1.5 Conjugate Acid-Base Pairs
- Page ID
- 32055
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \) \( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)\(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\) \(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\)\(\newcommand{\AA}{\unicode[.8,0]{x212B}}\)
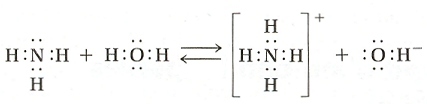
As you ponder the Brønsted-Lowry theory of acids and bases there are some important things to think about. Once again we'll examine ammonia:
 '
'
| Consider how \(\ce{NH3}\) changes to: \(\ce{NH4+}\) | \(\ce{NH3 -> NH4^{+}}\) |
The formulas differ by a single hydrogen; NH3 gains an H+ to become NH4+
| Consider how \(\ce{H2O}\) (or \(\ce{HOH}\)) changes to \(\ce{OH-}\): | \(\ce{HOH -> OH^{-}}\) |
Again the formulas differ only by a single hydrogen; \(\ce{H2O}\) lost a \(\ce{H+}\) forming \(\ce{OH-}\)
Now consider these two changes as reversible reactions. What if the reaction proceeds in the opposite direction:
| \(\ce{NH4+}\) can change back to \(\ce{NH3}\): | \(\ce{NH4^{+} -> NH3}\) |
| \(\ce{OH-}\) can change back into \(\ce{H2O}\): | \(\ce{OH^{-} -> HOH}\) |
Putting these observations together we see that:
- ammonia acts as a base because it can combine with a hydrogen ion. It's partner ammonium is now an acid, for it has a hydrogen ion that it can give up; once it does it is converted back into ammonia.
- water acts as an acid because it gives away a hydrogen ion to ammonia. Once it has lost the hydrogen ion and becomes hydroxide, the hydroxide in turn can act as a base and accept a hydrogen ion from ammonium.
What we have here are conjugate acid-base pairs
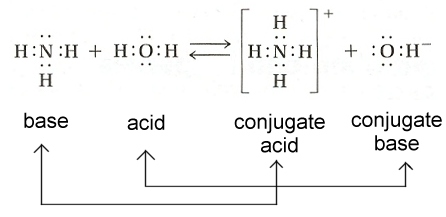
| Conjugate acid-base pairs differ from each other by the presence or absence of a single hydrogen ion (proton). Every acid has a conjugate base, and every base has a conjugate acid. |
The conjugates will always be listed on the product side of the reaction.

