2.4: Physical Properties
- Last updated
- Save as PDF
- Page ID
- 227012
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
The molecule is the smallest observable group of uniquely bonded atoms that represent the composition, configuration and characteristics of a pure compound. Our chief focus up to this point has been to discover and describe the ways in which atoms bond together to form molecules. Since all observable samples of compounds and mixtures contain a very large number of molecules (ca.!020), we must also concern ourselves with interactions between molecules, as well as with their individual structures. Indeed, many of the physical characteristics of compounds that are used to identify them (e.g. boiling points, melting points and solubilities) are due to intermolecular interactions.
All atoms and molecules have a weak attraction for one another, known as van der Waals attraction. This attractive force has its origin in the electrostatic attraction of the electrons of one molecule or atom for the nuclei of another. If there were no van der Waals forces, all matter would exist in a gaseous state, and life as we know it would not be possible. It should be noted that there are also smaller repulsive forces between molecules that increase rapidly at very small intermolecular distances.
Boiling & Melting Points |
|---|
Boiling and Melting Points
For general purposes it is useful to consider temperature to be a measure of the kinetic energy of all the atoms and molecules in a given system. As temperature is increased, there is a corresponding increase in the vigor of translational and rotation motions of all molecules, as well as the vibrations of atoms and groups of atoms within molecules. Experience shows that many compounds exist normally as liquids and solids; and that even low-density gases, such as hydrogen and helium, can be liquified at sufficiently low temperature and high pressure. A clear conclusion to be drawn from this fact is that intermolecular attractive forces vary considerably, and that the boiling point of a compound is a measure of the strength of these forces. Thus, in order to break the intermolecular attractions that hold the molecules of a compound in the condensed liquid state, it is necessary to increase their kinetic energy by raising the sample temperature to the characteristic boiling point of the compound.
The following table illustrates some of the factors that influence the strength of intermolecular attractions. The formula of each entry is followed by its formula weight in parentheses and the boiling point in degrees Celsius. First there is molecular size. Large molecules have more electrons and nuclei that create van der Waals attractive forces, so their compounds usually have higher boiling points than similar compounds made up of smaller molecules. It is very important to apply this rule only to like compounds. The examples given in the first two rows are similar in that the molecules or atoms are spherical in shape and do not have permanent dipoles. Molecular shape is also important, as the second group of compounds illustrate. The upper row consists of roughly spherical molecules, whereas the isomers in the lower row have cylindrical or linear shaped molecules. The attractive forces between the latter group are generally greater. Finally, permanent molecular dipoles generated by polar covalent bonds result in even greater attractive forces between molecules, provided they have the mobility to line up in appropriate orientations. The last entries in the table compare non-polar hydrocarbons with equal-sized compounds having polar bonds to oxygen and nitrogen. Halogens also form polar bonds to carbon, but they also increase the molecular mass, making it difficult to distinguish among these factors.
Boiling Points (ºC) of Selected Elements and Compounds |
||||
|---|---|---|---|---|
|
Increasing Size |
||||
| Atomic | Ar (40) -186 | Kr (83) -153 | Xe (131) -109 | |
| Molecular | CH4 (16) -161 | (CH3)4C (72) 9.5 | (CH3)4Si (88) 27 | CCl4 (154) 77 |
|
Molecular Shape |
||||
| Spherical: | (CH3)4C (72) 9.5 | (CH3)2CCl2 (113) 69 | (CH3)3CC(CH3)3 (114) 106 | |
| Linear: | CH3(CH2)3CH3 (72) 36 | Cl(CH2)3Cl (113) 121 | CH3(CH2)6CH3 (114) 126 | |
|
Molecular Polarity |
||||
| Non-polar: | H2C=CH2 (28) -104 | F2 (38) -188 | CH3C≡CCH3 (54) -32 | CF4 (88) -130 |
| Polar: | H2C=O (30) -21 | CH3CH=O (44) 20 | (CH3)3N (59) 3.5 | (CH3)2C=O (58) 56 |
| HC≡N (27) 26 | CH3C≡N (41) 82 | (CH2)3O (58) 50 | CH3NO2 (61) 101 | |
The melting points of crystalline solids cannot be categorized in as simple a fashion as boiling points. The distance between molecules in a crystal lattice is small and regular, with intermolecular forces serving to constrain the motion of the molecules more severely than in the liquid state. Molecular size is important, but shape is also critical, since individual molecules need to fit together cooperatively for the attractive lattice forces to be large. Spherically shaped molecules generally have relatively high melting points, which in some cases approach the boiling point. This reflects the fact that spheres can pack together more closely than other shapes. This structure or shape sensitivity is one of the reasons that melting points are widely used to identify specific compounds. The data in the following table serves to illustrate these points.
| Compound | Formula | Boiling Point | Melting Point |
|---|---|---|---|
| pentane | CH3(CH2)3CH3 | 36ºC | –130ºC |
| hexane | CH3(CH2)4CH3 | 69ºC | –95ºC |
| heptane | CH3(CH2)5CH3 | 98ºC | –91ºC |
| octane | CH3(CH2)6CH3 | 126ºC | –57ºC |
| nonane | CH3(CH2)7CH3 | 151ºC | –54ºC |
| decane | CH3(CH2)8CH3 | 174ºC | –30ºC |
| tetramethylbutane | (CH3)3C-C(CH3)3 | 106ºC | +100ºC |
Notice that the boiling points of the unbranched alkanes (pentane through decane) increase rather smoothly with molecular weight, but the melting points of the even-carbon chains increase more than those of the odd-carbon chains. Even-membered chains pack together in a uniform fashion more compactly than do odd-membered chains. The last compound, an isomer of octane, is nearly spherical and has an exceptionally high melting point (only 6º below the boiling point).
Hydrogen Bonding |
|---|
Hydrogen Bonding
The most powerful intermolecular force influencing neutral (uncharged) molecules is the hydrogen bond. If we compare the boiling points of methane (CH4) -161ºC, ammonia (NH3) -33ºC, water (H2O) 100ºC and hydrogen fluoride (HF) 19ºC, we see a greater variation for these similar sized molecules than expected from the data presented above for polar compounds. This is shown graphically in the following chart. Most of the simple hydrides of group IV, V, VI & VII elements display the expected rise in boiling point with molecular mass, but the hydrides of the most electronegative elements (nitrogen, oxygen and fluorine) have abnormally high boiling points for their mass.
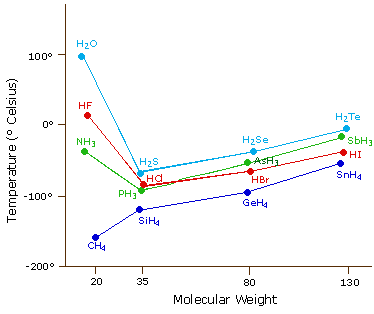
The exceptionally strong dipole-dipole attractions that cause this behavior are called the hydrogen bond. Hydrogen forms polar covalent bonds to more electronegative atoms such as oxygen, and because a hydrogen atom is quite small, the positive end of the bond dipole (the hydrogen) can approach neighboring nucleophilic or basic sites more closely than can other polar bonds. Coulombic forces are inversely proportional to the sixth power of the distance between dipoles, making these interactions relatively strong, although they are still weak (ca. 4 to 5 kcal per mole) compared with most covalent bonds. The unique properties of water are largely due to the strong hydrogen bonding that occurs between its molecules. In the following diagram the hydrogen bonds are depicted as magenta dashed lines.
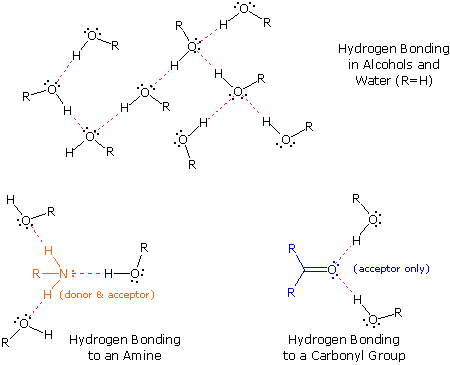
The molecule providing a polar hydrogen for a hydrogen bond is called a donor. The molecule that provides the electron rich site to which the hydrogen is attracted is called an acceptor. Water and alcohols may serve as both donors and acceptors, whereas ethers, aldehydes, ketones and esters can function only as acceptors. Similarly, primary and secondary amines are both donors and acceptors, but tertiary amines function only as acceptors. Once you are able to recognize compounds that can exhibit intermolecular hydrogen bonding, the relatively high boiling points they exhibit become understandable. The data in the following table serve to illustrate this point.
| Compound | Formula | Mol. Wt. | Boiling Point | Melting Point |
|---|---|---|---|---|
| dimethyl ether | CH3OCH3 | 46 | –24ºC | –138ºC |
| ethanol | CH3CH2OH | 46 | 78ºC | –130ºC |
| propanol | CH3(CH2)2OH | 60 | 98ºC | –127ºC |
| diethyl ether | (CH3CH2)2O | 74 | 34ºC | –116ºC |
| propyl amine | CH3(CH2)2NH2 | 59 | 48ºC | –83ºC |
| methylaminoethane | CH3CH2NHCH3 | 59 | 37ºC | |
| trimethylamine | (CH3)3N | 59 | 3ºC | –117ºC |
| ethylene glycol | HOCH2CH2OH | 62 | 197ºC | –13ºC |
| acetic acid | CH3CO2H | 60 | 118ºC | 17ºC |
| ethylene diamine | H2NCH2CH2NH2 | 60 | 118ºC | 8.5ºC |
Alcohols boil considerably higher than comparably sized ethers (first two entries), and isomeric 1º, 2º & 3º-amines, respectively, show decreasing boiling points, with the two hydrogen bonding isomers being substantially higher boiling than the 3º-amine (entries 5 to 7). Also, O–H---O hydrogen bonds are clearly stronger than N–H---N hydrogen bonds, as we see by comparing propanol with the amines. 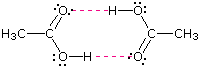 As expected, the presence of two hydrogen bonding functions in a compound raises the boiling point even further. Acetic acid (the ninth entry) is an interesting case. A dimeric species, shown on the right, held together by two hydrogen bonds is a major component of the liquid state. If this is an accurate representation of the composition of this compound then we would expect its boiling point to be equivalent to that of a C4H8O4 compound (formula weight = 120). A suitable approximation of such a compound is found in tetramethoxymethane, (CH3O)4C, which is actually a bit larger (formula weight = 136) and has a boiling point of 114ºC. Thus, the dimeric hydrogen bonded structure appears to be a good representation of acetic acid in the condensed state.
As expected, the presence of two hydrogen bonding functions in a compound raises the boiling point even further. Acetic acid (the ninth entry) is an interesting case. A dimeric species, shown on the right, held together by two hydrogen bonds is a major component of the liquid state. If this is an accurate representation of the composition of this compound then we would expect its boiling point to be equivalent to that of a C4H8O4 compound (formula weight = 120). A suitable approximation of such a compound is found in tetramethoxymethane, (CH3O)4C, which is actually a bit larger (formula weight = 136) and has a boiling point of 114ºC. Thus, the dimeric hydrogen bonded structure appears to be a good representation of acetic acid in the condensed state.
A related principle is worth noting at this point. Although the hydrogen bond is relatively weak (ca. 4 to 5 kcal per mole), when several such bonds exist the resulting structure can be quite robust. The hydrogen bonds between cellulose fibers confer great strength to wood and related materials.
Water Solubility |
|---|
Solubility in Water
Water has been referred to as the "universal solvent", and its widespread distribution on this planet and essential role in life make it the benchmark for discussions of solubility. Water dissolves many ionic salts thanks to its high dielectric constant and ability to solvate ions. The former reduces the attraction between oppositely charged ions and the latter stabilizes the ions by binding to them and delocalizing charge density. Many organic compounds, especially alkanes and other hydrocarbons, are nearly insoluble in water. Organic compounds that are water soluble, such as most of those listed in the above table, generally have hydrogen bond acceptor and donor groups. The least soluble of the listed compounds is diethyl ether, which can serve only as a hydrogen bond acceptor and is 75% hydrocarbon in nature. Even so, diethyl ether is about two hundred times more soluble in water than is pentane.
The chief characteristic of water that influences these solubilities is the extensive hydrogen bonded association of its molecules with each other. This hydrogen bonded network is stabilized by the sum of all the hydrogen bond energies, and if nonpolar molecules such as hexane were inserted into the network they would destroy local structure without contributing any hydrogen bonds of their own. Of course, hexane molecules experience significant van der Waals attraction to neighboring molecules, but these attractive forces are much weaker than the hydrogen bond. Consequently, when hexane or other nonpolar compounds are mixed with water, the strong association forces of the water network exclude the nonpolar molecules, which must then exist in a separate phase. This is shown in the following illustration, and since hexane is less dense than water, the hexane phase floats on the water phase.
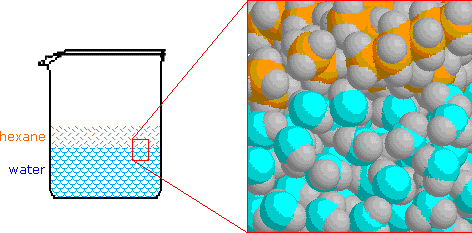
It is important to remember this tendency of water to exclude nonpolar molecules and groups, since it is a factor in the structure and behavior of many complex molecular systems. A common nomenclature used to describe molecules and regions within molecules is hydrophilic for polar, hydrogen bonding moieties and hydrophobic for nonpolar species.
Contributors
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry

