8.21: Diamond and Graphite
- Page ID
- 49473
The simplest example of a macromolecular solid is diamond. Crystals of diamond contain only carbon atoms, and these are linked to each other by covalent bonds in a giant three-dimensional network, as shown below. Note how each carbon atom is surrounded tetrahedrally by four bonds.
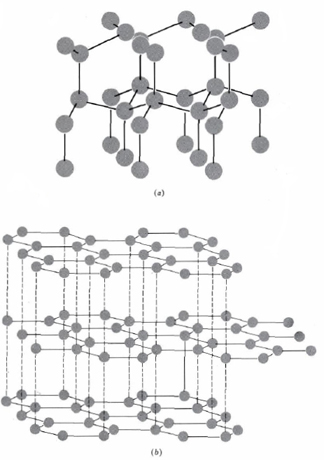
Such a network of carbon atoms extends throughout the crystal so that the whole diamond is one extremely large covalently bonded entity, i.e., a macromolecule.
Because strong covalent bonds, rather than London forces or dipole forces, hold the carbon atoms together in this crystal, it takes a great deal of energy to separate them. Accordingly, diamond has an extremely high melting point, 3550°C—much higher than any ionic solid. Diamond is also the hardest substance known. Each carbon atom is held firmly in its place from all sides and is thus very difficult to displace or remove.
Carbon also exists in a second, more familiar, crystalline form called graphite, whose crystal structure is also shown in part b of the figure. You use graphite every time you write with a pencil. (Pencil leads consist of C, not Pb!) The structure of graphite consists of flat layers. In each layer the carbon atoms are arranged in a regular hexagonal array. We can regard each layer as a large number of benzene rings fused together to form a gigantic honeycomb. All carbon-carbon bonds in this honeycomb are equivalent and intermediate in character between a single and a double bond.
While there are strong covalent bonds between the carbon atoms in a given plane, only weak London forces attract the planes together. The various layers can therefore slide past each other quite easily. When a pencil lead rubs across paper, the planes slide past each other and thin plates of crystal are left behind on the paper. These sliding plates also make graphite useful as a lubricant.
When an element can exist in more than one crystalline form, as carbon can in diamond and graphite, each form is said to be an allotrope. Other elements, such as sulfur and phosphorus, also form allotropes.


