Answers to Chapter 18 Study Questions
- Page ID
- 11196
-
- -3; \(\mathrm{[x + 3(1) = 0]}\)
- 0
- +4; \(\mathrm{[x +2(-2) = 0]}\)
- +5; \(\mathrm{[x + 3(-2) = -1]}\)
-
- \(\ce{Br2}\)
- \(\ce{I}\)
- \(\ce{Br}\)
- \(\ce{Br2}\)
- \(\ce{I-}\)
- First determine whether substances may be oxidized or reduced, then look up \(\mathrm{E^\circ_{ox}}\) for reducing agents and \(\mathrm{E^\circ_{red}}\) for oxidizing agents and list in order.
Oxidizing agents (are reduced): \(\ce{H+}\) (\(\mathrm{E^\circ_{red} = 0\, V}\)).
Reducing agents (are oxidized): \(\ce{Ni}\) (\(\mathrm{E^\circ_{ox} = +0.23\, V}\)), \(\ce{Au}\) (\(\mathrm{E^\circ_{ox} = -1.50\, V}\)), \(\ce{Mg}\) (\(\mathrm{E^\circ_{ox} = +2.37\, V}\)).
Both: \(\ce{Sn^2+}\) (\(\mathrm{E^\circ_{red} = -0.14\, V}\); \(\mathrm{E^\circ_{ox} = -0.15\, V}\)), \(\ce{Fe^2+}\) (\(\mathrm{E^\circ_{red} = -0.44\, V}\), \(\mathrm{E^\circ_{ox} = -0.77\, V}\)), \(\ce{Cl2}\) (\(\mathrm{E^\circ_{red} = 1.36\, V}\), \(\mathrm{E^\circ_{ox} = -1.47\, V}\)).
Oxidizing agents (list in order of decreasing \(\mathrm{E^\circ_{red}}\)): \(\mathrm{Cl_2 > H^+ > Sn^{2+} > Fe^{2+}}\)
Reducing agents (list in order of decreasing \(\mathrm{E^\circ_{ox}}\)): \(\mathrm{Mg > Ni > Sn^{2+} > Fe^{2+} > Cl_2 > Au}\)
-
- ox: \(\ce{[Ag \rightarrow Ag+ + e^- ]\times 2}\); red: \(\ce{Cu^2+ + 2 e^- \rightarrow Cu}\)
balanced: \(\ce{2 Ag(s) + Cu^2+ (aq) \rightarrow 2 Ag+ (aq) + Cu(s)}\)
\(\mathrm{E^\circ = E^\circ_{ox}(Ag) + E^\circ_{red}(Cu^{2+}) = -0.80\, V + 0.34\, V = -0.46\, V}\).
Not spontaneous. - ox: \(\ce{[Ni(s) \rightarrow Ni^2+ + 2 e^- ] \times 5}\); red: \(\ce{[5 e^- + 8 H+ + MnO_4- \rightarrow Mn^2+ + 4 H2O] \times 2}\)
balanced: \(\ce{5 Ni(s) + 16 H+ (aq) + 2 MnO_4- (aq) \rightarrow 5 Ni^2+ (aq) + 2 Mn^2+ (aq) + 8 H2O}\)
\(\mathrm{E^\circ = E^\circ_{ox}(Ni) + E^\circ_{red}(MnO_4^-) = +0.23\, V + 1.51\, V = +1.74\, V}\).
Spontaneous. - ox: \(\ce{[2 H2O + Mn^2+ \rightarrow MnO2 + 4 H+ + 2 e^- ] \times 3}\)
red: \(\ce{[3 e^- + 4 H+ + NO3- \rightarrow NO + 2 H2O] \times 2}\)
balanced: \(\ce{2 H2O + 3 Mn^2+ (aq) + 2 NO3- (aq) \rightarrow 3 MnO2(s) + 4 H+ (aq) + 2 NO(g)}\)
\(\mathrm{E^\circ = E^\circ_{ox}(Mn^{2+}) + E^\circ_{red}(NO_3^-) = -1.21\, V + 0.96\, V = -0.25\, V}\).
Not spontaneous.
- ox: \(\ce{[Ag \rightarrow Ag+ + e^- ]\times 2}\); red: \(\ce{Cu^2+ + 2 e^- \rightarrow Cu}\)
-
- \(\mathrm{E^\circ = E^\circ_{ox}(Fe) + E^\circ_{red}(Fe^{3+}) = +0.44\, V + 0.77\, V = +1.21\, V}\).
Spontaneous. - \(\mathrm{E^\circ = E^\circ_{ox}(I^-) + E^\circ_{red}(Zn^{2+}) = - 0.54\, V - 0.76\, V = -1.30\, V}\).
Not spontaneous.
- \(\mathrm{E^\circ = E^\circ_{ox}(Fe) + E^\circ_{red}(Fe^{3+}) = +0.44\, V + 0.77\, V = +1.21\, V}\).
- \(\mathrm{E^\circ_{ox}(Br^-) = -1.09\, V}\). An ion will oxidize \(\ce{Br-}\) if it has an \(\mathrm{E^\circ_{red} > +1.09\, V}\).
- \(\mathrm{E^\circ_{red}(Pb^{2+}) = -0.13\, V \Rightarrow}\) No.
- \(\mathrm{E^\circ_{red}(H^+) = 0 \Rightarrow}\) No.
- \(\mathrm{E^\circ_{red}(Au^{3+}) = +1.50\, V \Rightarrow}\) Yes.
- \(\mathrm{E^\circ_{red}(MnO_4^-) = +1.51\, V \Rightarrow}\) Yes.
Answer: \(\ce{Au^3+}\) and \(\ce{MnO4-}\)
-
- \(\mathrm{E^\circ_{ox}(Al) = +1.66\, V}\); \(\mathrm{E^\circ_{ox}(Pb) = +0.13\,V}\). \(\ce{Al}\) has a greater tendency to be oxidized. Thus, for spontaneous reaction, \(\ce{Al}\) will be oxidized and \(\ce{Pb}\) will be reduced:
\(\ce{2 Al(s) + 3 Pb^2+ (aq) \rightarrow 2 Al^3+ (aq) + 3 Pb(s)}\)
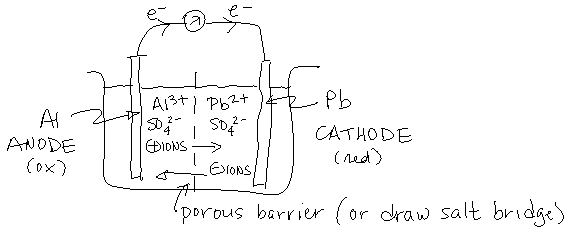
- lead (\(\ce{Pb}\)); since \(\ce{Pb^2+ \rightarrow Pb}\)
-
- Iron (\(\ce{Fe}\)) is the strongest reducing agent because it is always oxidized. It reduces all of the other metals.
- Gold (\(\ce{Au}\)) is the weakest reducing agent because it is never oxidized.
- Using the voltages of the other metals with \(\ce{Pb}\).
| Half-reaction | \(\mathrm{E^\circ_{red}}\) (volts) |
| \(\mathrm{Au^{3+} + 3 e^- \leftrightarrow Au}\) | + 0.80 V |
| \(\mathrm{Pb^{2+} + 2 e^- \leftrightarrow Pb}\) | 0.00 V |
| \(\mathrm{Ni^{2+} + 2 e^- \leftrightarrow Ni}\) | - 0.10 V |
| \(\mathrm{Fe^{2+} + 2 e^- \leftrightarrow Fe}\) | - 0.25 V |

