12: Homework Solutions
- Page ID
- 43946
Properties of Acids and Bases
(01) Arrhenius acid: releases hydrogen ions; Bronsted-Lowry acid: hydrogen ion donor; Bronsted-Lowry base: hydrogen ion acceptor
(02)
- \(HCN + H_2O \rightleftharpoons CN^- + H_3O^+\)
- \(CO_3^{2-} + H_2O \rightleftharpoons HCO_3^- + OH^-\)
- \(NH_4^+ + H_2O \rightleftharpoons NH_3 + H_3O^+\)
- \(H_2SO_4 + H_2O \rightarrow HSO_4^- + H_3O^+\)
- \(CH_3CO_2H + H_2O \rightleftharpoons CH_3CO_2^- + H_3O^+\)
Conjugate Acids and Bases
(03) For each of the following acids, determine the form of their conjugate bases.
- SO42-
- HCO3-
- F-
- PO43-
- HCOO-
(04) For each of the following bases, determine the form of their conjugate acids.
- HBr
- HNO3
- HS-
- NH4+
- H2CO3
Strengths of Acids and Bases
(05) strong acid: substances that fully ionize in water to create hydronium ions.
\[HNO)3 + H_2O \rightarrow NO_3^- + H_3O+\]
\[H_2S + H_2O \rightleftharpoons CN^- + H_3O^+\]
(06) strong base: substance that fully ionize to produce hydroxide ions in water
\[LiOH + H_2O \rightarrow H_2O + Li^+ + OH^- \]
which simplifies to
\[LiOH \rightarrow Li^+_{(aq)} + OH^-_{(aq)}\]
(07)
H3PO4 + H2O \(\rightleftharpoons\) H2PO4- + H3O+
H2PO4- + H2O \(\rightleftharpoons\) HPO42- + H3O+
HPO42- + H2O \(\rightleftharpoons\) PO43- + H3O+
(08) For each chemical reaction, determine whether they are representative of the dissociation reaction of a strong acid, a weak acid, a strong base, or a weak base.
- strong base
- weak acid
- weak base
- weak acid
- strong acid
(09)
- HClO4 \(\rightleftharpoons\) H+ + ClO
- HSCN \(\rightleftharpoons\) H+ + SCN-
- HBr → H+ + Br-
- H2CO3 \(\rightleftharpoons\) H+ + HCO3-
- CH3COOH \(\rightleftharpoons\) H+ + CH3COO-
(10)
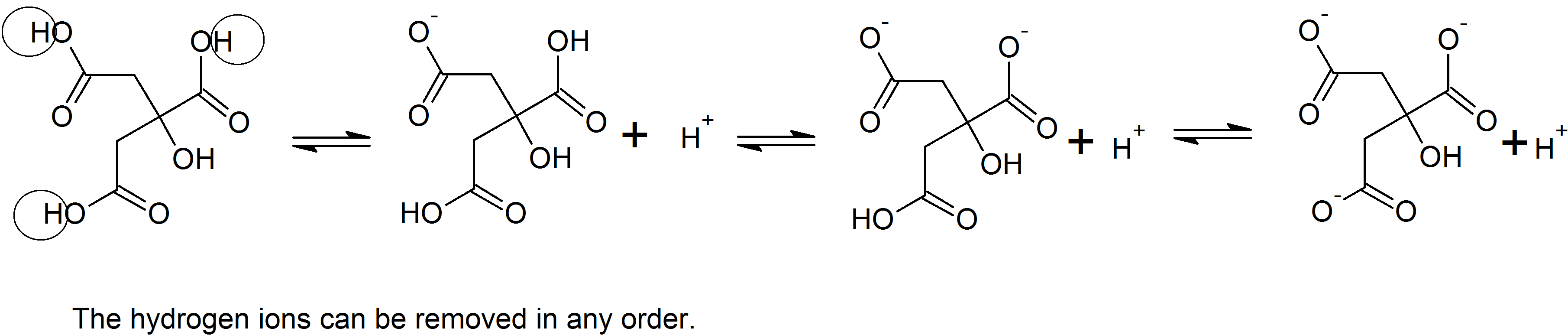
Acid-Base Equilibria
(11) equilibrium: a state in which the forward and reverse reactions are occurring at the same rate
H2O \(\rightleftharpoons\) OH- + H+
No, the concentrations of the reactants and products remain constant when a reaction is at equilibrium.
The double arrow represent the forward and reverse reactions occurring simultaneously.
Le Chatelier’s Principle
(12)
- left
- right
- right
- left
(13)
- left
- left
- left
- right
- right
(14)
- The hydrogen ion concentration increases.
- The hydrogen ion concentration decreases.
Acid-Base Neutralization Reactions
(15) Write out the neutralization reactions for the following pairs. Make sure to balance the equations as necessary and include phases.
- \(Ba(OH)_2\) + \(2HNO_3\) → Ba(NO3)2 + 2 H2O
- \(Ca(OH)_2\) + \(H_2SO_4\) → CaSO4 + 2 H2O
- \(RbOH\) + \(HI\) → RbI + H2O
- \(KOH\) + \(HClO_4\) → KClO4 + H2O
- \(Mg(OH)_2\) + 2 \(HBr\) → MgBr2 + 2 H2O
pH
(16) Examine the following pH measures and tell whether they are in the range to be considered acidic, basic or neutral.
- neutral (very slightly acidic)
- basic
- acidic
- acidic
- basic
(17) Using the formula for converting concentration into pH, determine whether each of the following molarities are acidic, basic or neutral. Remember, -log[H+]
- 7.0 neutral
- 11.3 basic
- 3.2 acidic
- 9.1 basic
- 1.0 acidic
(18) Fill in the blank portions of the table.
| \([H_3O^+]\) | \([OH^-]\) | \(pH\) | Acidic, Basic or Neutral |
| \(1.0 \times 10^{-1}\) | \(1.0 \times 10^{-13}\) | 1 | acidic |
| \(1.0 \times 10^{-10}\) | \(1.0 \times 10^{-4}\) | 10 | basic |
| \(1.0 \times 10^{-7}\) | \(1.0 \times 10^{-7}\) | 7 | neutral |
| \(1.0 \times 10^{-5}\) | \(1.0 \times 10^{-9}\) | 5 | acidic |
| \(1.0 \times 10^{-13}\) | \(1.0 \times 10^{-1}\) | 13 | basic |
(19) Given the following sets of information, calculate the requested quantities for each problem.
- pH = 3.4 and [OH-] = 2.4 x 10-11M
- pOH = 8.2 and [H3O+] = 1.5 x 10-6M
- [H3O+] = 1.0 x 10-9M, [OH-] = 1.0 x 10-5M and pOH = 5
- [OH-] = 3.2 x 10-13M and [H3O+] = 3.2 x 10-2M
- [H3O+] = 1 x 10-7M, [OH-] = 1 x 10-7M and pH = 7
Buffers
(20) Buffer (buffer system): a combination of a weak acid and weak base (often conjugate acid/base pairs) at similar concentrations used to maintain constant pH
- no
- yes
- no
- yes
- no
(21)
- HBr + HCO3- \(\rightleftharpoons\) H2CO3 + Br-
- LiOH + H2CO3 → HCO3- + H2O + Li+
- NH4+ + HCO3- \(\rightleftharpoons\) H2CO3 + NH3
- HCOO- + H2CO3 \(\rightleftharpoons\) HCOOH + HCO3-
- CH3NH2 + H2CO3 \(\rightleftharpoons\) CH3NH3+ + HCO3-
(22) CO2 levels increase in the body raising the concentration of hydrogen ions and lowering the pH.

