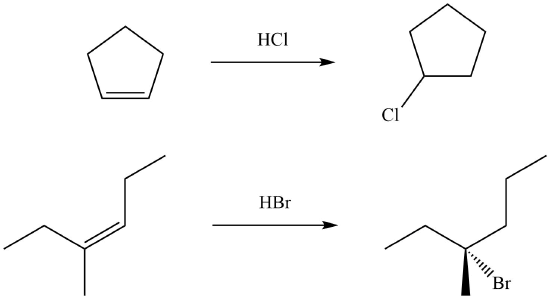
Exercise \(\PageIndex{2}\)
In each case, suggest an alkene that would give the product shown.
- Answer
-
Exercise \(\PageIndex{3}\)
Give the IUPAC name for the product of the following reaction.
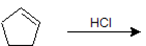
- Answer
-
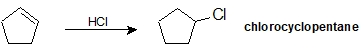
Exercise \(\PageIndex{4}\)
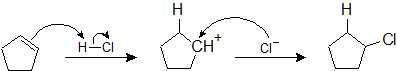
Draw the reaction mechanism of the previous problem
- Answer
-
Exercise \(\PageIndex{5}\)
Identify the products of the following reactions.
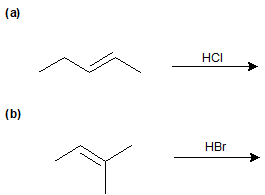
- Answer
-