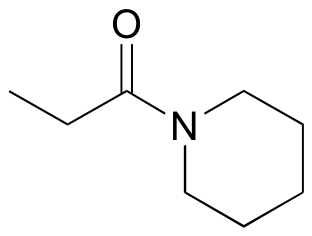
17.22: How Cells Activate Carboxylic Acids
- Page ID
- 16426
12.2A: Glutamine synthetase
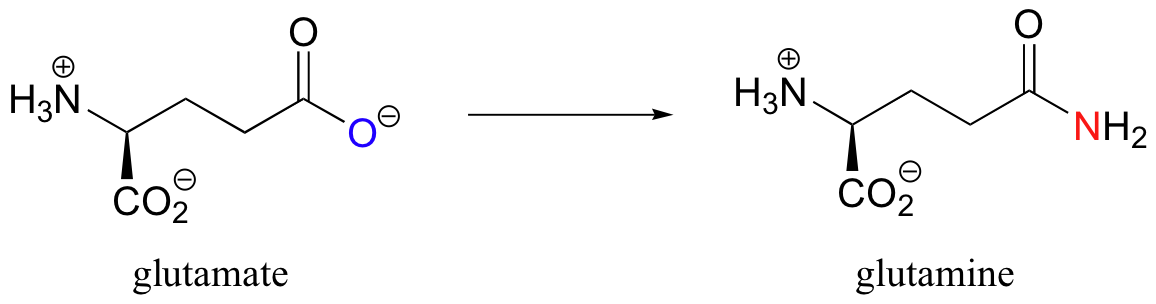
You have already learned that the carboxylate functional group is a very unreactive substrate for an enzyme-catalyzed acyl substitution reactions. How, then, does a living system accomplish an ‘uphill’ reaction such as the one shown below, where glutamate (a carboxylate) is converted to glutamine (an amide)?
It turns out that this conversion is not carried out directly. Rather, the first conversion is from a carboxylate (the least reactive acyl transfer substrate) to an acyl phosphate (the most reactive acyl transfer substrate). This transformation requires a reaction that we are familiar with from chapter 10: phosphorylation of a carboxylate oxygen with ATP as the phosphate donor.
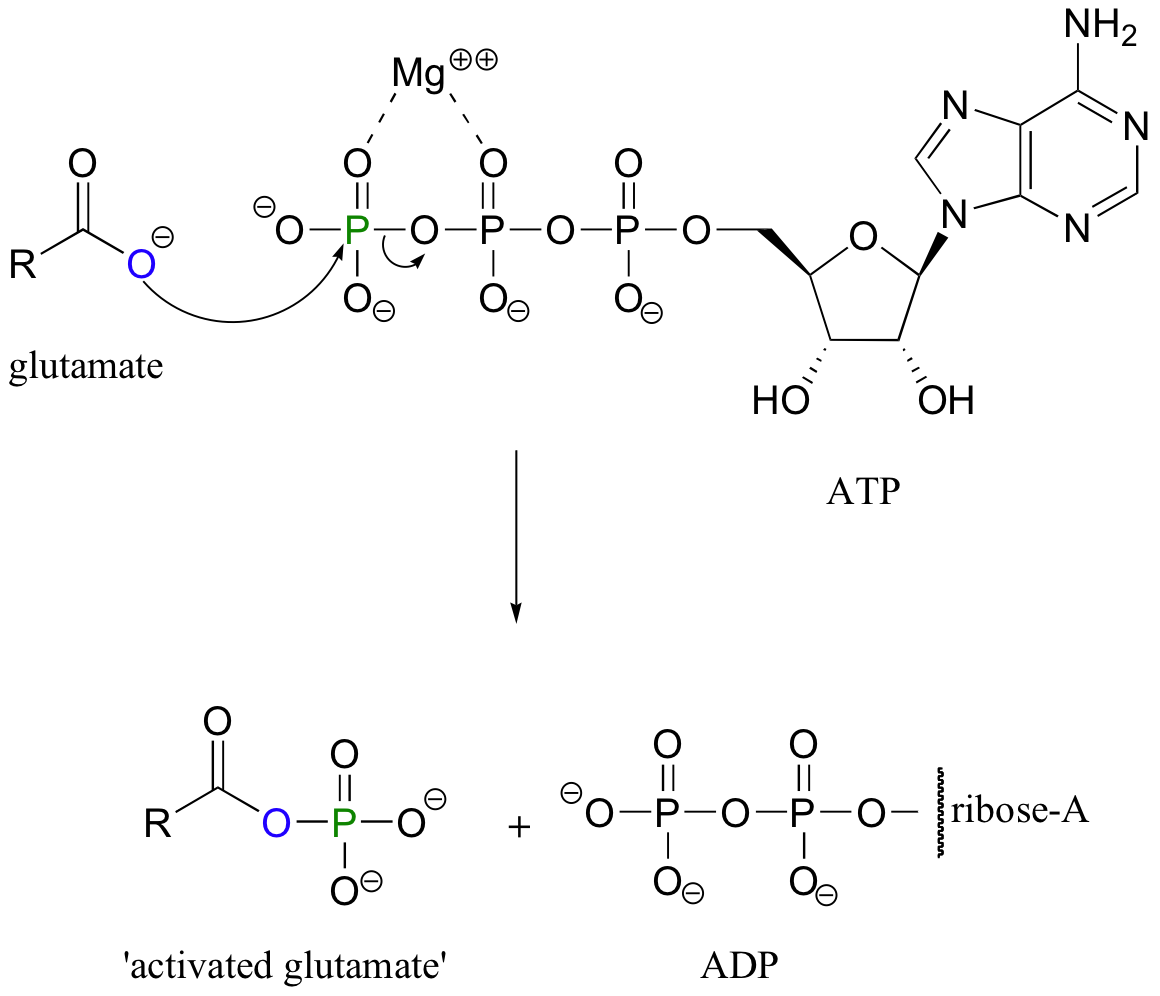
Note that this is just one of the many ways that ATP is used as a energy storage unit: in order to make a high energy acyl phosphate molecule from a low energy carboxylate, the cell must ‘spend’ the energy of one ATP molecule.
The acyl phosphate version of glutamate is now ready to be converted directly to an amide (glutamine) via a nucleophilic acyl substitution reaction, as an ammonia molecule attacks the carbonyl and the phosphate is expelled.
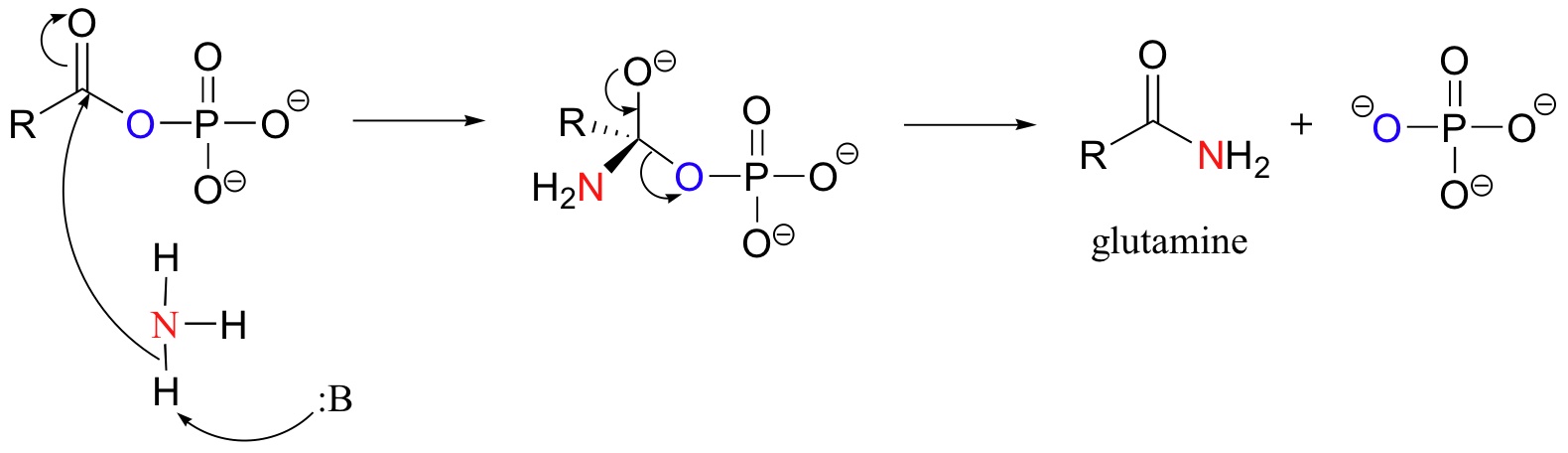
Overall, this reaction can be written as:

12.2B: Asparagine synthetase
Another common form of activated carboxylate group is an acyl adenosine phosphate. Consider another amino acid reaction, the conversion of aspartate to asparagine. In the first step, the carboxylate group of aspartate must be activated:
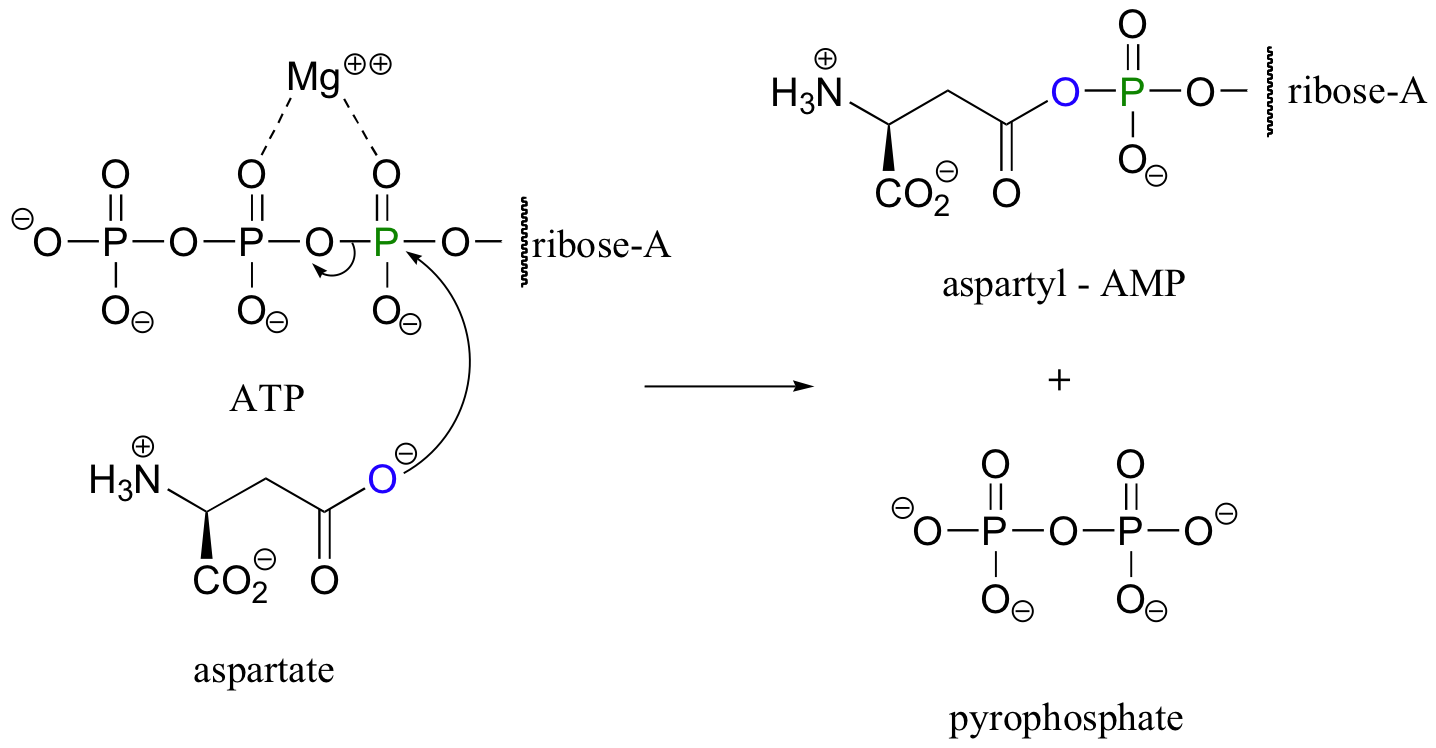
Once again, ATP provides the energy for driving the uphill reaction. This time, however, the activated carboxylate takes the form of an acyl adenosine (mono)phosphate. All that has happened is that the carboxylate oxygen has attacked the a-phosphate of ATP rather than the g-phosphate.
The reactive acyl-AMP version of aspartate is now ready to be converted to an amide (asparagine) via nucleophilic attack by ammonia. In the case of glutamine synthase, the source of ammonia was free ammonium ion in solution. In the case of asparagine synthase, the NH3 is derived from the hydrolysis of glutamine (this is simply another acyl substitution reaction):
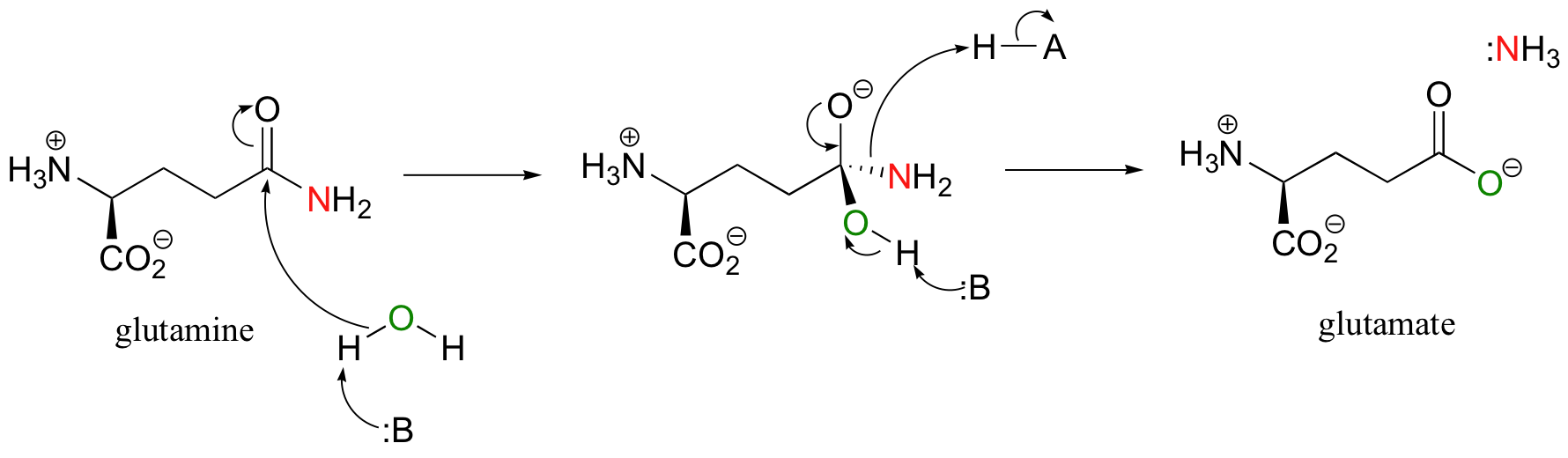
The hydrolysis reaction is happening in the same enzyme active site – as the NH3 is expelled in the hydrolysis of glutamine, it immediately turns around and acts as the nucleophile in the conversion of aspartyl-AMP to asparagine:
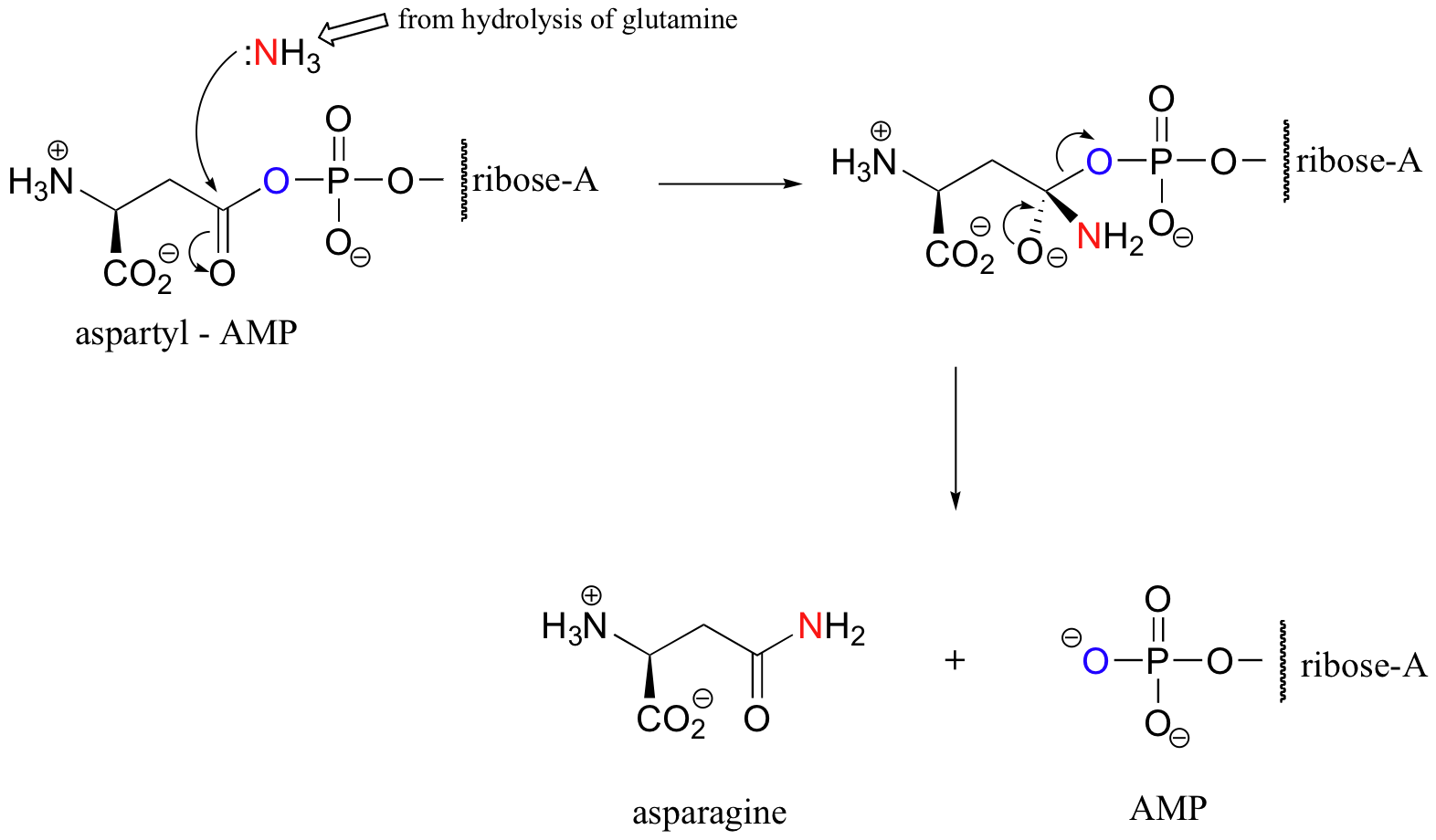
Keep in mind that the same enzyme is also binding ATP and using it to activate aspartate – this is a busy construction zone!
Overall, this reaction can be written in condensed form as:
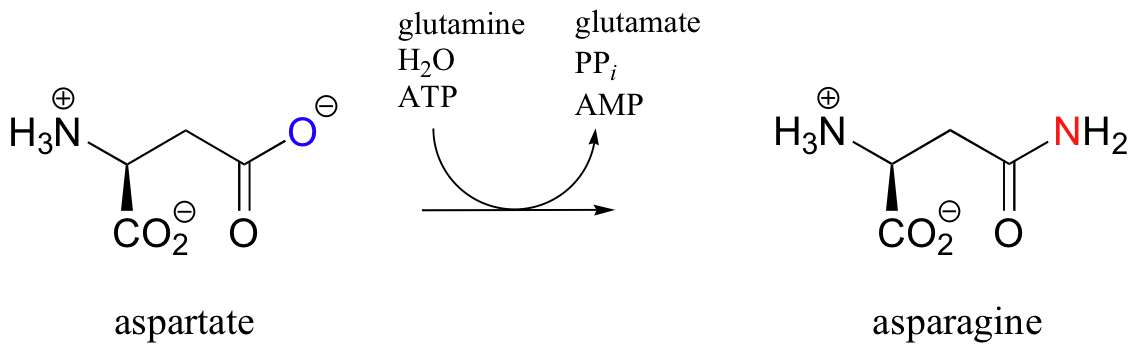
The use of glutamine as a ‘carrier’ for ammonia is a fairly common strategy in metabolic pathways. This strategy makes sense, as it allows cells to maintain a constant source of NH3 for reactions that require it, without the need for high solution concentrations of free ammonia.
12.2C: Glycinamide ribonucleotide synthetase
One of the early steps in the construction of purine bases (the adenine and guanine bases in DNA and RNA) involves an acyl substitution reaction with an acyl phosphate intermediate. In this case, the attacking nucleophile is not ammonia but a primary amine. The strategy, however, is similar to that of glutamine synthase. The carboxylate group on glycine is converted to an acyl phosphate, at the cost of one ATP molecule. The acyl group is then transferred to 5-phosphoribosylamine, resulting in an amide product.
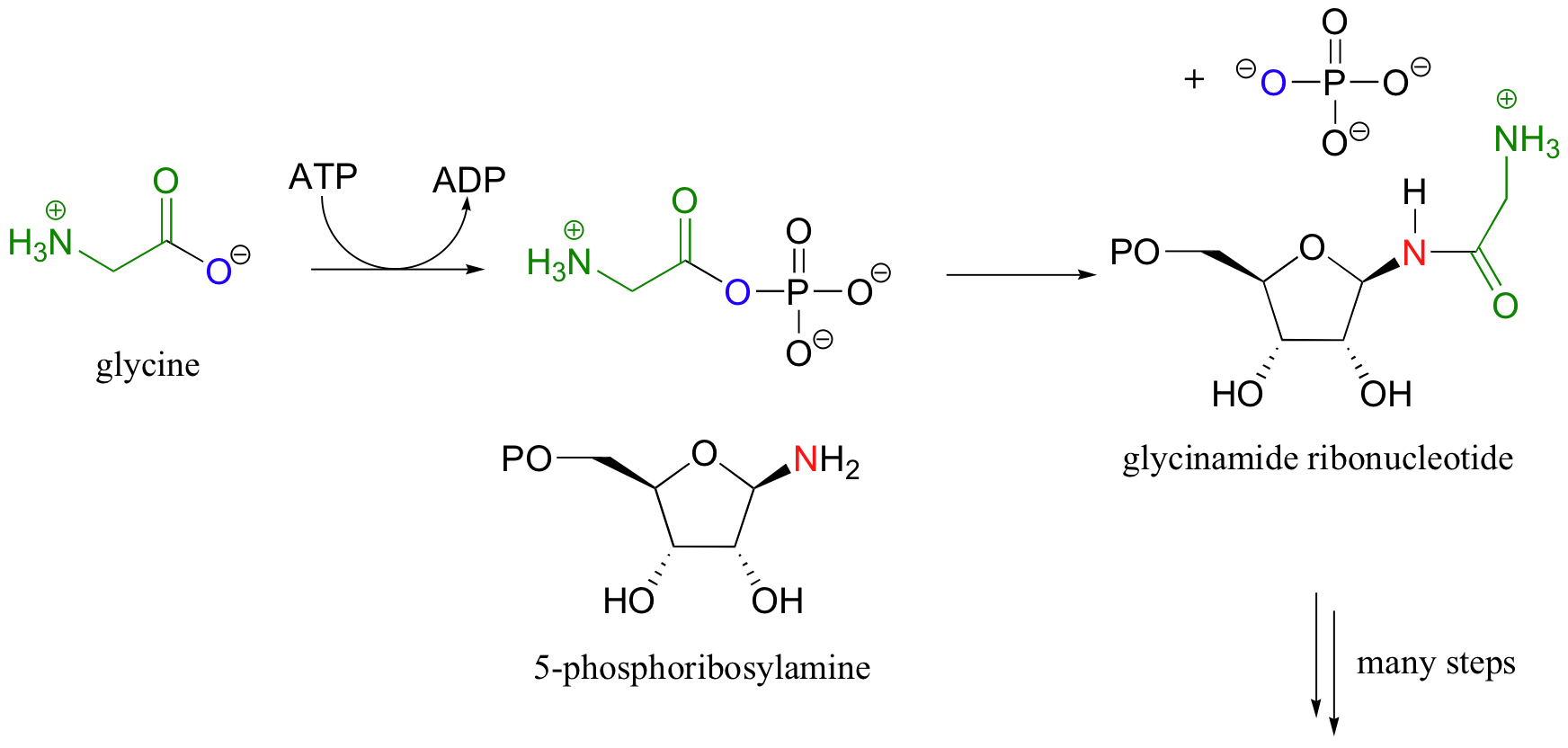
12.2D: Synthetic parallel - activated carboxylic acids in the lab
Just as enzymes need to activate carboxylate groups by first converting them to acyl phosphates, human chemists working in the lab have developed methods to create derivatives of carboxylic acids that are reactive in acyl substitution reactions. When treated with SOCl2 or PCl3, a carboxylic acid can be converted to an acid chloride, a derivative in which the acyl X group is a chloride ion. Note that these reactions are very similar to the reactions we saw in section 8.5B, in which alcohols were converted into alkyl chlorides or alkyl bromides by SOCl2 and PBr3, respectively.
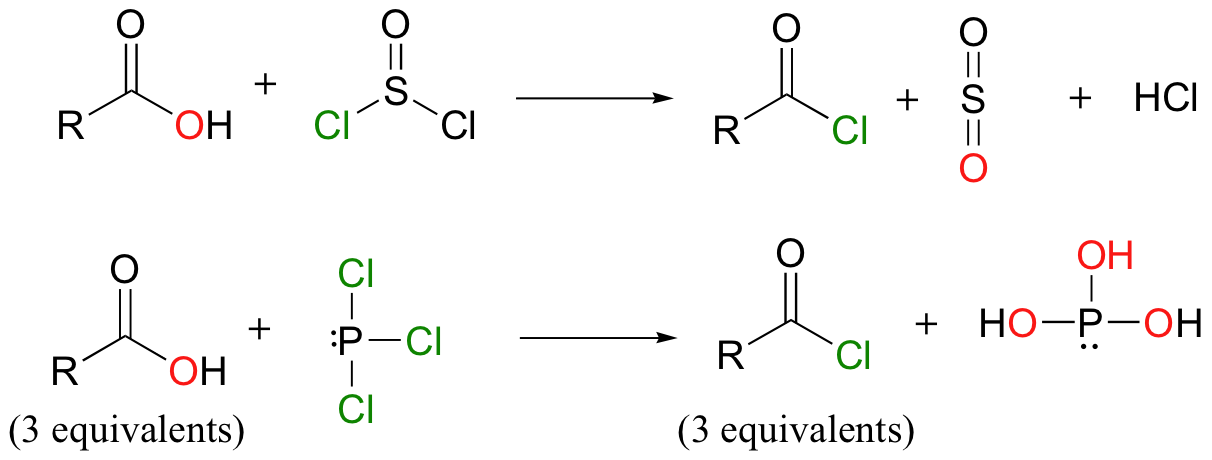
You will be asked to propose a mechanism for these transformations in the end-of-chapter problems.
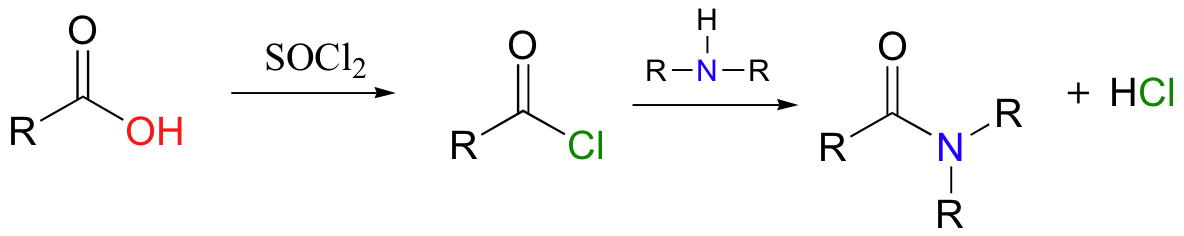
Acid chlorides are the laboratory equivalents of acyl phosphates in biochemistry, readily donating their acyl groups to form amides, esters, or other acyl compounds according to the nucleophile selected. The general example below shows the conversion of a carboxylic acid to an amide via an acid chloride intermediate.
Acid anyhydrides are also used as activated acyl groups in the lab, although they are less reactive than acid chlorides. Acetic anhydride is a convenient acetyl group donor, as shown in this synthesis of aspirin from salicylic acid (this is an easy transformation that is often carried out in undergraduate laboratory classes).

Exercise 12.3: Show how you might prepare the following compound, starting with a carboxylic acid and an amine.