20.10: Polysaccharides
- Page ID
- 49616
As the name suggests, polysaccharides are substances built up by the condensation of a very large number of monosaccharide units. Cellulose, for example, is a polymer of β-glucose, containing upwards of 3000 glucose units in a chain. Starch is largely a polymer of α-glucose.
These two substances are a classic example of how a minor difference in the monomer can lead to major differences in the macroscopic properties of the polymer. Good-quality cotton and paper are almost pure cellulose, and they give us a good idea of its properties. Cellulose forms strong but flexible fibers and does not dissolve in water. By contrast, starch has no mechanical strength at all, and some forms are water soluble. Part of the molecular structure of cellulose and starch are shown in Figure \(\PageIndex{1}\).
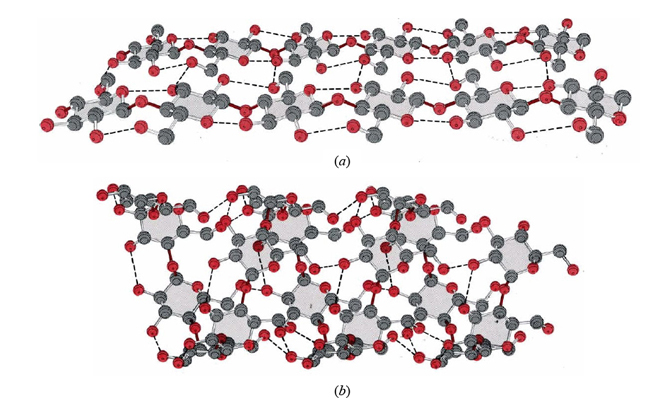
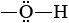 groups in the structure. As a consequence none are left exposed to hydrogen bond to solvent molecules except on the surface of the fiber. This explains why cellulose is insoluble in water.
groups in the structure. As a consequence none are left exposed to hydrogen bond to solvent molecules except on the surface of the fiber. This explains why cellulose is insoluble in water.
Cellulose and starch are different not only in overall structure and macroscopic properties. From a biochemical point of view they behave so differently that it is difficult to believe that they are both polymers of the same monosaccharide. Enzymes which are capable of hydrolyzing starch will not touch cellulose, and vice versa. From a plant’s point of view this is just as well since cellulose makes up structural material while starch serves as a storehouse for energy. If there were not a sharp biochemical distinction between the two, the need for a bit more energy by the plant might result in destruction of cell walls or other necessary structural components.
Bacteria, protozoa, termites, some cockroaches, and ruminant mammals (cattle, sheep, etc.) are capable of digesting cellulose. Ruminant mammals, termites and cockroaches themselves do not produce cellulase, the enzyme which breaks down cellulose, but rather, maintain a symbiotic relationship with bacteria in their guts which do breakdown the cellulose for digestion. Most organisms, including humans, are not capable of digesting cellulose, either through their own enzymes or through a symbiotic relationship with an organism which can. If our digestive enzymes could hydrolyze cellulose, humans would have available a much larger food supply. Quite literally we would be able to eat sawdust! It is possible to hydrolyze cellulose in the laboratory either with strong acid or with cellulase, the enzyme used by bacteria. So far, however, such processes produce more expensive (and less tasty!) food than we already have available.


