PR1. Introduction to Pericyclic Reactions
- Page ID
- 4341
Pericyclic reactions differ from the ones we have looked at so far because they are not easily understood in Lewis acid- Lewis base terms. There is not always a clear nucleophile and electrophile in these reactions. In fact, they may appear to involve completely non-polar reactants. The classic example of a pericyclic reaction is a Diels Alder reaction. A Diels Alder reaction is a reaction between two alkenes.
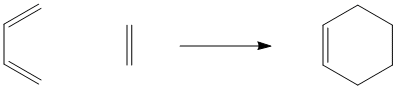
Figure PR1.1. A Diels Alder reaction between ethene and 1,3-butadiene.
Normally, we think of both of these compounds as nucleophiles. It isn't easy to see why one would react with the other. It isn't easy to see how electrons would be attracted from one molecule to the other.
Instead, pericyclic reactions rely on weak attractions between (or within) molecules that can lead to electronic interactions that result in new bond formation. Normally, pericyclic reactions are studied using molecular orbital calculations to map out these electronic interactions. They are also explained qualitatively using molecular orbital tools.


