1.7: Medicine
- Page ID
- 52290
How has chemistry made a contributions to medicine?
Diabetes mellitus is a disease characterized by the body's inability to use glucose (a component of table sugar). Glucose is needed to provide biochemical energy for all the cells of the body. When the body cannot make energy using glucose, it begins to break down fat and protein to provide the needed energy, eventually leading to death. Diabetes is the result of the pancreas losing the ability to make insulin, a protein that helps glucose to enter the body's cells and be used for biochemical energy. A key piece of the puzzle surrounding our understanding of diabetes came when Frederick Sanger, a British biochemist, carried out experiments that gave him the structure of the insulin molecule. Sanger used basic chemistry techniques and reactions, and took twelve years to complete his research. Today, automated instruments based on his approach can perform the same analysis in a matter of days. Sanger was awarded the Nobel Prize in Chemistry in 1958 for his insulin research.
Chemistry in Medicine
Major contributions to health care have been made by chemistry. The development of new drugs involves chemical analysis and synthesis of new compounds. Many recent television programs advertise the large number of new drugs produced by chemists.
The development of a new drug for any disease is long and complicated. The chemistry of the disease must be studied, as well as how the drug affects the human body. A drug may work well in animals, but not in humans. Out of one hundred drugs that offer the possibility of treating disease, only a small handful actually turn out to be both safe and effective.
Chemistry contributes to the preparation and use of materials for surgery (sutures, artificial skin, and sterile materials). The sutures used in many surgeries today do not have to be removed, because they simply dissolve in the body after a period of time. Replacement blood vessels for heart and other types of surgery are often made of chemicals that do not react with the tissues, so they will not be rejected by the body. Artificial skin can be used to replace human skin for burn patients.
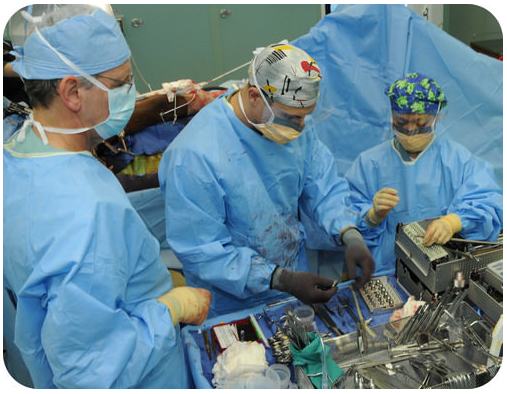
Clinical laboratory testing uses a wide variety of chemical techniques and instrumentation for analysis. Clinical laboratory testing allows us to answer commonly asked questions such as "is your cholesterol high?" and "do you have diabetes?" Some laboratory tests use simple techniques. Other processes involve complex equipment and computer analysis data, in order to perform measurements on large numbers of patient samples.
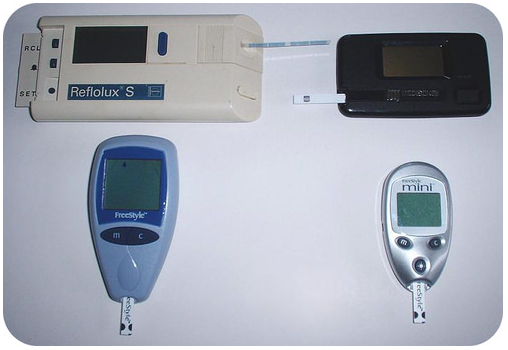
Laboratory testing has come to the local drug store or grocery store because of developments in chemistry. You can test your blood glucose using a simple portable device that runs a chemical test on the blood sample and tells you how much glucose is present, allowing a diabetic patient to regulate how much insulin to administer (chemistry is also used to produce the insulin and the disposable syringe that administers the drug).
How useful is worm spit? It turns out that worm spit, also known as silk, is a very useful material in medicine. In this video by Science Friday, Dr. David Kaplan describes how silk is used in a variety of medical applications.
Summary
- Chemistry finds many applications in the healthcare field.
- Development of medicines involves many complicated chemistry processes.
- Chemistry is used to create materials used in surgery.
- Much of laboratory testing is based on chemistry techniques.
Review
- What chemical is missing in the diabetic patient?
- Who discovered the structure of insulin?
- What two things need to be studied to develop a new drug?
- List two areas where chemistry has helped surgical patients
- What blood test can be performed using material purchased from your local drugstore?

