5.3: Ionizing Radiation and Non-ionizing Radiation
- Page ID
- 466601
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Qualitatively compare the ionizing and penetration power of alpha particles (α), beta particles (β), and gamma rays (γ).
- Describe the biological impact of ionizing radiation.
- Know the most common source of background radiation and how to minimize exposure to this source.
The Ionizing and Penetration Power of Radiation
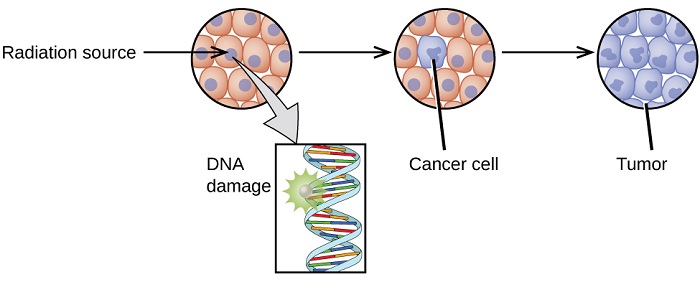
The increased use of radioisotopes has led to increased concerns over the effects of these materials on biological systems (such as humans). All radioactive nuclides emit high-energy particles or electromagnetic waves. When this radiation encounters living cells, it can cause increase heat, break chemical bonds, or ionize molecules. The most serious biological damage results when these radioactive emissions fragment or ionize molecules. For example, alpha and beta particles emitted from nuclear decay reactions possess much higher energies than ordinary chemical bond energies. When these particles strike and penetrate matter, they produce ions and molecular fragments that are extremely reactive. The damage this does to biomolecules in living organisms can cause serious malfunctions in normal cell processes, taxing the organism’s repair mechanisms and possibly causing illness or even death (Figure \(\PageIndex{1}\)).
The ability of radiation to damage molecules is analyzed in terms of ionizing power. When a radiation particle interacts with atoms, the interaction can cause the atom to lose electrons and thus become ionized. The greater the likelihood that damage will occur by an interaction means the radiation has greater ionizing power. Ionizing radiation could affect either the whole body (somatic damage) and/or eggs and sperm (genetic damage).
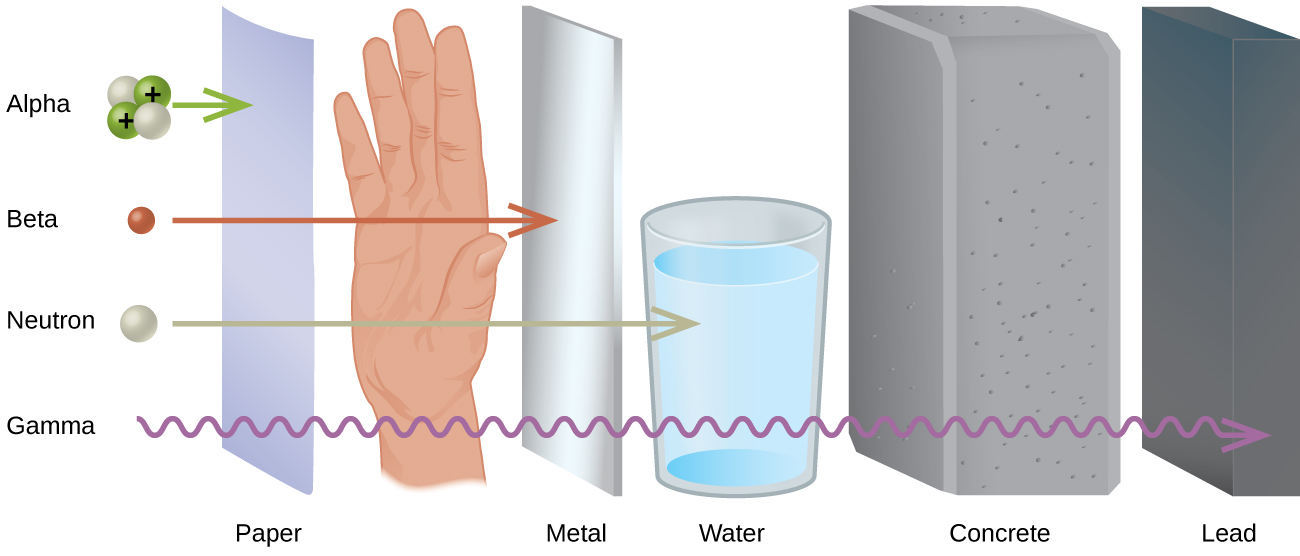
Much of the threat from radiation relates to the ability to protect oneself from the emitted radiation. The ability of each type of radiation to pass through matter is expressed in terms of penetration power. The more material the radiation can pass through and the easier the radiation passes through, the greater the penetration power and the more dangerous they are. In general, the greater the mass of the particle, the greater the ionizing power but the lower the penetration power.
When comparing the most common forms of ionizing radiation (alpha, beta, and gamma), alpha particles have the greatest mass. Alpha particles have approximately four times the mass of a proton or neutron and approximately ~8,000 times the mass of a beta particle (Figure \(\PageIndex{1}\)). Because of the large mass of the alpha particle, it has the highest ionizing power and the greatest ability to damage tissue. That same large size of alpha particles, however, makes them less able to penetrate matter. They collide with molecules very quickly when striking matter. This collision often results in the addition of two electrons, causing the alpha particle to become a harmless helium atom. Due to their relatively heavy mass, alpha particles have the least penetration power and can be stopped by a thick sheet of paper or even a layer of clothes. They are also stopped by the outer layer of dead skin on people. This may seem to remove the threat from alpha particles but only from external sources. In a situation like a nuclear explosion or a nuclear accident where radioactive emitters are spread around in the environment, the emitters can be inhaled or taken in with food or water. If the alpha emitter enters the body, you no longer have protection from the particles and bodily harm can result.
| Particle | Symbol | Mass | Penetrating Power | Ionizing Power | Shielding |
|---|---|---|---|---|---|
| alpha | \(\alpha\) | 4 amu | Very Low | Very High | Paper and Skin |
| beta | \(\beta\) | 1/1837 amu | Intermediate | Intermediate | Aluminum |
| gamma | \(\gamma\) | 0 (energy only) | Very High | Very Low | 2 inches lead |
Beta particles are much smaller than alpha particles, and therefore, they have much less ionizing power (less ability to damage tissue), but their small size gives them much greater penetration power. Most resources say that beta particles can be stopped by a one-quarter inch thick sheet of aluminum. Once again, however, the greatest danger occurs when the beta-emitting source gets inside your body.
Gamma rays are not particles, but a high energy form of electromagnetic radiation (like X-rays, except more powerful). Gamma rays are energy with no mass or charge. Gamma rays have tremendous penetration power and require several inches of dense material (like lead) to shield them. It is possible for gamma rays to pass completely through a human body without striking anything. They are considered to have the least ionizing power but the greatest penetration power.
ADAPT \(\PageIndex{1}\)
ADAPT \(\PageIndex{2}\)
ADAPT \(\PageIndex{3}\)
ADAPT \(\PageIndex{4}\)
The safest amount of radiation to the human body is zero. Unfortunately, it is not possible to completely avoid exposure to ionizing radiation. The next best goal is to minimize your exposure by limiting the length of time of your exposure and by increasing your distance from the source. The image below summarizes the key concepts of ionization and penetration abilities of alpha, beta, and gamma radiation.

ADAPT \(\PageIndex{5}\)
Nonionizing Radiation
There is a large difference in the magnitude of the biological effects of nonionizing radiation (for example, light and microwaves) and ionizing radiation, emissions energetic enough to knock electrons out of molecules, for example, \(α\) and \(β\) particles, \(γ\) rays, X-rays, and high-energy ultraviolet radiation (Figure \(\PageIndex{2}\)).
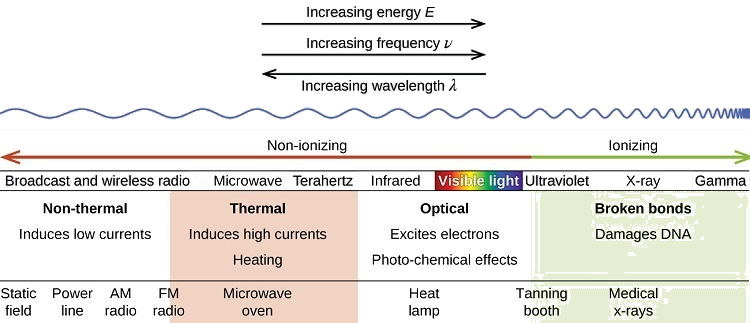
Energy absorbed from nonionizing radiation speeds up the movement of atoms and molecules, which is equivalent to heating the sample. Although biological systems are sensitive to heat (as we might know from touching a hot stove or spending a day outside on a hot day), a large amount of nonionizing radiation is tolerated before negative effects begin. Forms of nonionizing radiation include wave-like radiation shown on the left side of the image. This type of radiation would include the visible spectrum through radio waves.
Everyday Exposure to Radiation
Natural radiation provides the majority of exposure to the average person. Looking at the pie chart below, the largest sources of exposure to radiation is from radon gas (Rn-222). This isotope is an alpha emitter with a half–life of 3.82 days. Radon is produced through the radioactive decay of U-238, which is found in trace amounts in soil and rocks. In the environment, radon concentrations can vary depending on geographical location. Once the soil of a particular region is disturbed, this element can escape and cause serious health issues. Please be aware that radon, not nuclear reactors, affects more people in the United States.
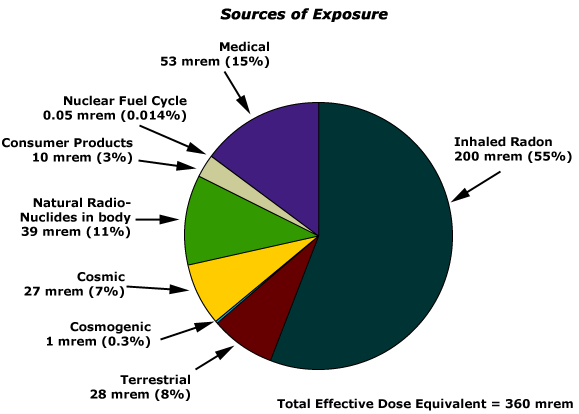
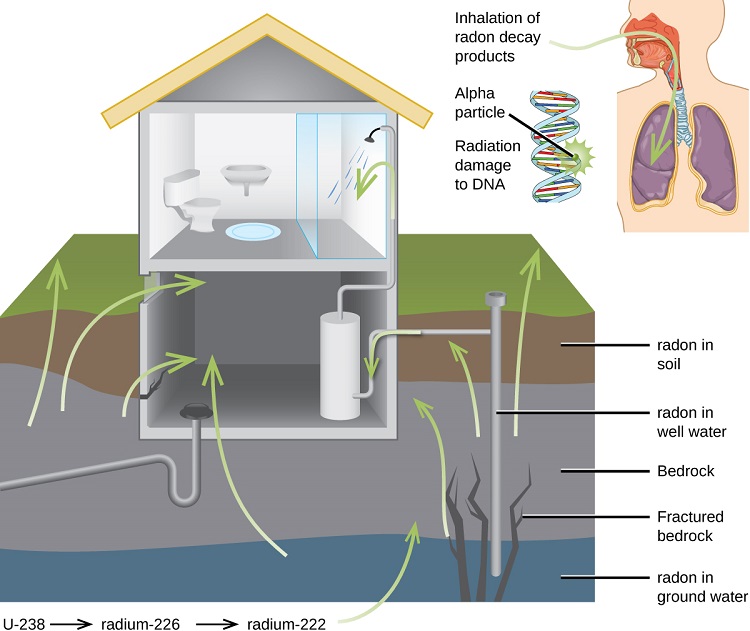
Radon gas escapes from the ground and gradually seeps into homes and other structures situated above. Since radon gas is about eight times more dense than air, it accumulates in basements and lower floors, slowly diffusing throughout buildings (Figure \(\PageIndex{5}\)). Once airborne, the radon enters the body through inhalation or ingestion. Through alpha emission, Rn-222 decays to produce large particles. These alpha particles then travel to the respiratory tract where they can ionize lung tissue. Exposure to radon increases one’s risk of getting cancer (especially lung cancer), and high radon levels can be as bad for health as smoking a carton of cigarettes a day. Radon is the number one cause of lung cancer in nonsmokers and the second leading cause of lung cancer overall. Radon exposure is believed to cause over 20,000 deaths in the US per year.
Radon is found in buildings across the country, with amounts varying depending on where you live. The average concentration of radon inside houses in the United States (1.25 pCi/L) is about three times the levels found in outside air. About one in six houses have radon levels high enough to warrant remediation efforts to reduce the radon concentration. The Environmental Protection Agency (EPA) sets regulations and monitors for radon throughout the United States. Regions are classified into three different zones using color-coded keys (Zone 1 (red): highest potential for radon; Zone 2 (orange): moderate potential for radon; Zone 3 (yellow): low potential for radon). Based on the zone for your region, an individual can assess their probability of being exposed to this deadly isotope and determine if they should have their home tested for radon. If the radon level in a home is greater than 4 pCi/L (picocuries per liter), the EPA recommends action is taken to reduce the radon in the home. Click on this link to see your state and county's radon-risk level.
Radon tests can be purchased through the EPA or at most hardware stores. If a consumer finds excessive radon in their home, then alterations to the existing construction can be made. Some of these changes include installing barriers between the soil and the home and placing ventilation on the group floor to release radon outside. For more information regarding these types of alterations, click on this link and select "how to fix your home."
Other sources of radiation in consumer products include smoke detectors, antique watches/clocks, and older ceramics/glass. Smoke detectors emit alpha particles of americium-241. Antique watches and clocks use hydrogen-3 (tritium), promethium-147, or radium-226 as a fluorescent light source. Antique ceramics (dating before 1970) could contain uranium-238 if the color of the ceramic is an orangey-red. If antique glassware is yellow or green in color, it could contain uranium-238 as well. Glassware with this isotope will glow under a black light.
Concrete, fertilizers, kitty litter, and even food (bananas and salt substitutes) can contain trace amounts of different types of radioactivity. Potassium-40 is present in all of these substances. You can estimate your yearly radiation exposure using the EPA's "Calculate Your Radiation Dose."
ADAPT \(\PageIndex{6}\)
References
Contributors and Attributions
Paul Flowers (University of North Carolina - Pembroke), Klaus Theopold (University of Delaware) and Richard Langley (Stephen F. Austin State University) with contributing authors. Textbook content produced by OpenStax College is licensed under a Creative Commons Attribution License 4.0 license. Download for free at http://cnx.org/contents/85abf193-2bd...a7ac8df6@9.110).
- Emma Gibney (Furman University)
- Erin Avram (Cleveland State University)

