20.5: Nonpolar Lipids
- Page ID
- 49606
A good example of a nonpolar lipid is the neutral fat glycerol tristearate. This most-common form of animal fat serves as a storehouse for energy and as insulation against heat loss. On a molecular level it is constructed from three molecules of stearic acid and one of glycerol:
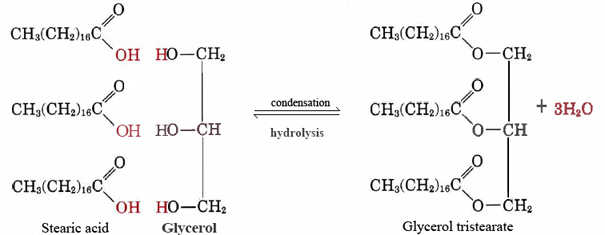 (1)
(1)
A great many nonpolar lipids can be made by combining different long- chain acids with glycerol. Because these acids were originally derived from fats, they are collectively referred to as fatty acids.
Notice that for each stearic or other fatty acid molecule that combines with one of the —OH groups of glycerol, a molecule of water is given off, and so the reaction is a condensation. It turns out that a great many important biological molecules are put together by condensation reactions during which water is given off. The reverse of Eq. (1), in which water reacts with a large molecule and splits it into smaller pieces, is called hydrolysis. By carrying out hydrolysis living organisms can break down molecules manufactured by other species. The simple building blocks obtained this way can then be recombined by condensation reactions to form structures appropriate to their new host.
By contrast with the glycerol tristearate found in animals, vegetable fats contain numerous double bonds in their long hydrocarbon chains. This polyunsaturation introduces “kinks” in the hydrocarbon chains because of the barrier to rotation and the 120° angles associated with the double bonds. Consequently it is more difficult to align the chains side by side (see Figure \(\PageIndex{1}\) ), and the unsaturated fats do not pack together as easily in a crystal lattice. As was true with alkanes, chain length also determines whether a fat is liquid or solid, and where the melting point occurs.
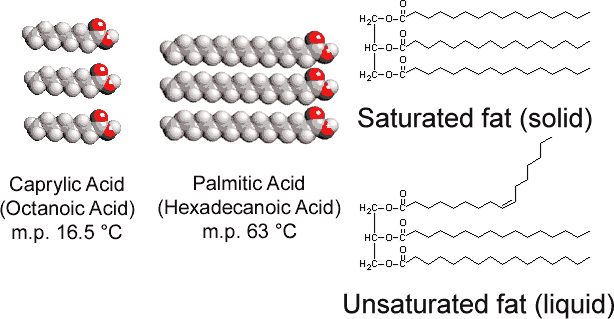
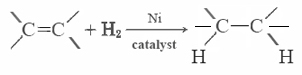
Most unsaturated fats (like corn oil) are liquids at ordinary temperatures, while saturated fats (like butter) are solids. Vegetable oils can be converted by hydrogenation to compounds that are solids. This process involves adding H2 catalytically to the double bonds:
Hydrolysis of fats [the reverse of Eq. (1)] is important in the manufacture of soaps. It can be speeded up by the addition of a strong base like NaOH or KOH, in which case the reaction is called saponification. Since saponification requires that the pH of the reaction mixture be high, the fatty acid that is produced will dissociate to its anion. When glycerol tristearate is saponified with NaOH for example, sodium stearate, a relatively water-soluble substance and a common soap, is formed.
The ability of soaps to clean grease and oil from soiled surfaces is a result of the dual hydrophobic-hydrophilic structures of their molecules. The stearate ion, for example, consists of a long nonpolar hydrocarbon chain with a highly polar —COO– group at one end.
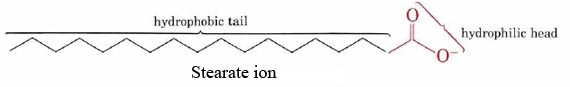
The hydrophobic hydrocarbon chain tries to avoid contact with aqueous media, while the anionic group readily accommodates the dipole attractions and hydrogen bonds of water molecules.
The two main ways that the hydrophobic portions of stearate ions can avoid water are to cluster together on the surface or to dissolve in a small quantity of oil or grease (see Figure \(\PageIndex{2}\) ).
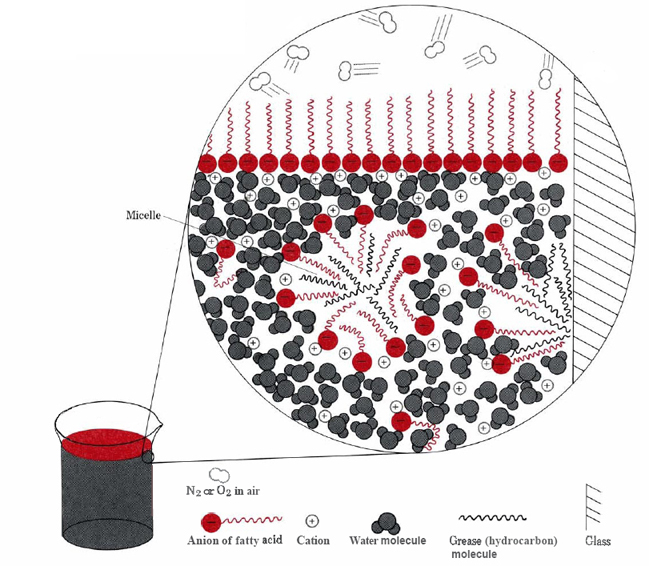
In the latter case the hydrophilic heads of the soap molecules contact the water outside the grease, forming a structure known as a micelle. Since the outsides of the micelles are negatively charged, they repel one another and prevent the grease droplets from recombining. The grease is therefore suspended (emulsified) in the water and can be washed away easily.
Natural soaps, such as sodium stearate, were originally made in the home by heating animal fat with wood ashes, which contained potash, K2CO3. Large quantities are still produced industrially, but to a considerable extent soaps have been replaced by detergents. This is a consequence of the undesirable behavior of soaps in hard water. Calcium, magnesium, and other hard-water cations form insoluble compounds when combined with the anions of fatty acids. This produces scummy precipitates and prevents the soap molecules from emulsifying grease unless a large excess is used.
Detergents such as alkylbenzenesulfonates (ABS) and linear alkylbenzenesulfonates (LAS) have structures very similar to sodium stearate except that the charged group in their hydrophilic heads is —SO3– attached to a benzene ring. The ABS detergents also have methyl (CH3) groups branching off their hydrocarbon chains.
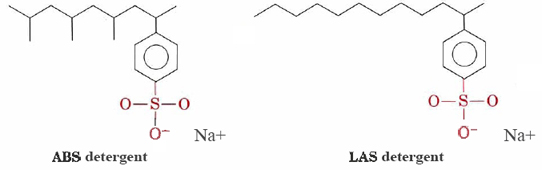
Such molecules do not precipitate with hard-water cations and therefore are more suitable for machine washing of clothes. The LAS detergents replaced ABS during the mid-nineteen-sixties when it was discovered that the latter were not biodegradable. They were causing rivers and even tap water to become covered with detergent suds and foam. Apparently the enzymes in microorganisms that had evolved to break down the unbranched hydrocarbon chains in natural fats and fatty acids were incapable of digesting the branched chains of ABS molecules. LAS detergents, though manufactured by humans, mimic the structures of naturally occurring molecules and are biodegradable.


