5.4: Titrations
- Last updated
- Save as PDF
- Page ID
- 98552
↵
Learning Objectives
- To use titration methods to analyze solutions quantitatively.
To determine the amounts or concentrations of substances present in a sample, chemists use a combination of chemical reactions and stoichiometric calculations in a methodology called quantitative analysis. Suppose, for example, we know the identity of a certain compound in a solution but not its concentration. If the compound reacts rapidly and completely with another reactant, we may be able to use the reaction to determine the concentration of the compound of interest. In a titration, a carefully measured volume of a solution of known concentration, called the titrant, is added to a measured volume of a solution containing a compound whose concentration is to be determined (the unknown). The reaction used in a titration can be an acid–base reaction, a precipitation reaction, or an oxidation–reduction reaction. In all cases, the reaction chosen for the analysis must be fast, complete, and specific; that is, only the compound of interest should react with the titrant. The equivalence point is reached when a stoichiometric amount of the titrant has been added—the amount required to react completely with the unknown.
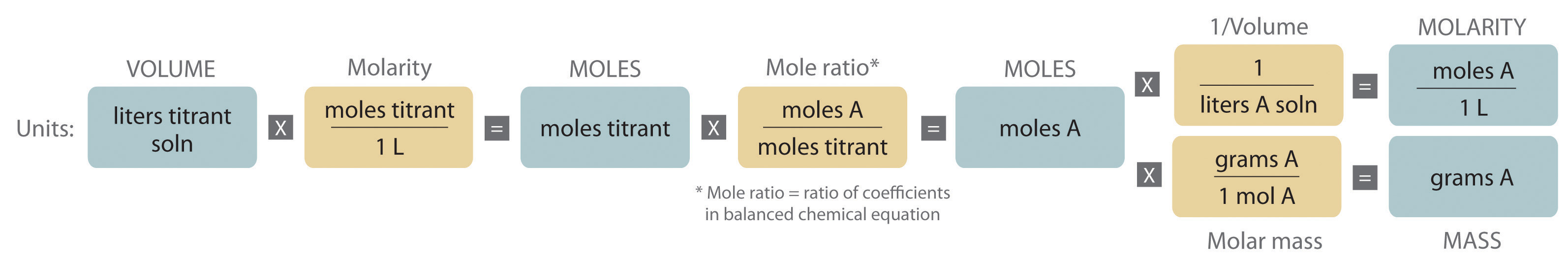
Determining the Concentration of an Unknown Solution Using a Titration
The chemical nature of the species present in the unknown dictates which type of reaction is most appropriate and also how to determine the equivalence point. The volume of titrant added, its concentration, and the coefficients from the balanced chemical equation for the reaction allow us to calculate the total number of moles of the unknown in the original solution. Because we have measured the volume of the solution that contains the unknown, we can calculate the molarity of the unknown substance. This procedure is summarized graphically here:
Example \(\PageIndex{1}\): Potassium Permanganate
The calcium salt of oxalic acid [Ca(O2CCO2)] is found in the sap and leaves of some vegetables, including spinach and rhubarb, and in many ornamental plants. Because oxalic acid and its salts are toxic, when a food such as rhubarb is processed commercially, the leaves must be removed, and the oxalate content carefully monitored.
The reaction of MnO4− with oxalic acid (HO2CCO2H) in acidic aqueous solution produces Mn2+ and CO2:
\( \begin{pmatrix}
MnO_{4}^{-}\left ( aq \right )+HO_{2}CCO_{2}H\left ( aq \right ) &\rightarrow Mn^{2+}\left ( aq \right )&+CO_{2}\left ( g \right )+H_{2}O\left ( l \right ) \\
purple & colorless &
\end{pmatrix} \)
Because this reaction is rapid and goes to completion, potassium permanganate (KMnO4) is widely used as a reactant for determining the concentration of oxalic acid.
Suppose you stirred a 10.0 g sample of canned rhubarb with enough dilute H2SO4(aq) to obtain 127 mL of colorless solution. Because the added permanganate is rapidly consumed, adding small volumes of a 0.0247 M KMnO4 solution, which has a deep purple color, to the rhubarb extract does not initially change the color of the extract. When 15.4 mL of the permanganate solution have been added, however, the solution becomes a faint purple due to the presence of a slight excess of permanganate. If we assume that oxalic acid is the only species in solution that reacts with permanganate, what percentage of the mass of the original sample was calcium oxalate? The video below demonstrates the titration when small, measured amounts of a known permaganate solution are added. At the endpoint, the number of moles of permaganate added equals the number of moles of oxalate in the solution, thus determining how many moles of oxalate we started with.
Given: equation, mass of sample, volume of solution, and molarity and volume of titrant
Asked for: mass percentage of unknown in sample
Strategy:
- Balance the chemical equation for the reaction using oxidation states.
- Calculate the number of moles of permanganate consumed by multiplying the volume of the titrant by its molarity. Then calculate the number of moles of oxalate in the solution by multiplying by the ratio of the coefficients in the balanced chemical equation. Because calcium oxalate contains a 1:1 ratio of Ca2+:−O2CCO2−, the number of moles of oxalate in the solution is the same as the number of moles of calcium oxalate in the original sample.
- Find the mass of calcium oxalate by multiplying the number of moles of calcium oxalate in the sample by its molar mass. Divide the mass of calcium oxalate by the mass of the sample and convert to a percentage to calculate the percentage by mass of calcium oxalate in the original sample.
Solution:
A As in all other problems of this type, the first requirement is a balanced chemical equation for the reaction. Using oxidation states gives
\[2MnO_4^-(aq) + 5HO_2CCO_2H(aq) + 6H^+(aq) \rightarrow 2Mn^{2+}(aq) + 10CO_2(g) + 8H_2O(l) \nonumber \]
Thus each mole of MnO4− added consumes 2.5 mol of oxalic acid.
B Because we know the concentration of permanganate (0.0247 M) and the volume of permanganate solution that was needed to consume all the oxalic acid (15.4 mL), we can calculate the number of moles of MnO4− consumed. To do this we first convert the volume in mL to a volume in liters. Then simply multiplying the molarity of the solution by the volume in liters we find the number of moles of \(MnO_4^−\)
\[ 15.4\; \cancel{mL}\left ( \dfrac{1 \: \cancel{L}}{1000\; \cancel{mL}} \right )\left ( \dfrac{0.0247\; mol\; MnO_{4}^{-}}{1\; \cancel{L}} \right )=3.80\times 10^{4\;} mol\; MnO_{4}^{-} \nonumber \]
The number of moles of oxalic acid, and thus oxalate, present can be calculated from the mole ratio of the reactants in the balanced chemical equation. We can abbreviate the table needed to calculate the number of moles of oxalic acid in the
\[\begin{align} moles\: HO_2 CCO_2 H & = 3 .80 \times 10^{-4}\: \cancel{mol\: MnO_4^-} \left( \dfrac{5\: mol\: HO_2 CCO_2 H} {2\:\cancel{mol\: MnO_4^-}} \right) \\ &= 9 .50 \times 10^{-4}\: mol\: HO_2 CCO_2 H \end{align} \nonumber \]
C The problem asks for the percentage of calcium oxalate by mass in the original 10.0 g sample of rhubarb, so we need to know the mass of calcium oxalate that produced 9.50 × 10−4 mol of oxalic acid. Because calcium oxalate is Ca(O2CCO2), 1 mol of calcium oxalate gave 1 mol of oxalic acid in the initial acid extraction:
\[Ca(O_2CCO_2)(s) + 2H^+(aq) \rightarrow Ca^{2+}(aq) + HO_2CCO_2H(aq) \nonumber \]
The mass of calcium oxalate originally present was thus
\(\begin{align} mass\: of\: CaC_2 O_4 &= 9 .50 \times 10^{-4}\: \cancel{mol\: HO_2 CCO_2 H} \left( \dfrac{1\: \cancel{mol\: CaC_2 O_4}} {1\: \cancel{mol\: HO_2 CCO_2 H}} \right) \left( \dfrac{128 .10\: g\: CaC_2 O_4} {1\: \cancel{mol\: CaC_2 O_4}} \right) \\ &= 0 .122\: g\: CaC_2 O_4 \end{align}\)
The original sample contained 0.122 g of calcium oxalate per 10.0 g of rhubarb. The percentage of calcium oxalate by mass was thus
\[ \% CaC_2 O_4 = \dfrac{0 .122\: g} {10 .0\: g} \times 100 = 1 .22\% \nonumber \]
Because the problem asked for the percentage by mass of calcium oxalate in the original sample rather than for the concentration of oxalic acid in the extract, we do not need to know the volume of the oxalic acid solution for this calculation.
Exercise \(\PageIndex{1}\): Glutathione
Glutathione is a low-molecular-weight compound found in living cells that is produced naturally by the liver. Health-care providers give glutathione intravenously to prevent side effects of chemotherapy and to prevent kidney problems after heart bypass surgery. Its structure is as follows:
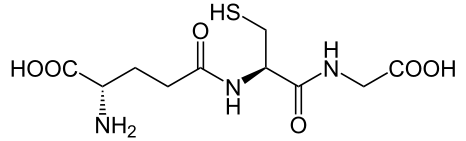
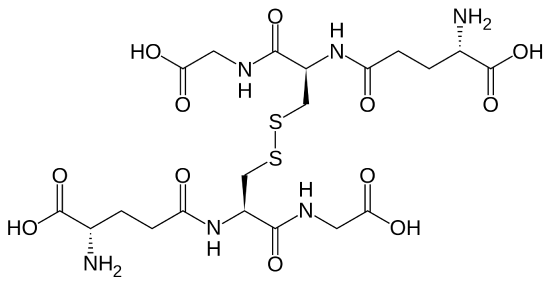
Glutathione is found in two forms: one abbreviated as (left) GSH (indicating the presence of an –SH group) and the other (right) as GSSG (the disulfide form, in which an S–S bond links two glutathione units). The GSH form is easily oxidized to GSSG with elemental iodine:
\[ \ce{2GSH(aq) + I_2(aq) \rightarrow GSSG(aq) + 2HI(aq)} \nonumber \]
A small amount of soluble starch is added as an indicator. Because starch reacts with excess I2 to give an intense blue color, the appearance of a blue color indicates that the equivalence point of the reaction has been reached.
Adding small volumes of a 0.0031 M aqueous solution of I2 to 194 mL of a solution that contains glutathione and a trace of soluble starch initially causes no change. After 16.3 mL of iodine solution have been added, however, a permanent pale blue color appears because of the formation of the starch-iodine complex. What is the concentration of glutathione in the original solution?
- Answer
-
5.2 × 10−4 M
Limiting Reactant Problems Using Molarities: Limiting Reactant Problems Using Molarities, YouTube(opens in new window) [youtu.be]
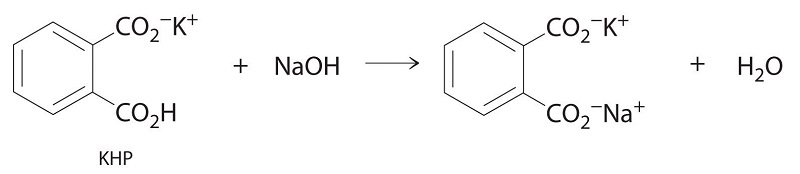
Standard Solutions
In Example \(\PageIndex{1}\), the concentration of the titrant (I2) was accurately known. The accuracy of any titration analysis depends on an accurate knowledge of the concentration of the titrant. Most titrants are first standardized; that is, their concentration is measured by titration with a standard solution, which is a solution whose concentration is known precisely. Only pure crystalline compounds that do not react with water or carbon dioxide are suitable for use in preparing a standard solution. One such compound is potassium hydrogen phthalate (KHP), a weak monoprotic acid suitable for standardizing solutions of bases such as sodium hydroxide. The reaction of KHP with NaOH is a simple acid–base reaction. If the concentration of the KHP solution is known accurately and the titration of a NaOH solution with the KHP solution is carried out carefully, then the concentration of the NaOH solution can be calculated precisely. The standardized NaOH solution can then be used to titrate a solution of an acid whose concentration is unknown.