12.2: Families of Organic Molecules - Functional Groups
- Page ID
- 86259
- Identify and describe functional groups in organic molecules.
Organic molecules can be classified into families based on structural similarities. Within a family, molecules have similar physical behavior and often have predictable chemical reactivity. The structural components differentiating different organic families involve specific arrangements of atoms or bonds, called functional groups. If you understand the behavior of a particular functional group, you can describe the general properties of that class of compounds.
The simplest organic compounds are in the alkane family and contain only carbon–carbon and carbon–hydrogen single bonds but do not have any specific functional group. Hydrocarbons containing at least one carbon–carbon double bond, (denoted C=C), are in the alkene family. Alkynes have at least one carbon–carbon triple bond (C≡C). Both carbon–carbon double bonds and triple bonds chemically react in specific ways that differ from reactions of alkanes and each other, making these specific functional groups.
In the next few chapters, we will learn more about additional functional groups that are made up of atoms or groups of atoms attached to hydrocarbons. Being able to recognize different functional groups will help to understand and describe common medications and biomolecules such as amino acids, carbohydrates, and fats. Table \(\PageIndex{1}\) and Figure \(\PageIndex{1}\) below list several of the functional groups to become familiar with as you learn about organic chemistry.
| Family Name |
Functional Group Structure |
Simple Example Structure | Simple Example Name | Name Suffix |
|---|---|---|---|---|
| alkane | none | CH3CH2CH3 | propane | -ane |
| alkene | 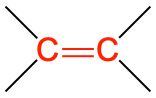 |
H2C=CH2 | ethene (ethylene) | -ene |
| alkyne |  |
HC≡CH | ethyne (acetylene) | -yne |
| aromatic | 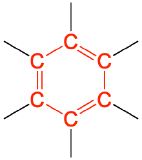 |
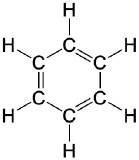 |
benzene | none |
| alkyl halide | 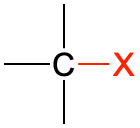 (X = F, Cl, Br, I) (X = F, Cl, Br, I) |
CH3CH2Cl | chloroethane | none |
| alcohol | 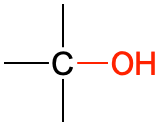 |
CH3CH2OH | ethanol | -ol |
| ether |  |
CH3CH2–O–CH2CH3 | diethyl ether | none* |
| amine | 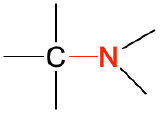 |
CH3CH2NH2 | ethylamine | -amine |
| aldehyde | 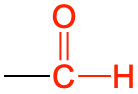 |
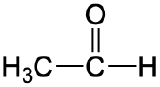 |
ethanal | -al |
| ketone | 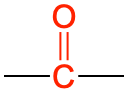 |
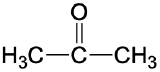 |
propanone (acetone) | -one |
| carboxylic acid | 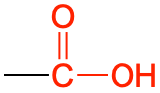 |
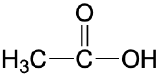 |
ethanoic acid (acetic acid) | -oic acid |
| anhydride | 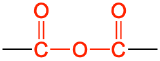 |
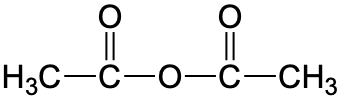 |
acetic anhydride | none |
| ester | 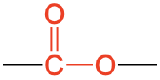 |
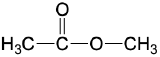 |
methyl ethanoate (methyl acetate) | -ate |
| amide | 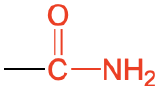 |
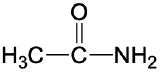 |
acetamide | -amide |
| thiol |  |
CH3CH2SH | ethanethiol | -thiol |
| disulfide |  |
CH3S–SCH3 | dimethyl disulfide | none |
| sulfide |  |
CH3CH2SCH3 | ethyl methyl sulfide | none |
|
Atoms and bonds in red indicate the functional group. Bonds not specified are attached to R groups (carbons and hydrogens). *Ethers do not have a suffix in their common name; all ethers end with the word ether. |
||||
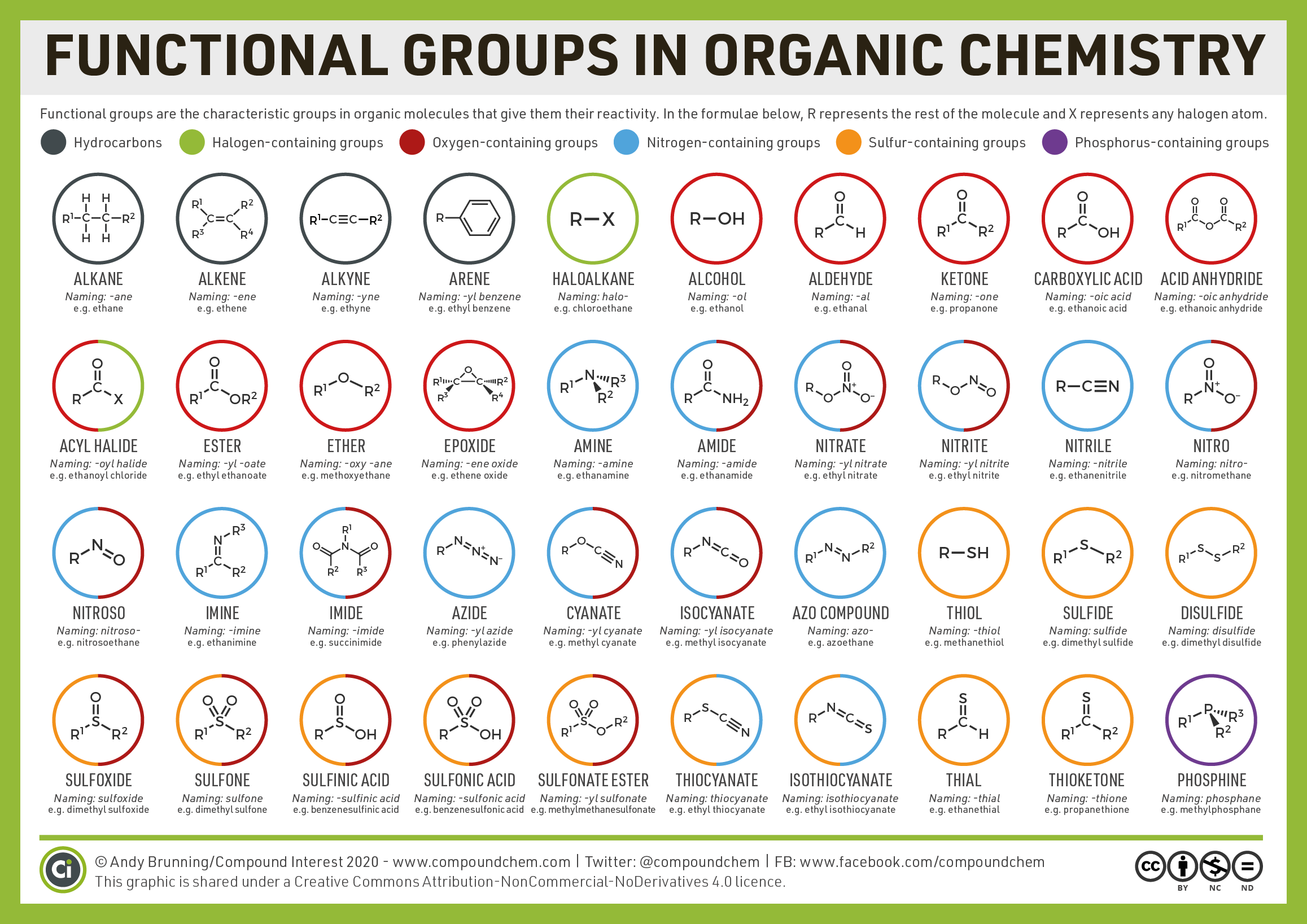
Figure \(\PageIndex{1}\): Functional groups in organic chemistry. (CC BY-NC-ND, CompoundChem.com).


