5.6: Intermolecular Forces
- Page ID
- 43780
The physical properties of melting point, boiling point, vapor pressure, evaporation, viscosity, surface tension, and solubility are related to the strength of attractive forces between molecules. These attractive forces are called Intermolecular Forces. The amount of "stick togetherness" is important in the interpretation of the various properties listed above.
Introduction
There are four types of intermolecular forces. Most of the intermolecular forces are identical to bonding between atoms in a single molecule. Intermolecular forces just extend the thinking to forces between molecules and follows the patterns already set by the bonding within molecules.
Ionic Forces
The forces holding ions together in ionic solids are electrostatic forces. Opposite charges attract each other. These are the strongest intermolecular forces. Ionic forces hold many ions in a crystal lattice structure.
Dipole Forces
Polar covalent molecules are sometimes described as "dipoles", meaning that the molecule has two "poles". One end (pole) of the molecule has a partial positive charge while the other end has a partial negative charge. The molecules will orientate themselves so that the opposite charges attract principle operates effectively.
For example, hydrochloric acid (\(HCl\)) is a polar molecule with the partial positive charge on the hydrogen and the partial negative charge on the chlorine. A network of partial + and - charges attract molecules to each other.

Hydrogen Bonding
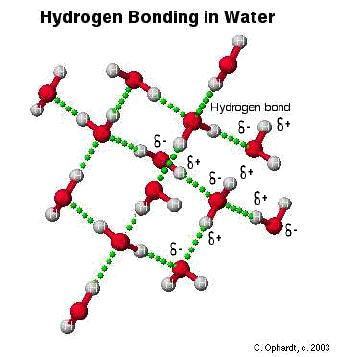
The hydrogen bond is really a special case of dipole forces. A hydrogen bond is the attractive force between the hydrogen attached to an electronegative atom of one molecule and an electronegative atom of a different molecule. Usually the electronegative atom is oxygen, nitrogen, or fluorine. In other words - The hydrogen on one molecule attached to O or N that is attracted to an O or N of a different molecule.
In the graphic below, the hydrogen is partially positive and attracted to the partially negative charge on the oxygen or nitrogen. Because oxygen has two lone pairs, two different hydrogen bonds can be made to each oxygen. This is a very specific bond as indicated. Some combinations that are not hydrogen bonds include: hydrogen to another hydrogen or hydrogen to a carbon.
London Dispersion or Induced Dipole or Van der Waals Forces
Forces between essentially non-polar molecules are the weakest of all intermolecular forces. "Temporary dipoles" are formed by the shifting of electron clouds within molecules. These temporary dipoles attract or repel the electron clouds of nearby non-polar molecules.
The temporary dipoles may exist for only a fraction of a second but a force of attraction also exist for that fraction of time. The strength of induced dipole forces depends on how easily electron clouds can be distorted. Large atoms or molecules with many electrons far removed from the nucleus are more easily distorted.

Contributors
- Charles Ophardt, Professor Emeritus, Elmhurst College
- Virtual Chembook

