3.4: Chemical Formulas
- Page ID
- 98535
Skills to Develop
- Symbolize the composition of molecules using molecular formulas and empirical formulas
- Represent the bonding arrangement of atoms within molecules using structural formulas
Just as we represent individual atoms and elements with symbols, we use combinations of atomic symbols to represent compounds in chemical formulas. Chemical formulas give a shorthand representation of the amounts of different elements present in a chemical compound and, to a trained eye, can also tell you something about the type of chemical bonding in the compound. Compounds are formed when two or more elements chemically combine, resulting in the formation of bonds. A chemical bond is an attraction between atoms or ions that holds them together in a molecule or an ionic crystal. For example, hydrogen and oxygen can react to form water (a molecular compound), and sodium and chlorine can react to form table salt (an ionic compound). We will discuss molecular compounds first.
Molecular Formulas
A molecule is a group of atoms bonded together in a particular arrangement that then functions as the smallest individual unit of a compound. A molecular formula is a representation of a molecule that uses chemical symbols to indicate the types of atoms followed by subscripts to show the number of atoms of each type in the molecule (Figure \(\PageIndex{1}\)). A subscript is used only when more than one atom of a given type is present. Molecular formulas are also used as abbreviations for the names of compounds.
The structural formula for a compound gives the same information as its molecular formula (the types and numbers of atoms in the molecule) but also shows how the atoms are connected in the molecule. The structural formula for methane contains symbols for one C atom and four H atoms, indicating the number of atoms in the molecule (Figure \(\PageIndex{1}\)). The lines represent the bonds that hold the atoms together. We will discuss chemical bonds and see how to predict the arrangement of atoms in a molecule later in this course. For now, simply know that the lines are an indication of how the atoms are connected in a molecule. More elaborate pictorial representations of molecules can also be constructed. A ball-and-stick model shows the geometric arrangement of the atoms in 3D space with atomic sizes not to scale, while a space-filling model shows the relative sizes of the atoms.
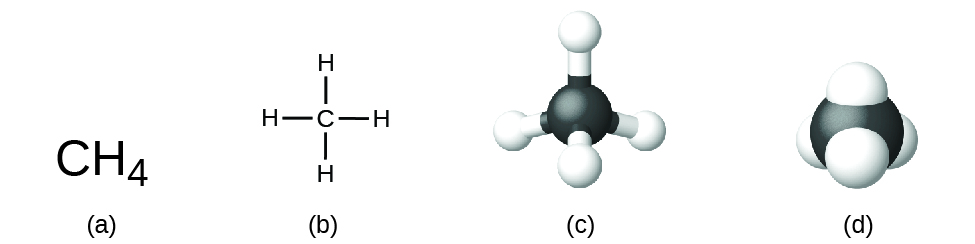
Figure \(\PageIndex{1}\): A methane molecule can be represented as (a) a molecular formula, (b) a structural formula, (c) a ball-and-stick model, and (d) a space-filling model. Carbon and hydrogen atoms are represented by black and white spheres, respectively.
Although many elements consist of discrete, individual atoms, some exist as molecules made up of two or more atoms of the same element chemically bonded together. For example, most samples of the elements hydrogen, oxygen, and nitrogen are composed of molecules that contain two atoms each (called diatomic molecules) and thus have the molecular formulas H2, O2, and N2, respectively. Other elements commonly found as diatomic molecules are fluorine (F2), chlorine (Cl2), bromine (Br2), and iodine (I2). The most common form of the element sulfur is composed of molecules that consist of eight atoms of sulfur; its molecular formula is S8 (Figure \(\PageIndex{2}\)).
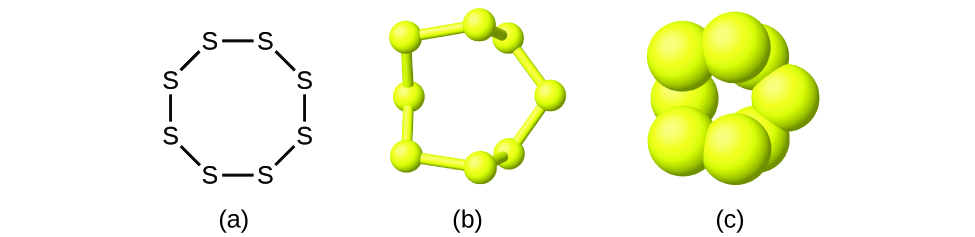
Figure \(\PageIndex{2}\): A molecule of sulfur is composed of eight sulfur atoms and is therefore written as S8. It can be represented as (a) a structural formula, (b) a ball-and-stick model, and (c) a space-filling model. Sulfur atoms are represented by yellow spheres.
It is important to note that a subscript following a symbol and a number in front of a symbol do not represent the same thing; for example, H2 and 2H represent distinctly different species. H2 is a molecular formula; it represents a diatomic molecule of hydrogen, consisting of two atoms of the element that are chemically bonded together. The expression 2H, on the other hand, indicates two separate hydrogen atoms that are not combined as a unit. The expression 2H2 represents two molecules of diatomic hydrogen (Figure \(\PageIndex{3}\)).
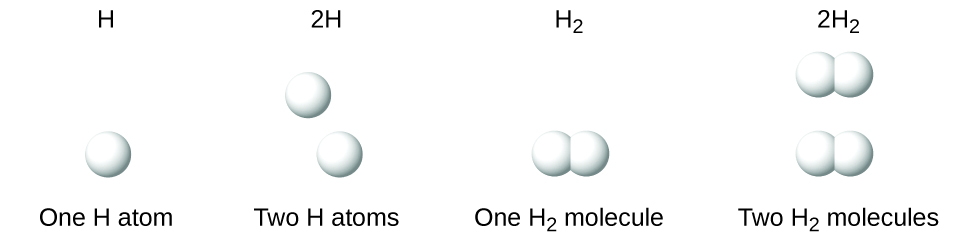
Figure \(\PageIndex{3}\): The symbols H, 2H, H2, and 2H2 represent very different entities.
Empirical Formulas
Some compounds do not form individual molecules but rather are bonded together in an extended network structure. This is especially the case with ionic compounds such as table salt, NaCl. In the case of salt, the chemical formula does not indicate that sodium and chlorine atoms pair up in individual Na-Cl molecules. Rather, the composition of this compound is represented by an empirical formula, which indicates the types of atoms present and the simplest whole-number ratio of the number of atoms (or ions) in the compound. They are called "empirical" because they can be directly determined by experiment, simply measuring the relative amounts of each element in a compound. In the case of NaCl, the empirical formula indicates that sodium and chlorine atoms are present in a 1:1 ratio. As another example, titanium dioxide (used as pigment in white paint and in the thick, white, blocking type of sunscreen) has an empirical formula of TiO2. This identifies the elements titanium (Ti) and oxygen (O) as the constituents of titanium dioxide, and indicates the presence of twice as many atoms of the element oxygen as atoms of the element titanium (Figure \(\PageIndex{4}\)).
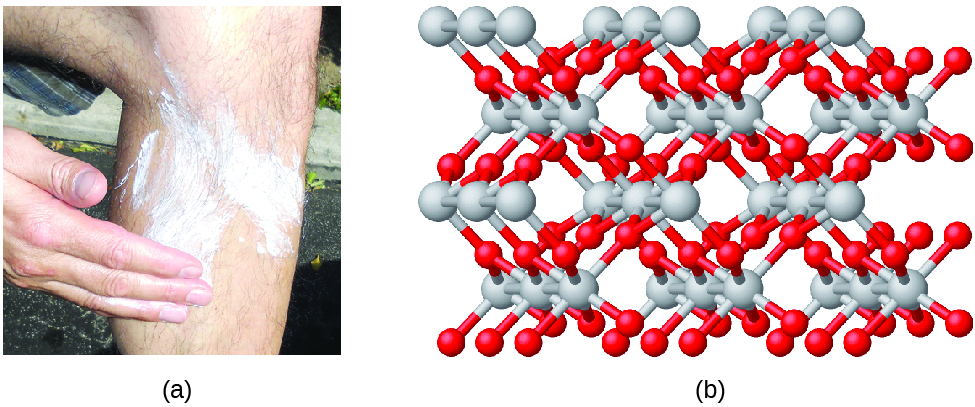
Figure \(\PageIndex{4}\): (a) The white compound titanium dioxide provides effective protection from the sun. (b) A crystal of titanium dioxide, TiO2, contains titanium and oxygen in a ratio of 1 to 2. The titanium atoms are gray and the oxygen atoms are red. (credit a: modification of work by “osseous”/Flickr).
Empirical formulas can also be derived for molecular compounds, where the subscripts would represent the simplest ratio of elements in the compound rather than the actual numbers of atoms of each element in a molecule of the compound (the molecular formula). The empirical and molecular formulas may be identical or different. In many cases, the molecular formula of a substance is derived from experimental determination of both its empirical formula and its molecular mass (the sum of atomic masses for all atoms composing the molecule). For example, it can be determined experimentally that benzene contains two elements, carbon (C) and hydrogen (H), and that for every carbon atom in benzene, there is one hydrogen atom. Thus, the empirical formula is CH. An experimental determination of the molecular mass reveals that a molecule of benzene contains six carbon atoms and six hydrogen atoms, so the molecular formula for benzene is C6H6 (Figure \(\PageIndex{5}\)).
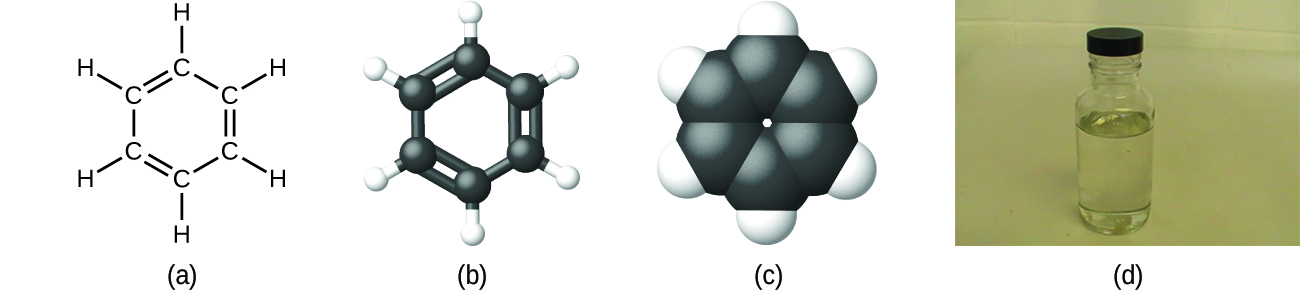
Figure \(\PageIndex{5}\): Benzene, C6H6, is produced during oil refining and has many industrial uses. A benzene molecule can be represented as (a) a structural formula, (b) a ball-and-stick model, and (c) a space-filling model. (d) Benzene is a clear liquid. (credit d: modification of work by Sahar Atwa).
If we know a compound’s formula, we can easily determine the empirical formula. (This is somewhat of an academic exercise; the reverse chronology is generally followed in actual practice.) For example, the molecular formula for acetic acid, the component that gives vinegar its sharp taste, is C2H4O2. This formula indicates that a molecule of acetic acid (Figure \(\PageIndex{6}\)) contains two carbon atoms, four hydrogen atoms, and two oxygen atoms. The ratio of atoms is 2:4:2. Dividing by the lowest common denominator (2) gives the simplest, whole-number ratio of atoms, 1:2:1, so the empirical formula is CH2O. Note that a molecular formula is always a whole-number multiple of an empirical formula.
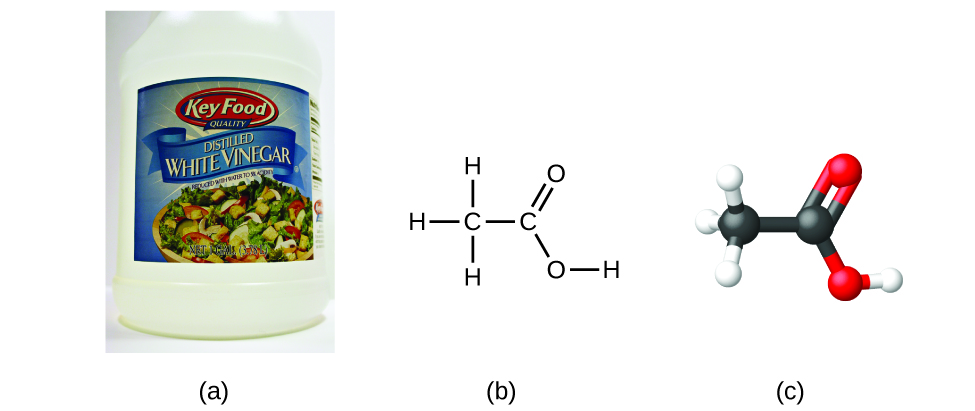
Figure \(\PageIndex{6}\): (a) Vinegar contains acetic acid, C2H4O2, which has an empirical formula of CH2O. It can be represented as (b) a structural formula and (c) as a ball-and-stick model. (credit a: modification of work by “HomeSpot HQ”/Flickr)
Example \(\PageIndex{1}\): Empirical and Molecular Formulas
Molecules of glucose (blood sugar) contain 6 carbon atoms, 12 hydrogen atoms, and 6 oxygen atoms. What are the molecular and empirical formulas of glucose?
Solution
The molecular formula is C6H12O6 because one molecule actually contains 6 C, 12 H, and 6 O atoms. The simplest whole-number ratio of C to H to O atoms in glucose is 1:2:1, so the empirical formula is CH2O.
Exercise \(\PageIndex{1}\)
A molecule of metaldehyde (a pesticide used for snails and slugs) contains 8 carbon atoms, 16 hydrogen atoms, and 4 oxygen atoms. What are the molecular and empirical formulas of metaldehyde?
- Answer
-
Molecular formula, C8H16O4; empirical formula, C2H4O
Isomers
It is important to be aware that it may be possible for the same atoms to be arranged in different ways: Compounds with the same molecular formula may have different atom-to-atom bonding and therefore different structures. For example, could there be another compound with the same formula as acetic acid, C2H4O2? And if so, what could be the structure of its molecules?
If you predicted that another compound with the formula C2H4O2 could exist, then you demonstrated good chemical insight and are correct. Two C atoms, four H atoms, and two O atoms can also be arranged to form methyl formate, which is used in manufacturing, as an insecticide, and for quick-drying finishes. Methyl formate molecules have one of the oxygen atoms between the two carbon atoms, differing from the arrangement in acetic acid molecules. Acetic acid and methyl formate are examples of isomers—compounds with the same chemical formula but different molecular structures (Figure \(\PageIndex{7}\)). Note that this small difference in the arrangement of the atoms has a major effect on their respective chemical properties. You would certainly not want to use a solution of methyl formate as a substitute for a solution of acetic acid (vinegar) when you make salad dressing!
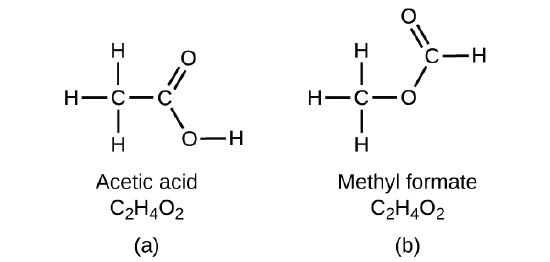
Figure \(\PageIndex{7}\): Molecules of (a) acetic acid and methyl formate (b) are structural isomers; they have the same formula (C2H4O2) but different structures (and therefore different chemical properties).
Many types of isomers exist (Figure \(\PageIndex{8}\)). Acetic acid and methyl formate are structural isomers, compounds in which the molecules differ in how the atoms are connected to each other. There are also various types of spatial isomers, in which the relative orientations of the atoms in space can be different. For example, the compound carvone (found in caraway seeds, spearmint, and mandarin orange peels) consists of two isomers that are mirror images of each other. S-(+)-carvone smells like caraway, and R-(−)-carvone smells like spearmint.
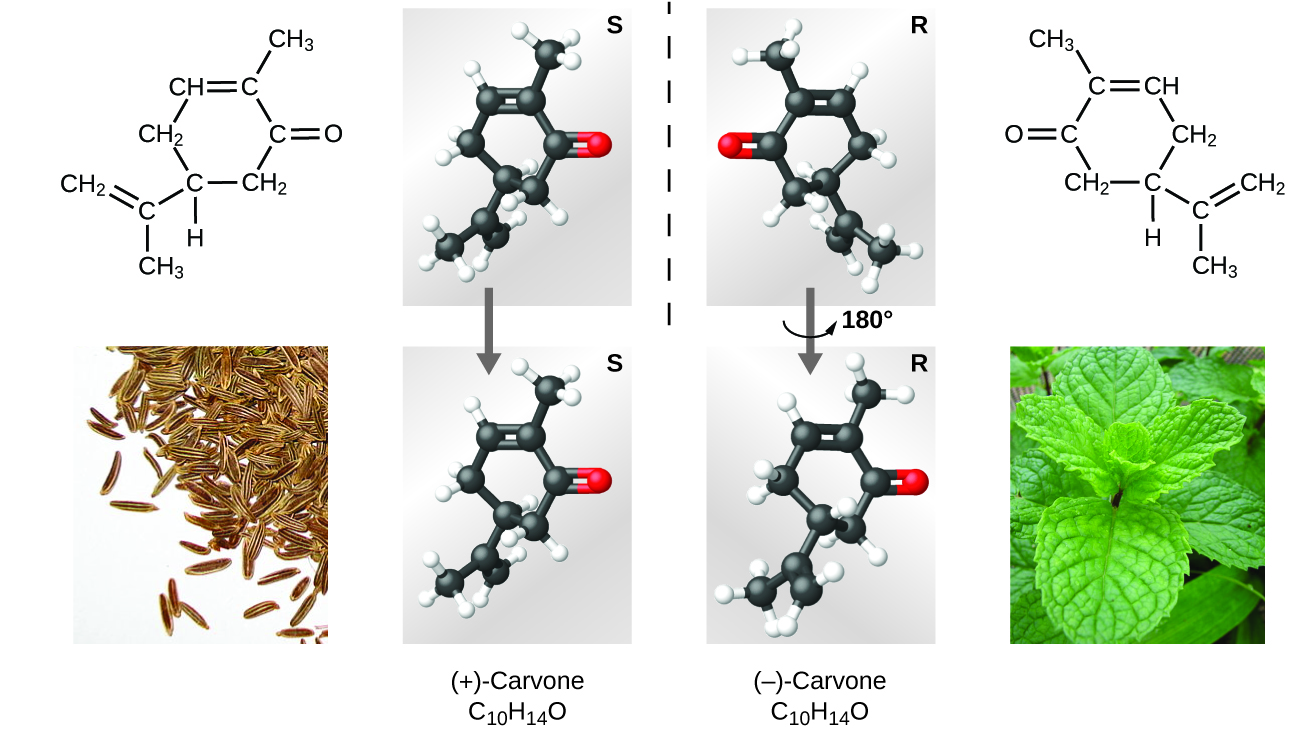
Figure \(\PageIndex{8}\): Molecules of carvone are spatial isomers; they only differ in the relative orientations of the atoms in space. (credit bottom left: modification of work by “Miansari66”/Wikimedia Commons; credit bottom right: modification of work by Forest & Kim Starr)
Summary
A molecular formula uses chemical symbols and subscripts to indicate the exact numbers of different atoms in a molecule or compound. An empirical formula gives the simplest, whole-number ratio of atoms in a compound. A structural formula indicates the bonding arrangement of the atoms in the molecule. Ball-and-stick and space-filling models show the geometric arrangement of atoms in a molecule. Isomers are compounds with the same molecular formula but different arrangements of atoms.
Contributors
Paul Flowers (University of North Carolina - Pembroke), Klaus Theopold (University of Delaware) and Richard Langley (Stephen F. Austin State University) with contributing authors. Textbook content produced by OpenStax College is licensed under a Creative Commons Attribution License 4.0 license. Download for free at http://cnx.org/contents/85abf193-2bd...a7ac8df6@9.110).
- Anna Christianson (Bellarmine University)

