7.1: Overview of acid-base reactions
- Page ID
- 21356
A: The Brønsted-Lowry definition of acidity and basicity
We’ll begin our discussion of acid-base chemistry with a couple of essential definitions. The first of these was proposed in 1923 by the Danish chemist Johannes Brønsted and the English chemist Thomas Lowry, and has come to be known as the Brønsted-Lowry definition of acidity and basicity. An acid, by the Brønsted-Lowry definition, is a species which acts as a proton donor, while a base is a proton acceptor. We have already discussed in the previous chapter one of the most familiar examples of a Brønsted-Lowry acid-base reaction, between hydrochloric acid and hydroxide ion:
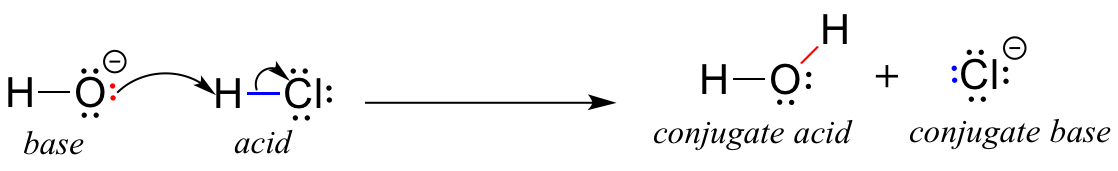
In this reaction, a proton is transferred from HCl (the acid, or proton donor) to hydroxide ion (the base, or proton acceptor). As we learned in the previous chapter, curved arrows depict the movement of electrons in this bond-breaking and bond-forming process.
After a Brønsted-Lowry acid donates a proton, what remains is called the conjugate base. Chloride ion is thus the conjugate base of hydrochloric acid. Conversely, when a Brønsted-Lowry base accepts a proton it is converted into its conjugate acid form: water is thus the conjugate acid of hydroxide ion.
Here is an organic acid-base reaction between acetic acid and methylamine:
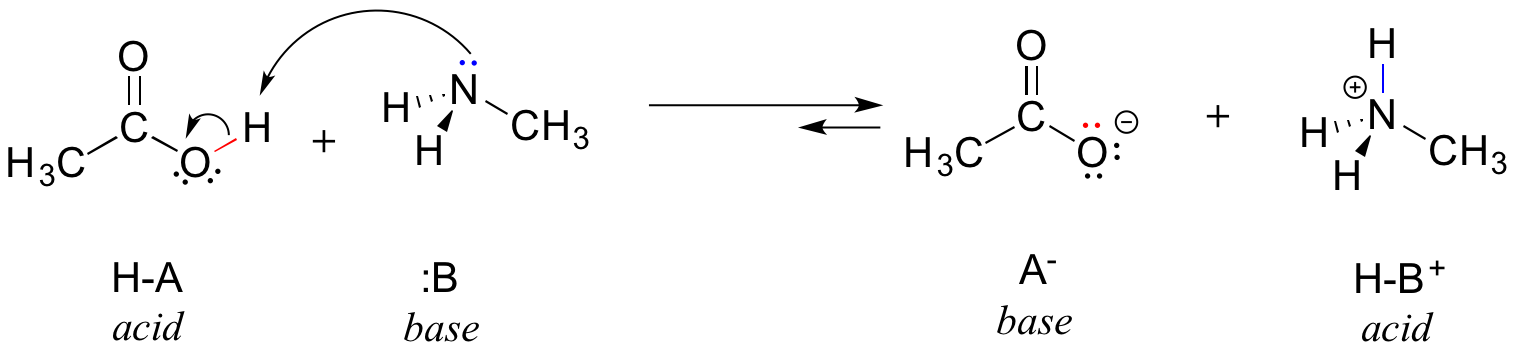
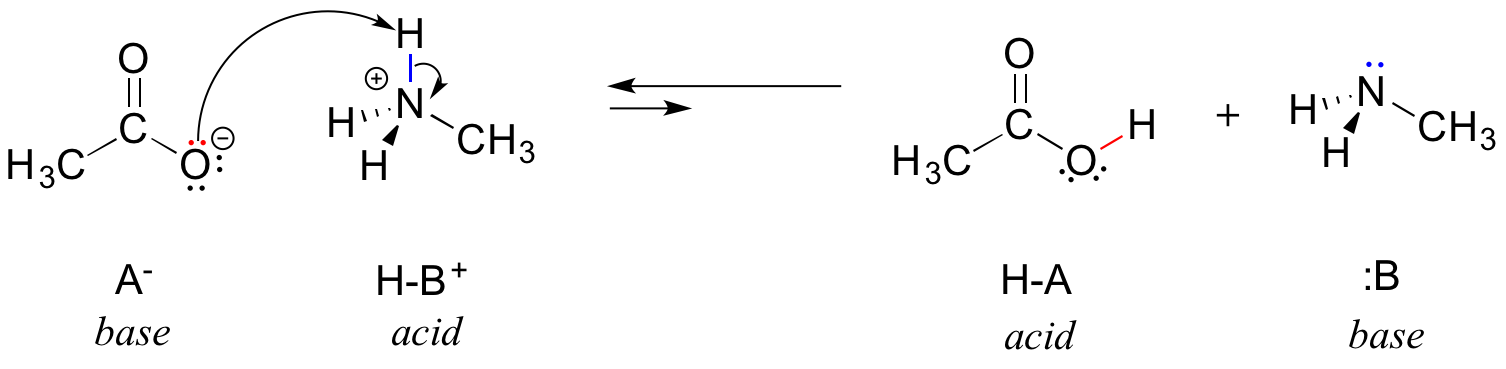
In the reverse of this reaction, acetate ion is the base and methylammonium ion (protonated methylamine) is the acid.
What makes a compound acidic (likely to donate a proton) or basic (likely to accept a proton)? Answering that question is one of our main jobs in this chapter, and will require us to put to use much of what we learned about organic structure in the first two chapters, as well as the ideas about thermodynamics that we reviewed in chapter 6.
For now, let's just consider one common property of bases: in order to act as a base, a molecule must have a reactive pair of electrons. In all of the acid-base reactions we'll see in this chapter, the basic species has an atom with a lone pair of electrons. When methylamine acts as a base, for example, the lone pair of electrons on the nitrogen atom is used to form a new bond to a proton.
Clearly, methylammonium ion cannot act as a base – it does not have a reactive pair of electrons with which to accept a proton.

Later, in chapter 14, we will study reactions in which a pair of electrons in a pi bond of an alkene or aromatic ring act in a basic fashion - but for now, will concentrate on the basicity of non-bonding (lone pair) electrons.
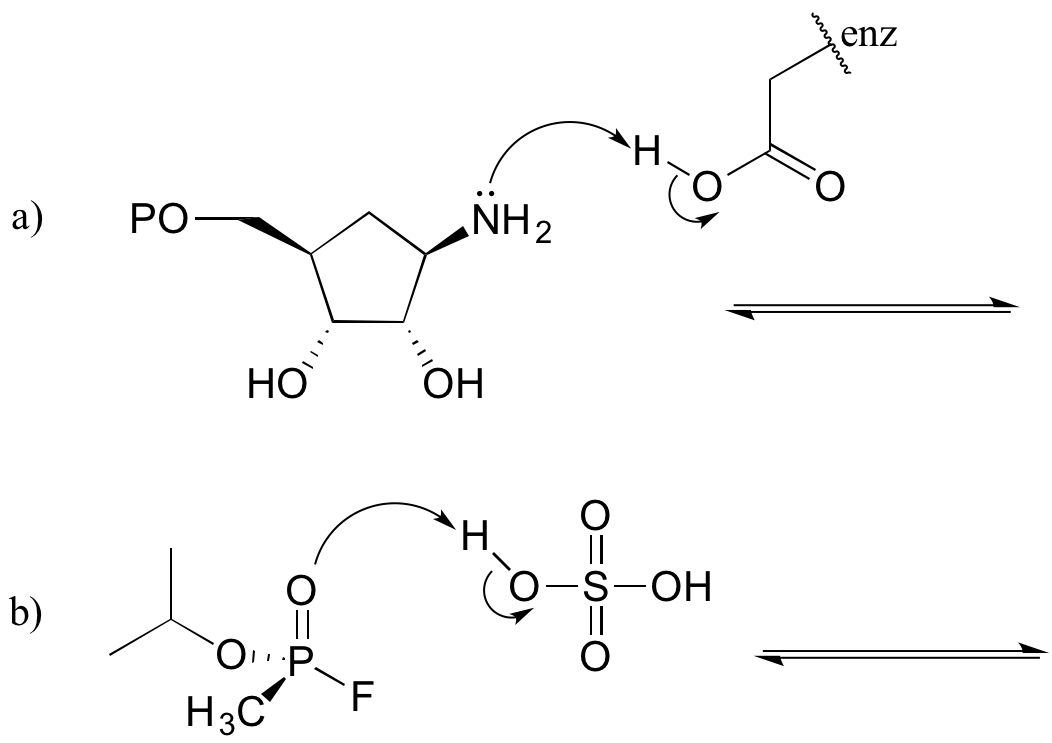
Exercise 7.1.1
Complete the reactions below - in other words, draw structures for the missing conjugate acids and conjugate bases that result from the curved arrows provided.
B: The Lewis definition of acidity and basicity
The Brønsted-Lowry picture of acids and bases as proton donors and acceptors is not the only definition in common use. A broader definition is provided by the Lewis definition of acidity and basicity, in which a Lewis acid is an electron-pair acceptor and a Lewis base is an electron-pair donor. This definition covers Brønsted-Lowry proton transfer reactions, but also includes reactions in which no proton transfer is involved. The interaction between a magnesium cation (Mg+2) and a carbonyl oxygen is a common example of a Lewis acid-base reaction in enzyme-catalyzed biological reactions. The carbonyl oxygen (the Lewis base) donates a pair of electrons to the magnesium cation (the Lewis acid).
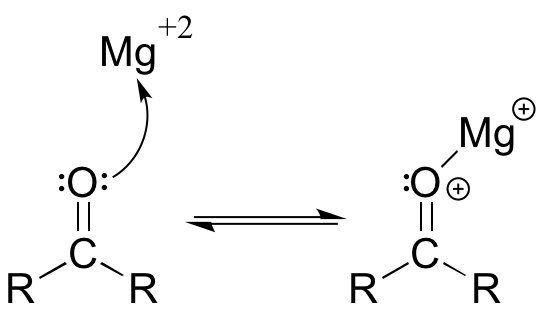
While it is important to be familiar with the Lewis definition of acidity, the focus throughout the remainder of this chapter will be on acid-base reactions of the (proton-transferring) Brønsted-Lowry type.


