4.7: The Mole
- Page ID
- 525764
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Describe the unit mole.
When objects are small or come in larger quantities, it is often inconvenient, inefficient, or even impossible to deal with the objects one at a time. For these reasons, we often deal with small objects or large quantities in groups, and have even invented names for various numbers of objects. The most common of these is "dozen" which refers to 12 objects. We frequently buy objects in groups of 12, like donuts or pencils. Other items such as bottle rockets – a type of fireworks – are sold in packages of 144, which is called a "gross", another name for a dozen dozen (122 = 144).

Avogadro's Number and the Mole
Chemists use a quantity called the mole (abbreviated as mol) to count items, it is the base SI unit for the amount of items. Just as a dozen implies 12 things, a mole of any item contains 602,200,000,000,000,000,000,000, or 6.022 × 1023, of those items. When writing numbers in scientific notation, or when seeing a bunch of zeroes at the end of a number, it is easy to lose the context of a number's sheer size. 6.022 × 1023 is also called Avogadro’s number, after the 19th-century Italian scientist who first proposed it.
For example, one mole of eggs (6.022×1023 eggs), the kind purchased at a grocery store, would be enough to cover the entire planet 60 miles deep with eggs!
Notice that we are applying the mole unit to different types of chemical entities. The word mole represents a number of things—6.022 × 1023 of them—but does not by itself specify what “they” are. The chemical entities can be atoms, molecules, formula units, or ions. This specific information needs to be specified accurately. Most students find this confusing hence, we need to review the composition of elements, covalent and ionic compounds. Figure \(\PageIndex{2}\) summarizes the basic units of elements, molecules, and ionic compounds.
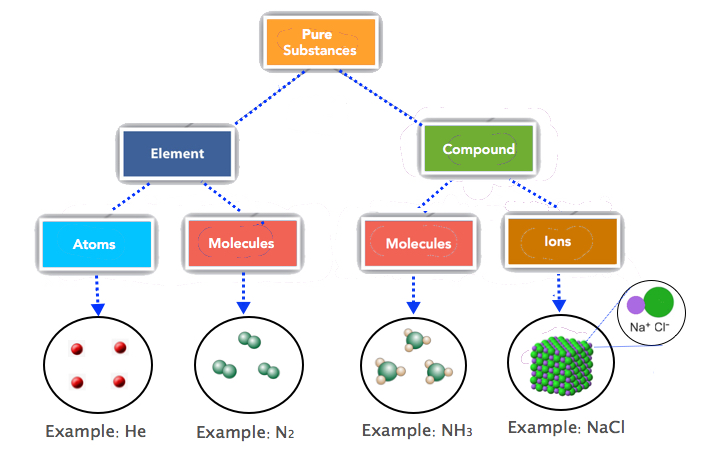
Counting Chemical Entities
|
Most elements are made up of individual atoms, such as helium. One mole of He consists of 6.022 × 1023 He atoms. |
 |
|
Some elements consist of molecules, such as the diatomic elements, nitrogen (N2), hydrogen (H2), oxygen (O2), etc. So, one mole of nitrogen contains 6.022 × 1023 N2 molecules. As each molecule is made of 2 atoms, 1 mole of nitrogen (N2) contains 2 moles of nitrogen atoms. |
 |
|
Ionic compounds, like NaCl, are represented by ionic formulas. One mole of NaCl, for example, refers to 6.022 × 1023 formula units of NaCl. And, one formula unit of NaCl consists of one sodium ion and one chloride ion. For every mole of Sodium Chloride, there is one mole of Sodium ion (6.022 × 1023 ions of Na+) and one mole of Chloride ion (6.022 × 1023 ions of Cl-). |
 |
Conversion between Moles and Atoms, Molecules, and Ions
Using the unit conversion techniques learned in Chapter 2, we can apply the mole relationship and chemical formulas to convert between moles and the number of chemical entities (atoms, molecules, or ions).
We can use Avogadro's number as a conversion factor, or ratio, in dimensional analysis problems. For example, if we are dealing with element X, the mole relationship is expressed as follows:
\[\text{1 mol X} = 6.022 \times 10^{23} \text{ X atoms} \nonumber \]
We can convert this relationship into two possible conversion factors shown below:
\(\mathrm{\dfrac{1\: mol\: X\: }{6.022\times 10^{23}\: X\: atoms}}\) or \(\mathrm{\dfrac{6.022\times 10^{23}\: X\: atoms}{1\: mol\: X\: }}\)
If the number of "atoms of element X" is given, we can convert it into "moles of X" by multiplying the given value with the conversion factor at the left. However, if the number of "mol of X" is given, the appropriate conversion factor to use is the one at the right.
If we are dealing with a molecular compound (such as C4H10), the mole relationship is expressed as follows:
\[\text{1 mol C4H10} = 6.022 \times 10^{23} \text{ C4H10 molecules} \nonumber \]
If working with ionic compounds (such as NaCl), the mole relationship is expressed as follows:
\[\text{1 mol NaCl} = 6.022 \times 10^{23} \text{ NaCl formula units} \nonumber \]
- How many formula units are present in 2.34 mol of NaCl?
- How many Na+ ions are in 2.34 mol of NaCl?
- How many Cl- ions are in 2.34 mol of NaCl?
- How Many (total) ions are in 2.34 mol of NaCl?
Solution
Typically, in a problem like this, we start with what we are given and apply the appropriate conversion factor. Here, we are given a quantity of 2.34 mol of NaCl, to which we can apply the definition of a mole as a conversion factor:

1. Formula units present in 2.34 mol of NaCl
\(\mathrm{2.34\: mol\: NaCl\times\dfrac{6.022\times10^{23}\: NaCl\: units}{1\: mol\: NaCl}=1.41\times10^{24}\: NaCl\: units}\)
2. Na+ ions present in 2.34 mol of NaCl
\(\mathrm{2.34\: mol\: NaCl\times\dfrac{1\: mol\: Na^{+}\: ions}{1\: mol\: NaCl}\times\dfrac{6.022\times10^{23}\: Na^{+}\: ions}{1\: mol\: Na^{+}\: ions}=1.41\times10^{24}\: Na^{+}\: ions}\)
3. Cl- ions present in 2.34 mol of NaCl
\(\mathrm{2.34\: mol\: NaCl\times\dfrac{1\: mol\: Cl^{-}\: ions}{1\: mol\: NaCl}\times\dfrac{6.022\times10^{23}\: Cl^{-}\: ions}{1\: mol\: Cl^{-}\: ions}=1.41\times10^{24}\: Cl^{-}\: ions}\)
4. Because there are two ions per formula unit, there are
\(\mathrm{1.41\times10^{24}\: NaCl\: units\times\dfrac{2\: ions}{NaCl\: units}=2.82\times10^{24}\: ions}\) in 2.34 mol of NaCl
How many molecules are present in 2.76 mol of H2O? How many atoms is this?
Solution
The definition of a mole is an equality that can be used to construct a conversion factor. Additionally, since we know that there are three atoms in each molecule of H2O, we can also determine the total number of atoms in the sample.
\[2.76\, \cancel{mol\, H_{2}O}\times \frac{6.022\times 10^{23}molecules\, H_{2}O}{\cancel{mol\, H_{2}O}}=1.66\times 10^{24}molecules\, H_{2}O \nonumber\nonumber \]
To determine the total number of atoms, the ratio 3 atoms per molecule should be used as conversion factor:
\[1.66\times 10^{24}\cancel{molecules\, H_{2}O}\times \frac{3\, atoms}{1\, molecule}=4.99\times 10^{24}\, atoms \nonumber\nonumber \]
If a sample consists of 2.5 mol of ethanol (C2H6O), how many moles of carbon atoms, hydrogen atoms, and oxygen atoms does it have?
Solution
Using formulas to indicate how many atoms of each element we have in a substance, we can relate the number of moles of molecules to the number of moles of atoms. For example, in 1 mol of ethanol (C2H6O), we can construct the following relationships (Table \(\PageIndex{1}\)):
| 1 Molecule of \(C_2H_6O\) Has | 1 Mol of \(C_2H_6O\) Has | Molecular Relationships |
|---|---|---|
| 2 C atoms | 2 mol of C atoms | \(\mathrm{\dfrac{2\: mol\: C\: atoms}{1\: mol\: C_2H_6O\: molecules}}\) or \(\mathrm{\dfrac{1\: mol\: C_2H_6O\: molecules}{2\: mol\: C\: atoms}}\) |
| 6 H atoms | 6 mol of H atoms | \(\mathrm{\dfrac{6\: mol\: H\: atoms}{1\: mol\: C_2H_6O\: molecules}}\) or \(\mathrm{\dfrac{1\: mol\: C_2H_6O\: molecules}{6\: mol\: H\: atoms}}\) |
| 1 O atom | 1 mol of O atoms | \(\mathrm{\dfrac{1\: mol\: O\: atoms}{1\: mol\: C_2H_6O\: molecules}}\) or \(\mathrm{\dfrac{1\: mol\: C_2H_6O\: molecules}{1\: mol\: O\: atoms}}\) |
Using the relationships in Table \(\PageIndex{1}\), we apply the appropriate conversion factor for each element:
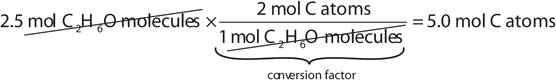
Note how the unit mol C2H6O molecules cancels algebraically. Similar equations can be constructed for determining the number of H and O atoms:
\(\mathrm{2.5\: mol\: C_2H_6O\: molecules\times\dfrac{6\: mol\: H\: atoms}{1\: mol\: C_2H_6O\: molecules}=15\: mol\: H\: atoms}\)
\(\mathrm{2.5\: mol\: C_2H_6O\: molecules\times\dfrac{1\: mol\: O\: atoms}{1\: mol\: C_2H_6O\: molecules}=2.5\: mol\: O\: atoms}\)
If a sample contains 6.75 mol of Na2SO4:
a. How many moles of sodium ions, sulfur atoms, and oxygen atoms does it have?
b. How many sodium ions, sulfur atoms, and oxygen atoms does it have? (Note: that it is asking for ions, not moles of ions)
- Answer
-
a. 13.5 mol Na+, 6.75 mol S, and 27 mol O.
b. 13.5 mol Na+ x \(\mathrm{\dfrac{6.022\times10^{23}\: Na^{+}\: ions}{1\: mol\: Na^{+}\: ions}}\)= [use the calculator], 6.75 mol S x \(\mathrm{\dfrac{6.022\times10^{23}\: atoms}{1\: mol\: atoms}}\)= [use the calculator], and 27 mol O x \(\mathrm{\dfrac{6.022\times10^{23}\: atoms}{1\: mol\: atoms}}\)= [use the calculator].
a. How many molecules are present in 16.02 mol of C4H10?
b. How many atoms are in 16.02 mol?
- Answer
-
a. 9.647 x 1024 molecules. b. Consider that there are 14 atoms per molecule = 1.351 x 1026 atoms.
How many molecules are present in 4.61 × 10−2 mol of \(\ce{O2}\)?
- Answer
-
2.78 × 1022 molecules
Key Takeaway
- A mole is 6.022 × 1023 things.


