9.7: General Features—Reactions of Alcohols, Ethers, and Epoxides
- Page ID
- 28201
Alcohols as a leaving group
Despite this promising background evidence, alcohols do not undergo the same SN2 reactions commonly observed with alkyl halides. For example, the rapid SN2 reaction of 1-bromobutane with sodium cyanide, shown below, has no parallel when 1-butanol is treated with sodium cyanide. In fact, ethyl alcohol is often used as a solvent for alkyl halide substitution reactions such as this.
CH3CH2CH2CH2–Br + Na(+) CN(–)  CH3CH2CH2CH2–CN + Na(+) Br(–) CH3CH2CH2CH2–CN + Na(+) Br(–) |
CH3CH2CH2CH2–OH + Na(+) CN(–)  No Reaction No Reaction |
The key factor here is the stability of the leaving anion (bromide vs. hydroxide). HBr is a much stronger acid than water (by more than 18 orders of magnitude), and this difference is reflected in reactions that generate their respective conjugate bases. The weaker base, bromide, is more stable, and its release in a substitution or elimination reaction is much more favorable than that of hydroxide ion, a stronger and less stable base.
A clear step toward improving the reactivity of alcohols in SN2 reactions would be to modify the –OH functional group in a way that improves its stability as a leaving anion. One such modification is to conduct the substitution reaction in strong acid, converting –OH to –OH2(+). Because the hydronium ion (H3O(+)) is a much stronger acid than water, its conjugate base (H2O) is a better leaving group than hydroxide ion. The only problem with this strategy is that many nucleophiles, including cyanide, are deactivated by protonation in strong acid, effectively removing the nucleophilic co-reactant needed for the substitution. The strong acids HCl, HBr and HI are not subject to this difficulty because their conjugate bases are good nucleophiles and are even weaker bases than alcohols. The following equations illustrate some substitution reactions of alcohols that may be effected by these acids. As with alkyl halides, the nucleophilic substitution of 1º-alcohols proceeds by an SN2 mechanism, whereas 3º-alcohols react by an SN1 mechanism. Reactions of 2º-alcohols may occur by both mechanisms and often produce some rearranged products. The numbers in parentheses next to the mineral acid formulas represent the weight percentage of a concentrated aqueous solution, the form in which these acids are normally used.
CH3CH2CH2CH2–OH + HBr (48%)  CH3CH2CH2CH2–OH2(+) Br(–) CH3CH2CH2CH2–OH2(+) Br(–)  CH3CH2CH2CH2–Br + H2O SN2 CH3CH2CH2CH2–Br + H2O SN2 |
(CH3)3C–OH + HCl (37%)  (CH3)3C–OH2(+) Cl(–) (CH3)3C–OH2(+) Cl(–)  (CH3)3C(+) Cl(–) + H2O (CH3)3C(+) Cl(–) + H2O  (CH3)3C–Cl + H2O SN1 (CH3)3C–Cl + H2O SN1 |
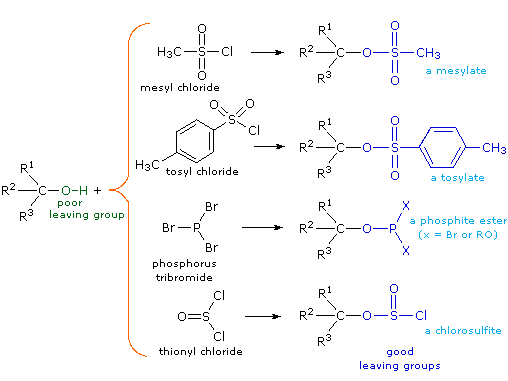
Although these reactions are sometimes referred to as "acid-catalyzed," this is not strictly correct. In the overall transformation a strong HX acid is converted to water, a very weak acid, so at least a stoichiometric quantity of HX is required for a complete conversion of alcohol to alkyl halide. The necessity of using equivalent quantities of very strong acids in this reaction limits its usefulness to simple alcohols of the type shown above. Alcohols with acid sensitive groups do not, of course, tolerate such treatment. Nevertheless, the idea of modifying the -OH functional group to improve its stability as a leaving anion can be pursued in other directions. The following diagram shows some modifications that have proven effective. In each case the hydroxyl group is converted to an ester of a strong acid. The first two examples show the sulfonate esters described earlier. The third and fourth examples show the formation of a phosphite ester (X represents remaining bromines or additional alcohol substituents) and a chlorosulfite ester respectively. All of these leaving groups (colored blue) have conjugate acids that are much stronger than water (by 13 to 16 powers of ten) so the leaving anion is correspondingly more stable than hydroxide ion. The mesylate and tosylate compounds are particularly useful in that they may be used in substitution reactions with a wide variety of nucleophiles. The intermediates produced in reactions of alcohols with phosphorus tribromide and thionyl chloride (last two examples) are seldom isolated, and these reactions continue on to alkyl bromide and chloride products.
Epoxides as a "Leaving Group"
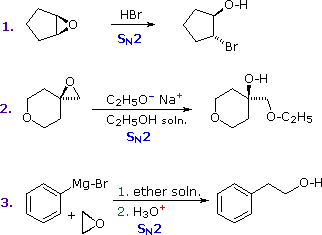
Epoxides (oxiranes) are three-membered cyclic ethers that are easily prepared from alkenes by reaction with peracids. Because of the large angle strain in this small ring, epoxides undergo acid and base-catalyzed C–O bond cleavage more easily than do larger ring ethers. Among the following examples, the first is unexceptional except for the fact that it occurs under milder conditions and more rapidly than other ether cleavages. The second and third examples clearly show the exceptional reactivity of epoxides, since unstrained ethers present in the same reactant or as solvent do not react. The aqueous acid used to work up the third reaction, following the Grignard reagent cleavage of the ethylene oxide, simply neutralizes the magnesium salt of the alcohol product.
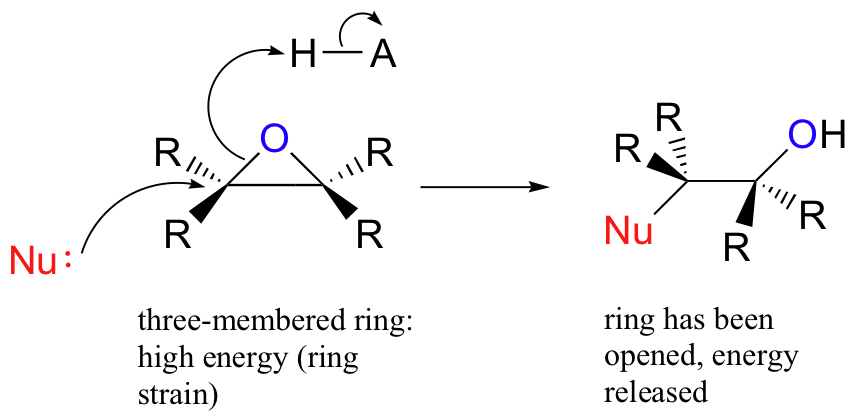
The carbons in an epoxide group are very reactive electrophiles, due in large part to the fact that substantial ring strain is relieved when the ring opens upon nucleophilic attack.
Examples
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry
-
Layne A. Morsch (University of Illinois Springfield)


