21.5: Brønsted-Lowry Acids and Bases
- Page ID
- 53934
The Arrhenius concept of acids and bases was a significant contribution to the scientific understanding of acids and bases. It replaced and expanded Lavoisier's original idea that all acids contained oxygen. However, the Arrhenius theory also had its shortcomings. It did not take into account the role of the solvent. In addition, this concept did not deal with acid-base behavior in solvents such as benzene, where there could be no ionization. So, formation of a new theory was imperative, which built on the findings of Arrhenius, but also went beyond them.
Brønsted-Lowry Acids and Bases
The Arrhenius definition of acids and bases is somewhat limited. There are some compounds whose properties suggest that they are either acidic or basic, but which do not qualify according to the Arrhenius definition. An example is ammonia \(\left( \ce{NH_3} \right)\). Its aqueous solution turns litmus blue, it reacts with acids, and displays all the other properties of a base. However, it does not contain the hydroxide ion. In 1923, a broader definition of acids and bases was independently proposed by Danish chemist Johannes Brønsted (1879-1947) and English chemist Thomas Lowry (1874-1936). A Brønsted-Lowry acid is a molecule or ion that donates a hydrogen ion in a reaction. A Brønsted-Lowry base is a molecule or ion that accepts a hydrogen ion in a reaction. A hydrogen ion is commonly referred to as a proton, and so acids and bases are proton donors and proton acceptors, respectively, according to the Brønsted-Lowry definition. All substances that are categorized as acids and bases under the Arrhenius definition are also defined as such under the Brønsted-Lowry definition. The new definition, however, includes some substances that are left out according to the Arrhenius definition.
What kind of molecule would qualify as a Brønsted-Lowry base? These molecules need to be able to accept a hydrogen ion (or proton). Two possibilities come to mind: an anion that can form a neutral compound with a proton, or a molecule in which one or more atoms has lone-pair electrons. The most obvious anion is the Arrhenius base \(\ce{OH^-}\). This ion can form a water molecule with a proton by accepting the proton. The acetate anion \(\ce{CH_3COO^-}\) is another anion that can combine with a proton to form acetic acid. Lone-pair electron groups would include the nitrogen atom (see figure below). The two electrons at the top of the nitrogen atom are not connected in any type of bond, but they do interact readily with a bare proton.
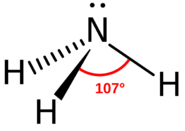
Oxygen is another atom with lone pair electrons that can function as a Brønsted-Lowry base.

The two single electrons (to the left and bottom of the atom) can form single covalent bonds with other atoms, while the two pairs of double electrons (top and right) are available to interact with a hydrogen ion.
Summary
- A Brønsted-Lowry acid is a molecule or ion that donates a hydrogen ion in a reaction.
- A Brønsted-Lowry base is a molecule or ion that accepts a hydrogen ion in a reaction.

