Carboxylic Derivatives - Reactions at the Alpha-Carbon
- Page ID
- 1218
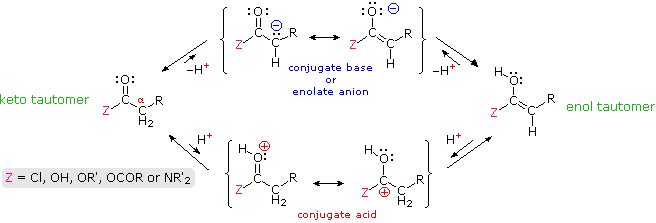
Many aldehydes and ketones were found to undergo electrophilic substitution at an alpha carbon. These reactions, which included halogenation, isotope exchange and the aldol reaction, take place by way of enol tautomer or enolate anion intermediates, a characteristic that requires at least one hydrogen on the α-carbon atom. In this section similar reactions of carboxylic acid derivatives will be examined. Formulas for the corresponding enol and enolate anion species that may be generated from these derivatives are drawn in the following diagram.
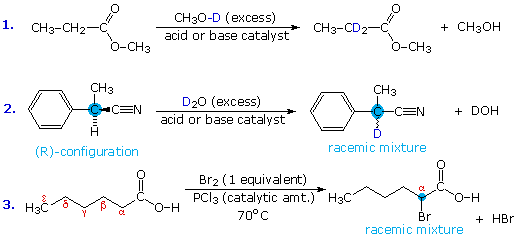
Acid-catalyzed alpha-chlorination and bromination reactions proceed more slowly with carboxylic acids, esters and nitriles than with ketones. This may reflect the smaller equilibrium enol concentrations found in these carboxylic acid derivatives. Nevertheless, acid and base catalyzed isotope exchange occurs as expected; some examples are shown in equations #1 and #2 below. The chiral alpha-carbon in equation #2 is racemized in the course of this exchange, and a small amount of nitrile is hydrolyzed to the corresponding carboxylic acid.
Acyl halides and anhydrides are more easily halogenated than esters and nitriles, probably because of their higher enol concentration. This difference may be used to facilitate the alpha-halogenation of carboxylic acids. Thus, conversion of the acid to its acyl chloride derivative is followed by alpha-bromination or chlorination, and the resulting halogenated acyl chloride is then hydrolyzed to the carboxylic acid product. This three-step sequence can be reduced to a single step by using a catalytic amount of phosphorus tribromide or phosphorus trichloride, as shown in equation #3. This simple modification works well because carboxylic acids and acyl chlorides exchange functionality as the reaction progresses. The final product is the alpha-halogenated acid, accompanied by a trace of the acyl halide. This halogenation procedure is called the Hell-Volhardt-Zelinski reaction.
To see a mechanism for the acyl halide-carboxylic acid exchange click the "Show Mechanism" button.
In a similar fashion, acetic anhydride serves as a halogenation catalyst for acetic acid (first equation below). Carboxylic acids that have a higher equilibrium enol concentration do not need to be activated for alpha-halogenation to occur, as demonstrated by the substituted malonic acid compound in the second equation below. The enol concentration of malonic acid (about 0.01%) is roughly ten thousand times greater than that of acetic acid. This influence of a second activating carbonyl function on equilibrium enol concentrations had been noted earlier in the case of 2,4-pentanedione.
|
CH3-CO2H + Br2 & (CH3CO)2O catalyst |
heat |
BrCH2CO2H + HBr |
|---|
(ii) |
RCH(CO2H)2 + Br2 |
 |
RCBr(CO2H)2 + HBr |
|---|
Contributors
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry


