20.13: The Amino Acids
- Page ID
- 49621
Altogether there are 20 amino acids which commonly occur in all organisms. Under most circumstances amino acids exist as zwitterions and have the general formula
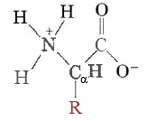
R represents a group called a side chain which varies from one amino acid to another. One aspect of amino acid structure not obvious in the above representation is the stereochemistry. Notice in the above image the carbon marked with an α, has 4 different groups attached to it, and thus amino acids are chiral. One of the most familiar chiral objects are hands, and therefore chirality is often thought of in terms of handedness. Below are the two enantiomers of the amino acid alanine, L-alanine and R-alanine, so you can see the difference.
The two enantiomers of alanine, L-alanine and R-alanine. Try as you might, it is impossible put these two molecules in the exact same position, they are mirror images. The L-form is the isomer seen in organisms.
How do you tell the difference between the two enantiomers? While most chemists use the R and S priority system to distinguish between enantiomers, many biochemical compounds, including amino acids and sugars use the D and L system. This is based upon a historical method for determining enantiomers using glyceraldehyde. Glyceraldehyde is a chiral three carbon sugar. with one form classified as D and the other as L. Amino acids and sugars can be synthesized from glyceraldehyde. The enantiomer synthesized from the D-form of glyceraldehyde are also labeled D, while those from the L-form, L. To determine if an amino acid is L or D, look at the α carbon, so that the hydrogen atom is directly behind it. This should place the three other functional groups in a circle. Follow from COOH to R to NH2, or CORN. If this is in a counterclockwise direction, the the amino acid is in the L-isomer. If this order is in the clockwise direction, the amino acid is a D-isomer. Try this trick with the two models of alanine.
If you assigned priority and used the R,S system, you will find that most amino acids are S-isomers. There is one exception however, which is cysteine. the sulfur in the R group gives it priority over the carboxylic acid group. This it is an R-isomer in the R,S system, but an L-isomer in the D,L system.
Structures of the 20 different R groups are given in Figure \(\PageIndex{1}\), where they are shown as part of a polypeptide chain. The figure also shows the three-letter code and one-letter code often used to identify amino acids in a polypeptide chain.
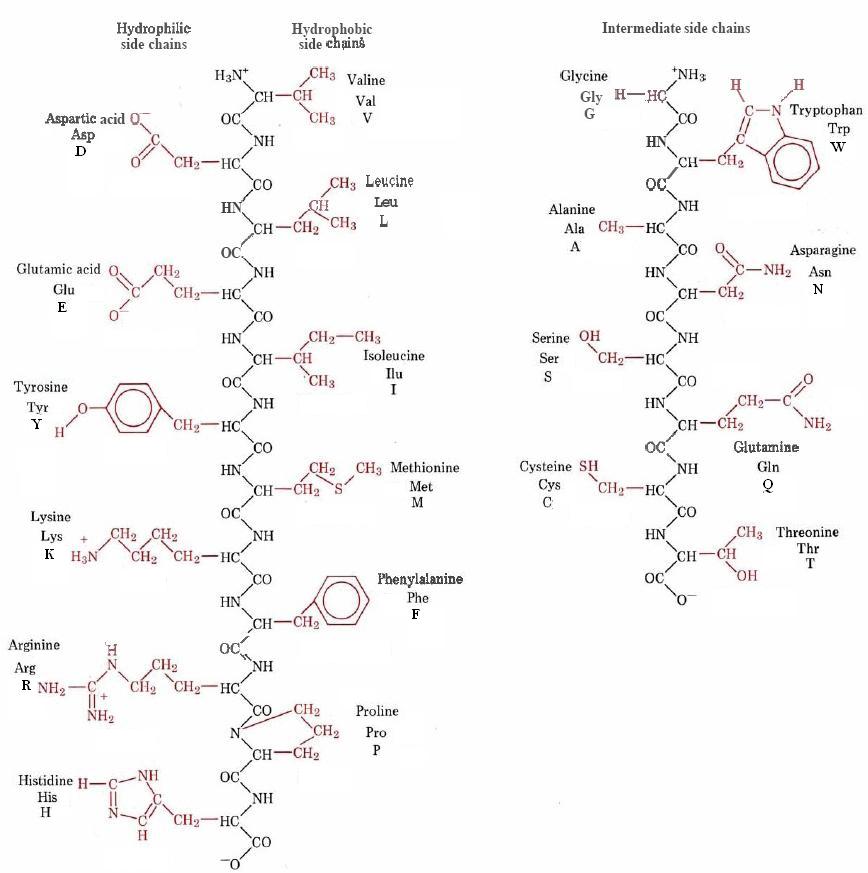
Although each amino acid side chain has its own individual properties, it is useful to divide them into several categories. In Figure \(\PageIndex{1}\) this has been done on the basis of how strongly hydrophilic or hydrophobic they are. On the extreme left of the figure are the six most hydrophilic side chains. All are polar, and four are actually ionic at a pH of 7. Next in the figure are the six most hydrophobic side chains. These are all large and nonpolar and contain no highly electronegative atoms like nitrogen and oxygen. The remaining eight side chains either contain small nonpolar groups or groups of fairly limited polarity, and are therefore not very strongly hydrophilic or very strongly hydrophobic.
Whether a side chain is hydrophilic or hydrophobic has considerable influence on the conformation of the polypeptide chain. Like the hydrocarbon tails of fatty acid molecules, hydrophobic amino acid side chains tend to avoid water and cluster together with other nonpolar groups. In a globular protein like trypsin for example, most hydrophobic residues are found among twisted chains deep within the molecule. Hydrophilic side chains, by contrast, tend to occupy positions on the outside surface, where they contact the surrounding water molecules. Hydrogen bonds and dipole forces attract these water molecules and help solubilize the globular protein. Were all the nonpolar R groups exposed to the aqueous medium, the protein would be much less soluble.


