25.10: Ethers
- Page ID
- 54002
Race car drivers are always looking for that "edge" that will make their car (legally) faster than their competitors' cars. One way to get more speed is to burn the gasoline in the car engine more efficiently. Methyl-t-butyl ether (MTBE) has been used for this purpose, but is being discontinued due to concerns about the contamination of drinking water by leaking fuel tanks that contain this compound.
Ethers
An ether is an organic compound in which two hydrocarbon groups are bonded to the same atom of oxygen. An ether is represented by the general formula \(\ce{R-O-R'}\). The \(\ce{R'}\) in the formula means that the hydrocarbon group can be the same as \(\ce{R}\), or it can be different. The steps for naming ethers are listed below.
- The parent compound is given by the word ether, which appears at the end of the name.
- The names of each alkyl group come before the word ether. If the two alkyl groups are the same, the prefix di- is used. If the two alkyl groups are different, they are listed in alphabetical order.
- Spaces are left between the names of the alkyl groups (if different) and before the word ether.
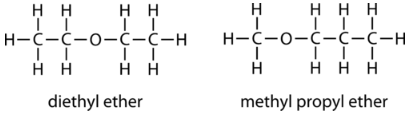
Shown below are two examples of ethers with their IUPAC names.
Properties of Ethers
Like alcohols, ethers are also quite soluble in water. The lone-pair electrons on the oxygen atom of the ether can form a hydrogen bond with the hydrogen atoms of water molecules. As with alcohols, the solubility is greater for ethers that have shorter length \(\ce{R}\) groups. The boiling points of ethers are much lower than the boiling points of alcohols. Ether molecules do not have hydrogen atoms that are covalently bonded to a highly electronegative atom, and so ether molecules cannot form hydrogen bonds with each other. The weaker intermolecular force acting between ether molecules results in boiling points which are much closer to alkanes of similar molar mass than to alcohols.
The anesthetic effects of ethers were first discovered in the 1840s. Diethyl ether was used as a general anesthetic for patients undergoing surgery for many years. However, ethers are very flammable and have undesirable side effects, such as nausea and vomiting. Safer alternatives to ethers are now used in anesthesia, and the primary use of ethers today is as a solvent for other organic compounds.
Summary
- An ether is an organic compound in which two hydrocarbon groups are bonded to the same atom of oxygen.
- IUPAC naming rules for ethers are dictated.
- Ethers are quite soluble in water.
- The primary use of ethers today is as a solvent for other organic compounds.

